Abstract
Pseudomyxoma peritonei (PMP) is a rare clinical entity characterized by widespread mucinous implants in the peritoneal cavity. Commonly seen in females in their 50s, PMP typically originates from ruptured appendiceal mucoceles that find refuge in the peritoneal space. Rarely, PMP may originate from the ovary, stomach, colon, or pancreas. Pseudomyxoma peritonei of colorectal origin is more malignant and has a lower survival rate. We report a case of a 59-year-old Hispanic woman with PMP who presented to the emergency room with a 3-month history of progressive abdominal distention. Pseudomyxoma peritonei was confirmed by computed tomography (CT) scan of the abdomen and pelvis and histopathology, and the patient underwent partial cytoreductive surgery. Given her Eastern Cooperative Oncology Group (ECOG) performance status of 1 despite extensive carcinomatosis, our patient may benefit from hyperthermic intraperitoneal chemotherapy (HIPEC) in the future.
Keywords
Introduction
Pseudomyxoma peritonei (PMP) is a rare neoplasm characterized by the dissemination of mucous ascites within the peritoneal cavity.1,2 These cancer metastases often originate from a ruptured appendiceal or ovarian neoplasm, and more rarely from the colon, stomach, or pancreas.3,4 Pseudomyxoma peritonei is more common in females around the ages of 50 to 59 years and is often an incidental finding during a laparotomy. 3 Long-term survival rates are poor, with a 5-year survival of 50% and a 10-year survival of 10% to 30%. 5
Pseudomyxoma peritonei is often asymptomatic until late in the disease course, when patients present with abdominal distention, ascites, abdominal mass, pelvic pressure, an acute abdomen, or new-onset hernia.2,3 Complete cytoreductive surgery (CRS) in combination with hyperthermic intraperitoneal chemotherapy (HIPEC) is now regarded as the cornerstone of therapy due to increased survival benefits2,6 compared with several drainage and debulking procedures done historically. 3 Be that as it may, recurrence rates are very high, even in patients with low-grade lesions. Herein, we present a rare case of PMP in a female patient with a surgical history of total hysterectomy with salpingo-oophorectomy.
Case Report
A 59-year-old Hispanic woman with a history of type 2 diabetes mellitus, hyperlipidemia, and fatty liver disease presented to the emergency department (ED) with a 1-day history of nausea and abdominal discomfort. She reported that her abdominal girth progressively increased over the last 3 months and caused her discomfort. She denied any vomiting, fever, chills, diarrhea, constipation, hematemesis, hematochezia, melena, weight loss, changes in her appetite, or early satiety. Notably, the patient had a surgical history of total hysterectomy with unilateral salpingo-oophorectomy due to menorrhagia.
In the ED, the patient was afebrile, and the rest of her vital signs were within normal limits. On examination, her abdomen was distended and showed a positive fluid wave. A palpable mass was first observed in the periumbilical region, the size of an adult fist, and the liver was grossly enlarged. However, there was no rebound tenderness, guarding, or rigidity. The remainder of the physical examination was unremarkable.
Laboratory results showed a hemoglobin level of 9.7 g/dL (normal range, 12-16 g/dL) and hemoglobin A1c of 6.2% (normal range, 4%-6%). The rest of the cell lines, liver enzymes, coagulation panel, and serum lipase levels were all unremarkable. Urinalysis and hepatitis panel results were also negative. Contrast-enhanced computed tomography (CT) scan of the abdomen and pelvis revealed large ascites with a mass effect, omental caking, and focal peritoneal soft tissue lesions, raising concerns of peritoneal carcinomatosis (Figure 1).

A computed tomography scan of the abdomen and pelvis showing liver scalloping (red arrows) and omental caking (black arrows) consistent with pseudomyxoma peritonei.
A tumor marker panel revealed an elevated carcinoembryonic antigen (CEA) level of 450 ng/mL (normal level: <3 ng/mL), a normal alpha fetoprotein level of 1 ng/mL (normal level: 0.5-9 ng/mL), an elevated cancer antigen (CA) 19-9 level of 1725 U/mL (normal level: 0-35 U/mL), and an elevated CA125 level of 93.9 U/mL (normal level: <38.1 U/mL).
Diagnostic paracentesis was attempted, which yielded 2 mL of mucinous ascites consistent with neoplastic etiology, but the ascitic fluid cytology was negative for malignancy. The patient underwent diagnostic laparoscopy (Figure 2), peritoneal biopsy, abdominal washout, and right subclavian chemoport placement. Peritoneal biopsy findings showed a low-grade mucinous neoplasia/carcinoma, consistent with PMP. Computed tomography scans of the chest and brain did not show any metastases. The patient was discharged with instructions for close follow-up with oncology and surgery for further treatment.

Intraoperative view: a jelly-like mass seen in the peritoneal cavity during a diagnostic laparotomy.
A month after discharge, the patient followed up with the oncology and surgical clinics for continued care. After discussing the biopsy results, the multidisciplinary tumor board reached a consensus to proceed with cytoreductive surgery (CRS), including the removal of the appendix. During the surgery, the patient was noted to have mucinous deposits and carcinomatosis of the entire abdomen and all 13 regions of the abdomen. The surgeons calculated a peritoneal cancer index (PCI) score of 24.
Exploratory laparotomy, evacuation of 2 L of mucinous ascites, abdominal washout, near-total omentectomy, and appendectomy were performed. Complete CRS was avoided as the risks outweighed the benefits. Residual tumor deposits could be seen at the top of the mesentery of the small bowel. The surgical pathology report was significant for a metastatic appendiceal low-grade mucinous neoplasm (Figure 3). The tumor involved the omentum, falciform ligament, peritoneum, and hernial sac. At the postoperative follow-up appointment with the surgical clinic, the patient reported feeling well and denied abdominal pain or changes in stool caliber. It has been 4 months since the patient underwent CRS and she continues to defer the chemotherapy. She may be a candidate for HIPEC in the future.
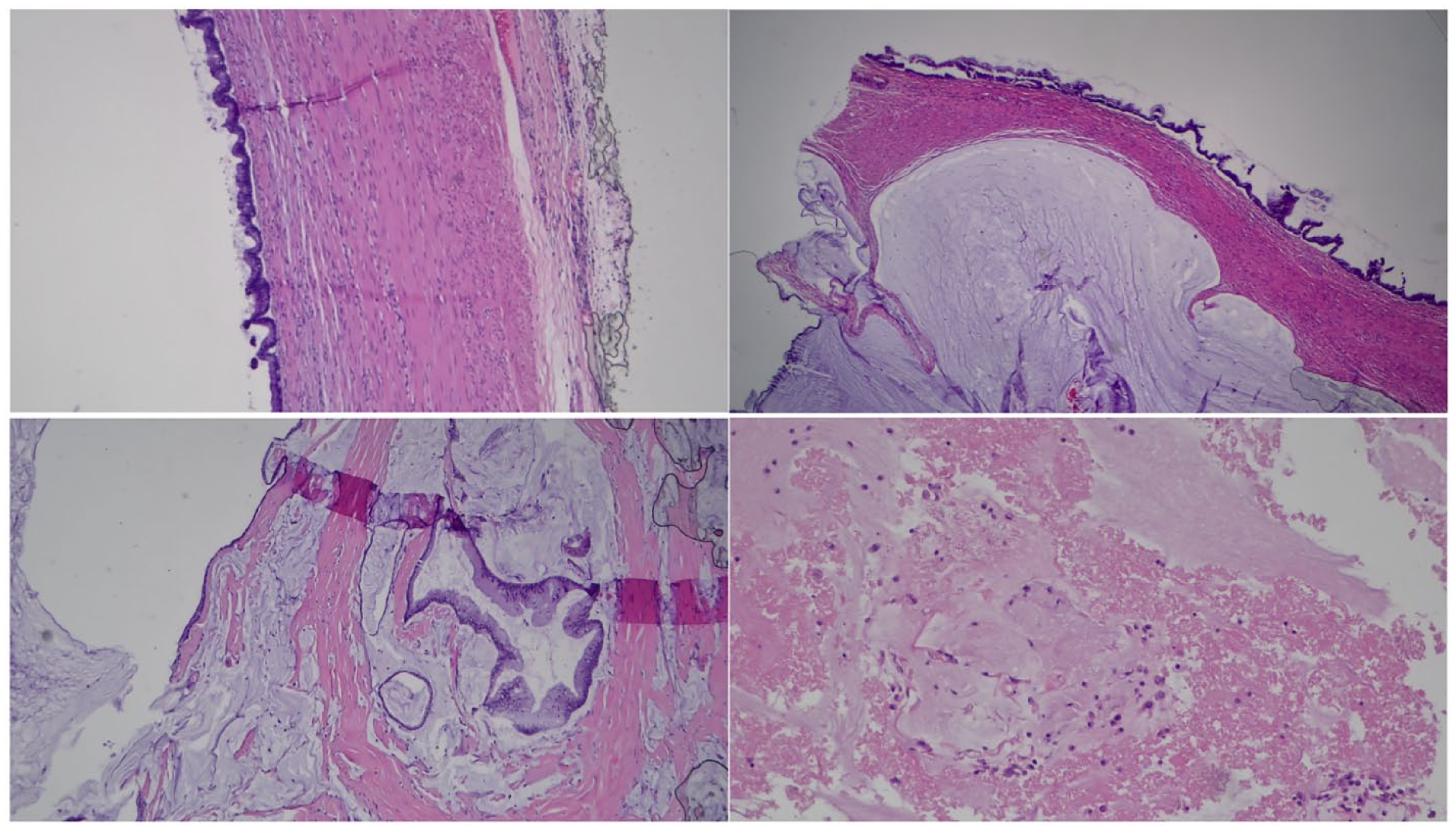
Histology slides showing abundant mucin dissecting through the fibroconnective tissue, with focal scattered fragments of mucinous glandular epithelium showing low-grade cytology. No infiltrative type of invasion or desmoplastic reactions are seen. The histological findings are most consistent with a low-grade mucinous neoplasia/carcinoma in the setting of pseudomyxoma peritonei.
Discussion
Pseudomyxoma peritonei is a rare medical condition involving thick, gelatinous mucinous accumulation within the peritoneal cavity.5-9 Its pathogenesis starts with rupture of the appendiceal mucinous tumor, which eventually finds refuge in the peritoneal space1,6 causing obstruction and fibrosis. Pseudomyxoma peritonei of ovarian origin is uncommon, and the few cases in the literature are associated with mature cystic teratomas. 7 Pseudomyxoma peritonei was previously known as “jelly belly” because of the mucinous nature of the ascites. 9 Pseudomyxoma peritonei incidence is estimated at 1 to 2 cases per million people per year,5,9 and it has female predominance.1,6,7,9
Pseudomyxoma peritonei nomenclature has evolved over the years 7 and so has its clinicopathological features. In 2010, PMP was classified into 2 subcategories by the American Joint Committee on Cancer (AJCC) and the World Health Organization (WHO) based on the grade of the epithelium in peritoneal mucin. 7 The 2 subtypes of PMP are low- and high-grade mucinous adenocarcinoma. 7 The WHO criteria were used to categorize 274 cases of PMP, and survival data was correlated with these categories. 7 The 5-year survival rate was 63% for low-grade mucinous adenocarcinoma and 23% for high-grade mucinous adenocarcinoma. 7 Low-grade lesions were typically of appendiceal origin, whereas some high-grade lesions were associated with colorectal adenocarcinomas. 7
Pseudomyxoma peritonei has a slow clinical course 1 but may present as abdominal pain, distended abdominal girth, or symptoms akin to acute appendicitis.7,9 Clinical symptoms in female patients may also include palpable ovarian masses and pelvic pressure.7,9 Serum tumor markers such as carbohydrate antigen (CA) 19-9 and CEA can aid in the diagnosis of PMP.1,7 These markers may also be used to monitor treatment response in patients undergoing CRS and HIPEC4,7 and as a measure of recurrence. 9 Liver scalloping, a radiologic sign made by the mass effect of the mucinous neoplasm against the liver and other viscera, is pathognomonic for PMP.1,2,4-7,10 In a retrospective study of 159 patients with PMP, Hotta et al 6 found that visceral scalloping on CT scans correlated with cancer recurrence after CRS. Identifying patients at risk of recurrence is crucial for selecting the appropriate treatment strategy. 7
Although not commonly indicated, magnetic resonance imaging (MRI) of the abdomen may show the extent of the cancer spread before surgical intervention. 1 Since most of the PMP cases are of appendiceal origin, colonoscopy is recommended as part of the workup for finding the primary tumor. 11 Diagnostic laparotomies are used as a minimally invasive tool to gain visuals on lesions undetected by cross-sectional imaging, such as CT, and better analyze tumor resectability.4,11 A PCI score is also calculated to quantify the extent of peritoneal spread of the tumor.4,11
Cytoreductive surgery combined with HIPEC is the optimal treatment for PMP in a curative setting, while tumor debulking is ideal in a palliative context. 1 Cytoreductive surgery and HIPEC are not without limitations and complications. Complete CRS may require extensive resections of the greater omentum or visceral peritoneum, which can lead to increased complications, decreased quality of life, 2 ileus, fistulas, gastroparesis, and abscesses. 4 Appendectomy is indicated even if the appendix appears normal or the tumor is of ovarian origin.7,9
Systemic chemotherapy, while an option for PMP treatment, has limited efficacy and is typically reserved for patients exhibiting progressive or recurrent disease symptoms. 7 Despite undergoing intensive treatment, the health of several patients tends to worsen over time, necessitating a shift in care focus toward palliative measures. 9 Small and frequent meals may alleviate early satiety, and anti-emetics can help with intractable nausea and vomiting. Bowel rest and adequate nutrition can be provided through intermittent or long-term parenteral alimentation. 9 Given end-of-life concerns, patients and their family members require sustained emotional support alongside medical management. 9 Further research is needed to establish optimal treatment strategies for PMP.
Conclusion
Pseudomyxoma peritonei is an uncommon neoplasm that manifests as disseminated mucinous ascites in the peritoneal cavity. Pseudomyxoma peritonei is more prevalent in females aged 50 to 59 years and often presents as abdominal pain, distended abdomen, pelvic pressure, or an acute abdomen. Ruptured appendiceal or ovarian neoplasms may be implanted in the peritoneal cavity, and excess mucinous ascites may cause visceral adhesions, obstructions, or fibrosis. Cytoreductive surgery and HIPEC are the gold standard for PMP but have higher recurrence rates. More research is needed to understand the pathogenesis of PMP and develop optimal therapies.
Footnotes
Author Contributions
L.B. and S.S.O. conceptualized the idea for this case report and wrote the manuscript. M.J., J.K., and S.A. helped write the paper. M.K. edited and proofread the final version of the manuscript. A.A. prepared the pathology slides and helped with the interpretations.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require IRB approval/waiver for case reports.
Informed Consent
The patient consented for the publication of this case report.
Data Availability Statement
Further enquiries can be directed to the corresponding author.
