Abstract
Stress cardiomyopathy is a transient left ventricular dysfunction caused by physiologic or pathologic stressors. Anaphylaxis is a hypersensitivity disorder that can lead to a rapid life-threatening respiratory collapse. It happens due to exposure to allergens including medications. During anaphylaxis, there is a compensatory release of catecholamines that can lead to stress cardiomyopathy. In this case, nab-paclitaxel infusion led to anaphylaxis with respiratory failure. Echocardiogram showed features of diffuse hypokinesis with preserved basal segment contractility, and cardiac catheterization did not show any evidence of obstructive coronary artery disease. The overall clinical picture suggested stress cardiomyopathy. The patient was treated with guideline-directed medical therapy which resulted in normalization of the ejection fraction with no symptoms of congestive heart failure at any point. The patient was thereafter resumed on a reduced dose of nab-paclitaxel. This case report adds to the spectrum of infusion-related reactions associated with paclitaxel and demonstrates the course of events in the management of anaphylaxis and stress cardiomyopathy in this scenario.
Introduction
Stress cardiomyopathy (SC) is characterized by sudden and transient onset of wall motion irregularities and akinesis involving the left ventricular apex. It is often precipitated by emotional or physical stressors and mimics the symptoms, electrocardiogram (EKG) manifestations, and troponin trends of acute coronary syndrome (ACS). However, unlike ACS, there is no significant evidence of obstructive coronary artery disease and a rapid return of left ventricular ejection fraction from 20%-49.9% to 59%-76% within a mean time of 7 to 37 days and an in-hospital mortality rate of 1.7%. 1
Despite it being an uncommon disease with a prevalence of 2% in patients presenting with the clinical manifestations of ACS, 2 there has been evidence of several classes of cardiotoxic chemotherapeutic agents being linked to SC in cancer patients. 3 Here we present a patient who developed SC during induction therapy with nab-paclitaxel.
Case Presentation
A 64-year-old woman with lung adenocarcinoma presented to the oncology infusion clinic for the first cycle of nab-paclitaxel and carboplatin. Prior to the infusion, she was premedicated with dexamethasone, diphenhydramine, and famotidine. Immediately after the initiation of nab-paclitaxel infusion, she started feeling severely short of breath, was found to be hypoxic with oxygen saturation (SpO2) dropping to 72%, and was placed on 15 L/min of supplemental oxygen via a non-rebreather mask. Blood pressure was 145/83 mm Hg, with normal heart rate and respiratory rate. She received 2 doses of 0.3 mg intramuscular epinephrine, intravenous dexamethasone, and diphenhydramine and was sent to the emergency room. Epinephrine was given in the anticipation of a hypotensive episode. She was unable to speak due to severe shortness of breath and was found to have accessory muscle retraction. On auscultation, there was bilateral inspiratory and expiratory wheezing with reduced air entry. She received another round of 0.3 mg intramuscular epinephrine, intravenous methylprednisone, diphenhydramine, and albuterol-ipratropium nebulization and was put on bilevel non-invasive positive pressure ventilation for oxygenation and increased work of breathing.
A 12-lead electrocardiogram showed sinus tachycardia with a heart rate of 115 beats per minute, with non-specific ST segment and T wave abnormalities in leads V1, V2, and V3 with a septal Q wave suggestive of prior septal infarction (Figure 1). Chest x-ray showed a previous area of malignancy with no interval change (Figure 2). In the next few minutes, she started feeling improvement in her breathing and was weaned off the bilevel ventilation with adequate saturation on ambient air. The complete blood count and comprehensive metabolic profile were unremarkable except for an elevated white blood cell count of 16 900/dL and high-sensitivity troponin (normal <14 ng/L) of 637, 901, and 846 ng/L checked at 3-hour intervals. A transthoracic echocardiogram (TTE) showed an ejection fraction of 25%, akinesis of the apical anterior segment, and hypokinesis of multiple segments including the entire septum and apex suggesting multivessel disease or stress-induced/Takotsubo cardiomyopathy (Figure 3).

EKG showing tachycardia with ST-segment abnormalities in the anterior leads and poor R wave progression in the anterior leads suggestive of a prior septal infarction.
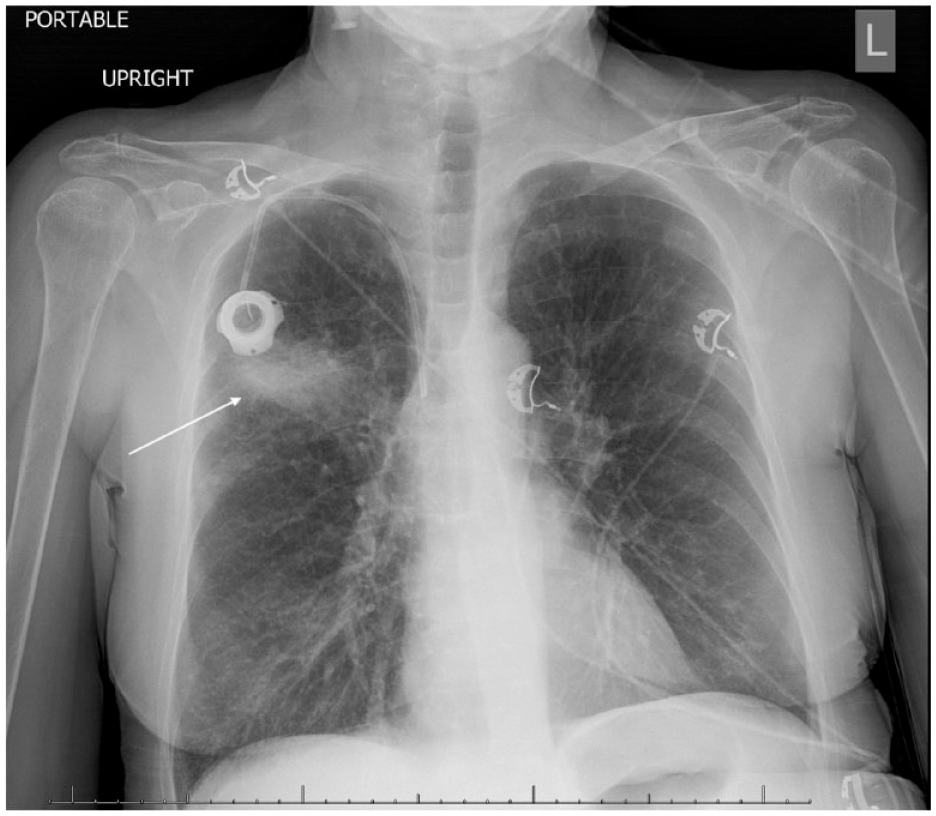
Posteroanterior chest radiograph showing opacification in the right upper lobe suggesting the previous area of malignancy (arrow) and hyperinflated lungs. These findings were unchanged from the previous chest x-rays.

Transthoracic echocardiogram with perfluten contrast in showing apical ballooning and diffuse hypokinesis with sparing of basal segments suggesting stress cardiomyopathy (arrows): (a) end-systolic view and (b) end-diastolic view.
Cardiovascular catheterization showed no significant obstructive disease. Hence, the cause of the cardiomyopathy was deemed to be SC. The patient’s condition stabilized over the next few days. She was discharged home on sacubitril-valsartan and metoprolol succinate as guideline-directed medical therapy for Takotsubo cardiomyopathy. Three months later, she was seen in the cardiology clinic, and a repeat TTE showed normalization of her left ventricular function with an ejection fraction of 60% and no wall motion abnormalities. The patient remained asymptomatic with no clinical features of congestive heart failure. Due to this previous infusion reaction, the chemotherapy regimen was modified with a lower dose of nab-paclitaxel and carboplatin.
Discussion
Stress cardiomyopathy is commonly seen in postmenopausal women and in most cases is attributable to physiologic or emotional stress. 4 Patients with malignancy are known to have a higher incidence of 10% as opposed to 1%-2% estimated in the general population. 5 Therefore, it is recommended to consider appropriate cancer screening in patients with SC. Furthermore, all-cause hospital mortality is significantly higher in chemotherapy-treated patients with SC versus those without an SC event. 6 The proposed mechanisms for SC include myocardial ischemia, left ventricular outflow tract obstruction, blood-borne catecholamine myocardial toxicity, epinephrine-induced switch in signal trafficking, and hyperactivation of the sympathetic nervous system. 7 Cancer is noted to accompany increased physiological stress and elevated sympathetic nervous tone. Paraneoplastic mediators, if associated, are noted to directly alter cardiac adrenoreceptors causing increased preponderance to the catecholamine response. Furthermore, various chemotherapies related to multiple hypersensitivity reactions can cause epinephrine surge leading to SC. 8
Clinical features of SC include dyspnea, acute chest pain, and tachyarrhythmias. Cardiac biomarkers such as troponin are known to be elevated without the presence of coronary artery occlusion. 7 Echocardiographic changes include left ventricular wall motion abnormality that extends beyond the coronary supply region which may be localized to the apical, midapical, midventricular, or basal segments of the left ventricle. 9 In this case, the echocardiogram showed akinesis of the anterior segment and hypokinesis of the apex.
Another differential to be considered in this scenario is Kounis syndrome which is associated with SC. It is described as a concurrence of acute coronary syndrome with anaphylactic or anaphylactoid reactions. The proposed mechanism is that the enormous amounts of epinephrine with heightened cardiovascular activity lead to endothelial injury, myocardial damage, and induction of adhesion molecules on the endothelial cells leading to significant coronary artery occlusion. 10 A scenario linking all the above differentials could be ATAK-adrenaline, Takotsubo, anaphylaxis, and Kounis syndrome. 11 Anaphylaxis-induced release of inflammatory cytokines, endogenous and exogenous epinephrine administration, and development of Takotsubo syndrome can perpetuate this vicious cycle and cause Kounis syndrome or worsen preexisting SC.
The chemotherapeutic agent most commonly reported with SC is 5-fluorouracil. Other reported associations are with paclitaxel, trastuzumab, bevacizumab, and rituximab. 12 Both platins and taxanes are related to a variety of infusion-related reactions (IRR). The IRRs to paclitaxel can be IgE mediated, due to mast cell degranulation, or complement activation by Cremophor EL (compounded with paclitaxel). 13 Nab-paclitaxel is devoid of Cremophor EL and therefore has resulted in the reduction but not complete elimination of IRRs. Premedication with glucocorticoids and antihistamines is advised. In the event of an IRR with paclitaxel, it is not discontinued for future cycles; rather it is either reintroduced again or desensitization is provided in the event of a severe reaction. 13
The management of Takotsubo cardiomyopathy involves standard guideline-directed medical therapy for heart failure with reduced ejection fraction which involves beta-blocker and angiotensin-converting enzyme inhibitor (or angiotensin receptor blocker) with or without angiotensin receptor/neprilysin inhibitor with fluid status management with loop diuretics if needed. 14 This patient was started on similar therapy for SC which would have contributed to the improvement in the ejection fraction along with the natural course of reversibility of the SC.
Conclusion
This case highlights an IRR with nab-paclitaxel which led to the development of SC. The novel presentation in this case is the development of SC with the use of nab-paclitaxel. In retrospect, the patient may have been predisposed to SC due to the presence of lung adenocarcinoma. Reducing the dose of nab-paclitaxel was sufficient to prevent any future similar event. This case serves as an addition to the literature of previously described IRR and SC with anticancer drugs.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal consent was obtained from the patient for their anonymized information to be published in this article.
Prior Presentation of Abstract Statement
The abstract was presented at the American College of Cardiology—Advancing the Cardiovascular Care of the Oncology Patient conference in a virtual meeting session on February 11, 2022, with the title Takotsubo cardiomyopathy secondary to taxol infusion reaction, Abdolmaleki, A., Singh, H., Hawes, E., Haynes, J.
