Abstract
Myxoinflammatory fibroblastic sarcoma (MIFS) is a rare tumor of soft tissue. It typically presents as a low-grade sarcoma with myxoid stroma, has a predilection for distal extremities, and displays a high propensity for local recurrence, but low metastatic potential. The risk factors associated with high-risk lesions metastasizing are poorly defined. In cases where the tumor metastasizes, therapeutic options are few, and death is rare. Our case discusses an aggressive MIFS that progressed from a painless lesion on a patient’s calf, to her death from a malignant pleural effusion within 21 months. The 58-year-old woman presented with a mass on her left calf. It was excised and was originally thought to be a benign process. It re-grew quickly after the initial resection, and she underwent re-excision of the mass. The pathologic examination was consistent with an MIFS. Despite negative margins on her second resection and an attempt at local control with radiotherapy, it metastasized to her lungs within less than 2 years. This resulted in a malignant pleural effusion that caused her death. An MIFS is typically benign but can metastasize in atypical cases. Even if the disease is metastatic, it is unlikely to be the cause of death. Treatment of metastatic MIFS is poorly defined, but there are suggested therapies beyond surgical resection and radiotherapy. Successful treatment of an MIFS should include a high index of suspicion in extremity lesions, screening for metastasis, and possible targeted therapies based on tumor genomics.
Keywords
Introduction
Myxoinflammatory fibroblastic sarcoma (MIFS) is an uncommon tumor of soft tissue that has been described since the late 1990s. They typically present as low-grade sarcomas with myxoid stroma and a predilection for distal extremities. They often recur locally but rarely metastasize to distant sites.1-4 Histologically, the architecture of MIFS is marked by fibrous septae dividing the tumor into lobules, comprising loose myxoid stroma and focal hyalinized areas associated with capillary proliferation. At higher power, neoplastic cells are intermixed with inflammatory infiltrate with emperipolesis of variable sizes containing intracytoplasmic neutrophils. Neoplastic epithelioid cells with marked nuclear atypia and prominent nucleoli are characteristic, and these have been termed “Reed-Sternberg-like” or “ganglion-like,” while multivacuolated pseudolipoblast cells are also variably present.2,5,6
Although MIFS was traditionally considered an acral sarcoma, these tumors have been reported on proximal extremities, trunk, head, and neck. It is thought that around 2% of MIFSs metastasize, but it is unclear to what extent histologic features associated with high-grade signal increased metastatic potential.4,6 Outside of a few studies, deaths resulting from metastatic disease in MIFS are rare and poorly understood.7-9 Consequently, an in-depth, longitudinal description of disease progression is absent in the literature. To add to this body of literature, we present an unusual case of an aggressive and fatal MIFS, which progressed from presentation to death from disease in less than 2 years. While there are around 20 cases of metastatic MIFS described, our case provides the most complete, longitudinal clinical, and pathologic examination of the progression of this disease. 7 We hope to expand on the current understanding of high-grade MIFS, its metastatic potential, and treatment options.
Case Discussion
A 58-year-old woman presented to our hospital for a second opinion for a fast-growing, recurrent mass on her posterior left calf. She originally presented to an outside physician complaining of a subdermal growth on the posterior left lower leg, 2 months prior. She only reported a past medical history of hypertension that was controlled with single-agent oral medication. She denied any recent trauma to the left calf. She did not recall any change in her exercise habits or lifestyle that would account for this mass. The soft tissue mass was approximately 4 cm at the time of the first resection by an outside surgeon. Pathologic examination measured 4 cm × 4 cm × 1.5 cm, with an initial pathologic diagnosis of an “epidermal inclusion cyst with underlying nodular fasciitis.” Over the next 2 months, the mass recurred on her left calf. At her initial consultation, her original slides were reviewed at our institution. Histologic review (Figure 1A) at our institution for treatment purposes, acknowledging the possibility of myxoid nodular fasciitis, considered a number of additional myxoid neoplasms and demonstrated no rearrangement of the EWSR1 locus by break-apart fluorescence in situ hybridization. Because the mass extended broadly to the edges of the original specimen, the pathologist recommended re-excision for full characterization. A computed tomography (CT) scan was ordered, which displayed a nonspecific appearance (Figure 1B). Based on the positive margins and rapid regrowth, the decision was made for the patient to undergo a wide local excision of the skin and subcutaneous fat of the left calf down to the level of the gastrocnemius (Figure 2). This was performed 3 months after the initial resection. Plastic surgery was consulted and Integra was placed over the wound. The final pathology of the 14 cm × 9.8 cm skin and subcutaneous fat displayed an MIFS with high-grade features, 5 cm in greatest diameter. Histologically, this tumor showed a nodular lesion (Figure 3A) based in the subcutis. It was variably but moderately cellular, with nodules of spindled to atypical epithelioid ganglion-like cells scattered in a myxoid background, some with the bilobed “Reed-Sternberg-like cell” nuclear morphology; emperipolesis was present (Figure 3B-C). The mitotic activity was ~4/10 high power fields; focal coagulative necrosis was present, as were areas of marked nuclear pleomorphism and solid growth, as have been described in MIFSs with high-grade features, 8 an impression also shared upon review by additional extramural consultants. Immunohistochemical staining with CD34 was focally positive involving the cells and highlighted lesion-associated capillaries. The lesional cells were negative for cytokeratin AE1/AE3, SOX10, S-100, Myogenin, SMA (smooth muscle actin), and Desmin. Cyclin D-1 was diffusely positive, highlighting emperipolesis. The margins were free but close (0.1 cm).

(A) At low power, the original biopsy sample showed reactive skin and dermal scar (upper right) overlying the superficial aspect of a hemorrhagic lesion with myxoid stroma in the superficial subcutis at lower left. While broadly transected at the base, the mixed inflammatory infiltrate and extravasated erythrocytes likely contributed to the original case contributor’s favoring nodular fasciitis as the diagnosis (H&E, photographed at 40X). (B) Computed tomography (CT) of the patient’s left calf taken after the initial excision and before the secondary excision. Sagittal section with a hyperdense lesion located within the soft tissue of the posterior aspect of the calf. Consistent with a local recurrence of the tumor after the initial surgery.

Gross image of resected tumor at the completion of the re-excision of the tumor. Wide local excision with resection of the fascia overlying the muscle belly of the gastrocnemius and Achilles tendon. Ruler displayed for scale is slightly larger than 6 inches and 15 cm in length.

(A) Upon resection, a distinctly nodular tumor with myxoid stroma and interstitial hemorrhage is seen in the subcutis (H&E, photographed at 12.5X). (B) At higher power, the tumor’s inflammatory and myxoid stroma is apparent, as are the atypical plump epithelioid to spindled lesional cells. A characteristic “Reed-Sternberg-like” cell with bilobed nucleus and prominent nucleoli is apparent at center (H&E, 200X). (C) At high power, the nuclear atypia apparent is marked, as is a cell with engulfed inflammatory cells (neutrophils), representing the emperipolesis characteristic of this tumor type (H&E, 400X). (D) In the patient’s re-resections, higher grade features were apparent, including very dense cellular areas with marked cellular and nuclear pleomorphism (left) as well as coagulative necrosis (right; H&E, 200X).
The patient underwent a split-thickness skin graft for wound coverage 1.5 months after the second resection and experienced excellent incorporation into the wound bed. Given the close margin, she was scheduled to undergo radiation therapy with 60 Gy in 30 fractions delivered to the wound bed after the skin graft healed. Medical oncology was also consulted, and no adjuvant chemotherapy was recommended at that time. A CT-scan of the chest, abdomen, and pelvis was obtained to evaluate for metastatic disease at 2 months from her secondary resection. A pelvic, left-sided 2.4 cm soft tissue density was noted, along with a 7.4 cm right adnexal mass, and a subtle, ill-defined enhancing lesion in the hilar plate at segment 4 of the liver. An MRI (magnetic resonance imaging) was obtained and the liver and left pelvic lesion were concerning for metastasis. The adnexal mass was deemed most likely benign by radiographic criteria. Percutaneous biopsy of the liver revealed cholangiocarcinoma, adenocarcinoma type. A biopsy of the pelvic mass was diagnosed as an incidental, benign schwannoma.
In the context of these multiple, unusual neoplasms, the patient was referred for genetic counseling and testing. The patient’s germline genetic examination, testing 83 loci (Invitae Multi-Cancer Panel, 2018) related to heritable cancer syndromes, displayed only 2 variants of uncertain significance. The first mutation was a heterozygous c.1906A>G variant of PTCH1, the locus associated with Gorlin (basal cell nevus) syndrome. 10 The other mutation was a heterozygous c.1582T>A in WRN, the locus associated with the autosomal recessive Werner syndrome, a progeria with cancer diathesis. Unfortunately, neither of these uncertain significance variants were known pathogenic, nor bear any evident causal relationship to the patient’s neoplasms.
Four months after her secondary resection, the patient experienced 2 new growths in the wound bed of her left calf. Fine needle aspiration confirmed local recurrence. Radiation therapy had not yet commenced, awaiting complete remodeling of her skin graft. We discussed her case at the multidisciplinary tumor board, and the decision was made for radiation therapy to both her calf and her liver for the 2 primary tumors. Medical oncology planned adjuvant therapy with gemcitabine and docetaxel after her radiation therapy was complete. She completed her radiotherapy and chemotherapy and a follow-up PET-CT (positron emission tomography–computed tomography) displayed avid lesions in the wound bed in the left leg and liver. A biopsy of the left lower leg displayed recurrent sarcoma. At the time, the patient was presented with treatment options including changing chemotherapy to gemcitabine and carboplatin, as well as an above the knee amputation of the left lower extremity, and she obtained a third opinion at a different institution. Now 9 months into her treatment from her secondary excision, she elected to change her chemotherapy. After 2 cycles of chemotherapy with gemcitabine and carboplatin, the patient stopped her infusions because of her declining quality of life.
Twelve months after her secondary excision, the patient’s left calf developed an area of skin ulceration, indicating further progression of her locally recurrent tumor (Figure 4). An updated biopsy displayed recurrent, high-grade sarcoma (Figure 3D). An outside institution reviewed her case and recommended isolated limb perfusion for possible limb preservation versus amputation. However, the wound worsened before her planned limb perfusion date and, at the patient’s request, she underwent a left above the knee amputation, revealing a now 12.8 cm tumor with multifocal lymphovascular invasion. All the margins were widely free of tumor. At 17 months after her second operation, she presented to our emergency department tachycardic, and hypoxic. She was admitted for acute hypoxic respiratory failure with a large right pleural effusion (Figure 5A). A thoracentesis obtained 1500 mL of sanguineous fluid that re-accumulated within 24 hours. An aspirate smear was positive for metastatic myxoid sarcoma (Figure 5B), consistent with the patient’s previous high-grade sarcoma. A CT-scan of the abdomen showed a decrease in the cholangiocarcinoma from 3.2 cm to 2.1 cm over the past year. Given her worsening respiratory status and rapid C of the malignant effusion, the patient and her family elected for hospice and she died 21 months after her original diagnosis and 17 months after her secondary excision.
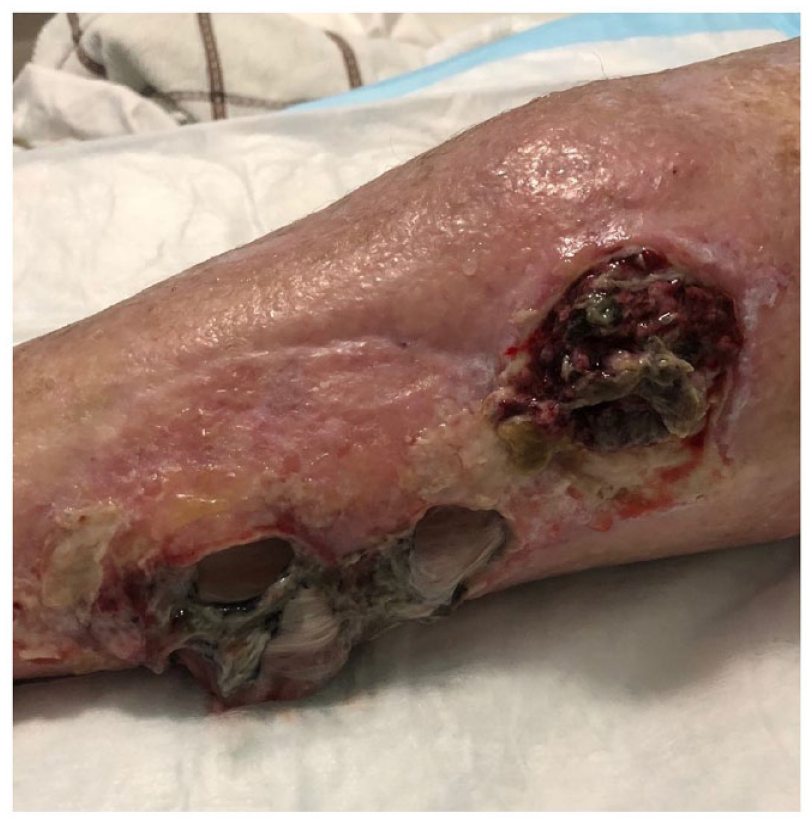
Gross image of the patient’s left calf approximately 1 year after her second excision. The wound displays local recurrence with ulceration of the calf.

(A) CT-scan of the patient’s thorax consistent with a 1500 mL malignant pleural effusion of the right thorax. (B) A representative field from the cell block preparation from the patient’s pleural effusion demonstrated an atypical cellular population comprising epithelioid and spindle cells and myxoid stroma, again with admixed inflammatory cells, documenting distant metastatic thoracic recurrence for this tumor (H&E, 200X).
Discussion
We have described our experience with an integrated clinical, pathological, and germline genetic description of a patient with high-grade, aggressive MIFS, directly leading to her death. She died 21 months after her initial diagnosis, with interventions comprising 2 wide local excisions, 1 with negative margins, radiotherapy, chemotherapy, and an above the knee amputation. While cases of metastatic MIFS have been described, death from this disease has been described in less than 20 cases to date.
An MIFS is typically considered to have a low but real risk of metastasis, according to the 2020 WHO (World Health Organization) classification. Many described cases have been treated with local excision, re-excision if necessary, and, occasionally, radiotherapy. They can carry at least some metastatic potential, which has been estimated as occurring in approximately 2% of patients diagnosed with MIFS.4,11
Laskin et al reviewed all extremity sarcomas from the Armed Forces Institute of Pathology/Joint Pathology Center from 1961 to 2004. Pathologic cases that were consistent with MIFS (n = 104) were identified and examined. Of these, 59 had documented clinical follow-up, ranging from 9 months post-resection to 372 months. 5 Of the 59 patients with clinical follow-up, an overall recurrence rate of 51% was documented. This study reported that 16 of the 59 patients with follow-up were deceased, but none of these deaths were attributed to MIFS per se. 5 Reviewing their metastatic cases for possible prognostic factors, the group evaluated the original size of the tumor, stromal composition, presence of necrosis, hallmark cells, whether the histology was conventional or atypical, the surgical resection margin, and whether 2 or more atypical features were present. They found that the only statistically significant predictor of recurrence was an R1 (microscopically positive margins) resection at the initial surgery. 5 A literature review by Lombardi et al found 138 reported patients diagnosed with MIFS by 2013. In their cohort, the rates of positive margins for initial versus re-excision subsets of the cohort were 63% and 6%, respectively (P < .001), but no difference in recurrence between the groups was apparent from their review. Utilizing meta-analysis of published data on these cases, no risk factors for recurrence were identified. Parameters assessed included the patients’ ages, gender, tumor site, pain at presentation, duration of symptoms, tumor size, type of surgical treatment, margin status, and use of adjuvant treatment. 9
Locoregional recurrence of MIFS is estimated to occur in 20% to 70% of cases and is typically managed by re-excision, with or without radiotherapy. Suggested high-risk features for evaluating which MIFS may become metastatic have been described in the literature, but given the limited data known about this sarcoma, no definitive evidence exists.6,7 High-grade MIFSs are classified as such by their morphology. Blastic tumor cells with a high degree of pleomorphism, with numerous atypical to bizarre mitosis, and frequent foci of necrosis are associated with higher grade lesions.6,8 Our patient’s tumor possessed these features and was located on her calf, as opposed to the more traditional acral locations, thus placing her tumor as high grade with metastatic potential. The immunophenotypic profile of MIFS is not helpful for diagnostic or grading purposes as tumor cells can variably express multiple markers.11,12 Metastatic cases of MIFS are either described as similar to the primary tumor or altered in appearance with higher cytologic atypia and cellularity, similar to that seen in the recurrence of this case.6,8 Hassanein et al reported their experience, including cases of 2 of 5 patients who developed distant metastases. The first patient developed a local recurrence 1 year post her original diagnosis that progressed to the regional nodal basin and eventually to the neck and base of the skull. With the metastasis, the patient survived at least 79 months after the original diagnosis. Their second patient developed 3 local regional recurrences, the earliest at 5 months following their initial excision. They underwent an amputation of the affected limb but developed a pulmonary metastasis that eventually led to the patient’s death, 3 years after the original diagnosis. 7 Following this pattern of malignant lung metastasis, an analysis of 23 MIFSs with high-grade features similar to our case found lung metastasis in all patients with metastatic disease, 7 of whom died from metastatic disease within 2 years after the first resection. 8 A subsequent analytical review of 138 cases by Lombardi et al found 4 cases presenting with lung metastasis, with 1 patient presenting with unresectable pulmonary metastases associated with pleural effusion at diagnosis. 9 Finally, Srivastava et al presented a case in 2018 of an aggressive presentation of MIFS in a 38-year-old patient with extensive local spread from subcutaneous tissue to the ankle joint and bones, with multiple synchronous metastases to the lung, sixth rib, and vertebrae. 13 The lung appears to be the most common distant metastatic site for MIFS, regardless of the presenting location. We have listed all found metastatic cases in the English literature (Table 1).
List of All Metastatic Cases Found in Studies in English.

Abbreviations: E, excision; CH, chemotherapy; RT, radiotherapy; AMP, amputation.
While wide local excision and radiotherapy to the tumor bed seem to confer a decrease in the recurrence of the tumor, it has been documented that patients with metastatic MIFS have received chemotherapy, but the regimen was not discussed. It seemed to be effective at disease control for some cases; patients have survived up to 9 months after diagnosis with regression of disease on imaging. 13 Our patient received gemcitabine and docetaxel partially for therapy of her cholangiocarcinoma but experienced no treatment benefit for her MIFS. Her concurrent cholangiocarcinoma responded to her regimen, and she was never able to complete a subsequent line of therapy due to her preference and aggressive progression of the disease. Kinase fusions have been the basis for successful treatment with imatinib for a small subgroup of MIFS patients. 5 Given the recent identification of BRAF rearrangements in a subset of MIFS, 16 the exploration of targeted therapy for these tumors is a promising avenue for future investigation.
Summary
Our patient presented with a high-grade MIFS that progressed to her death in less than 2 years despite multiple therapies. We have displayed the clinical and pathologic course of her disease in greater detail than those currently in the literature, in hopes that it may help guide future studies for successful therapy for these cases.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Informed consent for patient information to be published in this article was obtained from the patient’s family.
