Abstract
Checkpoint inhibitors have gained increased traction in recent years as they have improved prognosis in various malignancies. Pembrolizumab, an anti-programmed cell death protein (PD-1) monoclonal antibody, has become a first-line chemotherapeutic agent for stage II non–small cell lung cancer since 2019. Although much more common with nivolumab, several immune-related adverse effects, particularly endocrinopathies, have been linked with pembrolizumab. We describe a case of a 59-year-old man with a history of unspecified lung cancer who presented with severe hyponatremia later attributed to secondary adrenal insufficiency and accompanying primary hypothyroidism secondary to pembrolizumab. Diagnosing adrenal insufficiency in patients on immune checkpoint inhibitors like pembrolizumab can be challenging due to nonspecific symptoms, making it crucial to rule out other causes of hyponatremia. Immunotherapy is known to cause thyroid immune-related adverse events, and anti-thyroid antibodies may not always be present in the diagnosis of hypothyroidism. Although there are some reported cases of pembrolizumab-induced adrenal insufficiency, the link between immunotherapy and endocrine disorders remains unclear. To our knowledge, no case reports exist that describe both primary hypothyroidism and secondary adrenal insufficiency after taking pembrolizumab, although such cases have been documented with axitinib. Timely diagnosis and treatment of adrenal insufficiency is crucial to prevent adverse effects, especially in patients with cancer receiving immunotherapy, as highlighted in this case.
Introduction
Immune checkpoint inhibitors (ICIs), such as pembrolizumab and nivolumab, are essential in cancer treatment. 1 Pembrolizumab, a humanized IgG4 monoclonal antibody, inhibits the PD-1 receptor and is approved for various malignancies, including melanoma, non–small cell lung cancer (NSCLC), Hodgkin lymphoma, cervical cancer, and renal cell carcinoma.2,3 Pembrolizumab’s mechanism involves selectively silencing immunological cell surface proteins, thereby shielding both healthy and tumor cells from immune system–induced apoptosis. 4
Although ICIs have improved the survival rates in those suffering from malignancy, rare immune-related adverse effects such as endocrinopathies can occur. 3 One such endocrinopathy is adrenal insufficiency (AI), which has a reported incidence of 0.98%. 2 We describe a case of secondary AI and primary hypothyroidism in a 59-year-old man secondary to pembrolizumab use for lung cancer.
Case Presentation
A 59-year-old man with a history of hypertension, epilepsy, and unspecified lung cancer currently treated with pembrolizumab presented to the emergency department with altered mental status and fatigue. The review of systems was unremarkable for any pertinent findings. On physical examination, the patient was lethargic, weak, and had acute changes in mental status. Vital signs were found to be within normal limits. He had received his last pembrolizumab dose within the same week he presented to the emergency department. Initial sodium level was 129 mEq/L. However, the patient’s sodium levels continued to be between 120 and 122 mEq/L over the next few days (Table 1).
Comprehensive Metabolic Panel for Patient’s Visit.

The patient had a recent hospital admission a week earlier with a similar presentation that included hyponatremia, during which he was found to have COVID-19 infection treated with a 5-day duration of dexamethasone (10 mg) and remdesivir with improved clinical status. On this admission, workup of hyponatremia included evaluation of early morning adrenocorticotropic hormone (ACTH), cortisol, FT4, and thyroid-stimulating hormone (TSH) levels (Table 2). The interval time between stopping dexamethasone and checking cortisol was 7 days. A magnetic resonance imaging of the brain was ordered to rule out any pituitary lesion (Figure 1). Thyroid-stimulating hormone levels (74.95 μIU/mL) were noted to be elevated, whereas free T4 was found to be severely decreased. Thyroid-stimulating hormone was compared with earlier values, dating back to 2014, in which TSH was 0.74 (reference range: 0.35-5.6 μIU/mL). Unfortunately, free T4 levels dating back to 2014 were not available. Thyroid peroxidase (TPO) antibody measurements were conducted, but no TPO antibodies were detected.
Other Relevant Laboratory Studies.
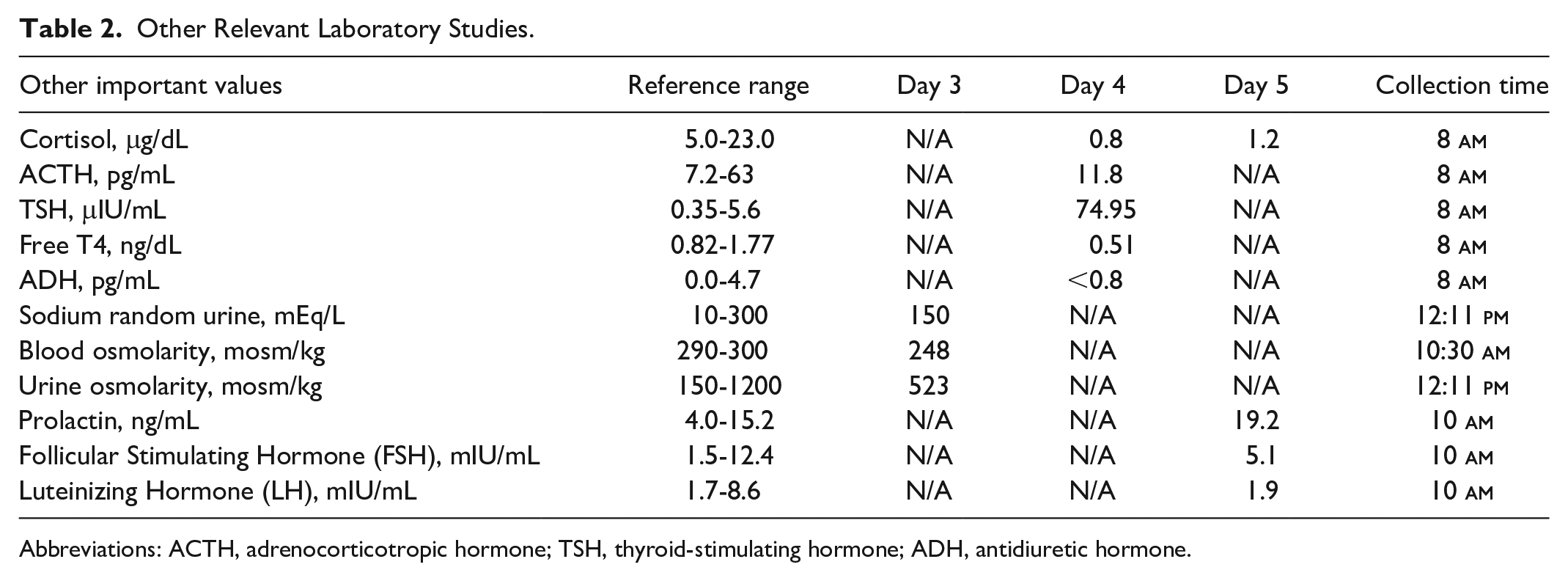
Abbreviations: ACTH, adrenocorticotropic hormone; TSH, thyroid-stimulating hormone; ADH, antidiuretic hormone.

Magnetic resonance imaging of the brain. Red arrow shows the normal pituitary gland.
The ACTH and cortisol levels were low and an ACTH stimulation test was performed, which showed an increase in cortisol levels from 0.8 to 5.9 μg/dL after 1 hour of injecting cosyntropin intravenously. The patient was diagnosed with secondary AI and primary hypothyroidism secondary to pembrolizumab. Initially, the patient was treated with water restriction; however, after the confirmation of the diagnosis of secondary AI and primary hypothyroidism, he was started on hydrocortisone followed with levothyroxine replacement (day 6 in Table 1). He showed improvement in sodium levels to 128 to 130 mEq/L (Table 1) and was discharged home with outpatient follow-up. Patient was followed on an outpatient basis with an oncologist outside our medical system.
Discussion
The ICIs have been linked with several endocrine disorders, and this usually occurs weeks or months after they are started. In fact, about 29% of patients treated with ICIs show signs of an endocrine disorder, usually around the sixth to seventh week with a median onset of 7 to 20 weeks. 5 However, AI has a rare association with ICIs and is oftentimes difficult to diagnose due to the vague signs that it presents with. Fatigue is the most common symptom reported by 16% to 24% of patients treated with chemotherapeutic agents with a similar mechanism to pembrolizumab. 5 Other nonspecific signs include nausea, vomiting, weakness, abdominal pain, headache, and weight loss.
The mechanism behind pembrolizumab and AI is poorly understood, though the relationship between hyponatremia and cancer has been extensively studied. Hyponatremia observed in patients with cancer has been linked to the nonosmotic release of vasopressin with consequent hypoosmolar hyponatremia.6,7 Nausea, pain, and syndrome of inappropriate antidiuretic hormone (ADH) are common causes of excess ADH release, while ectopic production of ACTH from tumors is also possible. 6 Adrenocorticotropic hormone, cortisol, free thyroxine, and TSH level labs are ordered to help in ruling out causes of hyponatremia. This patient had decreased ADH values, which effectively ruled out excess ADH release as a cause of hyponatremia in this patient. Adrenal insufficiency should be suspected if morning serum cortisol levels are <3 μg/dL. 5 In this case, cortisol levels were 0.8 μg/dL, and ACTH levels were low. Based on the patient’s response to the cosyntropin stimulation test, evident by the increase in cortisol from 0.8 to 5.9 μg/dL, and the observation that ACTH levels were within the low/normal range, it was concluded that the most likely mechanism in this case was secondary AI. Early morning ACTH and cortisol levels were low and an ACTH stimulation test was performed, which showed an increase in cortisol levels from 0.8 to 5.9 μg/dL after 1 hour of injecting cosyntropin intravenously. Similar to other irAEs that are associated with ICIs, the patient was able to continue treatment with pembrolizumab without interruption and did not develop any other endocrinopathies.
Several case reports have demonstrated AI after treatment of nivolumab; however, to our knowledge, there are only 4 other case reports of pembrolizumab-induced AI. In 2017, Paepegaey et al described a case of a 55-year-old woman who was started on pembrolizumab immunotherapy for a metastatic choroidal melanoma, and 1 month after discontinuation of the agent, the patient developed primary AI, which was confirmed with a computed tomographic scan showing atrophic adrenal glands and elevated 21-alpha hydroxylase levels. 8 Furthermore, this patient developed thyroiditis 4 months after treatment with pembrolizumab. In contrast, our patient developed concurrent primary hypothyroidism and secondary AI within the same week of his last dose of pembrolizumab. In 2020, Bekki et al described a case of a 65-year-old Japanese woman who was treated with pembrolizumab for metastatic cecal cancer and developed secondary AI after 7 weeks of the first dose of pembrolizumab. 5 Similar to our case, the main presenting symptom was fatigue and the patient was treated with hydrocortisone. In the same year, Tanaka et al described an 85-year-old Japanese woman who was treated with pembrolizumab for advanced squamous cell lung cancer and presented with generalized fatigue, appetite, and weight loss. 9 She was diagnosed with isolated ACTH deficiency and later treated with hydrocortisone. Last, Uppal et al described a case of a 68-year-old woman treated for metastatic NSCLC and diagnosed with isolated ACTH deficiency and treated with hydrocortisone. 6 To our knowledge, there are no case reports that exist describing both primary hypothyroidism and secondary AI after taking pembrolizumab, though several have been described with axitinib. 3
Primary hypothyroidism presents with elevated levels of TSH often in conjunction with low free T4. Thyroid-stimulating hormone measurement is the preferred and more sensitive test to diagnose ICI-induced hypothyroidism. Our index patient had low free T4 levels with elevated TSH concentration, which supports the diagnosis of primary hypothyroidism.
Thyroid irAEs, including hypothyroidism, hyperthyroidism, painless thyroiditis, and transient thyrotoxicosis, are known to occur with anti-PD1 or anti-PDL-1 therapy, affecting approximately 10% of patients. 10 Hypothyroidism and hyperthyroidism were reported in 7.9% and 3.8% of patients, respectively, in a meta-analysis of 38 studies, regardless of cancer type. 11 While the median time to onset of hypothyroidism is 6 weeks from ICI initiation, it can occur at any point during therapy. 12 Although ICI-induced hypothyroidism often lacks anti-thyroid antibodies, the presence of these antibodies increases the risk of developing ICI-induced thyroid adverse events. 13
As this patient presented with both secondary AI and primary hypothyroidism, a clinical decision needed to be made regarding the order of treatment. In this case, we chose to address AI before treating the hypothyroidism. A relevant case report by Murray et al warns about the potential worsening of primary AI and the risk of adrenal crisis when levothyroxine therapy is initiated, suggesting that hydrocortisone treatment should be prioritized to prevent clinical deterioration. 14 Schneider et al provide a similar recommendation stating that thyroid hormone replacement therapy before hydrocortisone increases the rate of metabolism of glucocorticoids, which can lead to worsening adrenal crisis. 15
This study presents a unique case of a patient with unspecified lung cancer who developed both secondary AI and primary hypothyroidism after treatment with pembrolizumab. The simultaneous presence of thyroid and adrenal involvement related to anti-PD1 therapy has only been reported in a handful of cases. Zeng et al describe a case of nivolumab-induced primary hypothyroidism followed by isolated ACTH deficiency. 16 Perhaps the biggest limitation in this case was the fact that a detailed workup for hyponatremia was not done on the first admission, which may have led to delayed treatment for our patient.
Conclusion
The diagnosis of AI may be delayed due to the vast differential diagnosis of fatigue. Thus, physicians should maintain a high index of suspicion for AI and initiate prompt treatment to prevent adverse effects on the patient’s health and quality of life, as highlighted in this case. Furthermore, it is crucial to conduct thyroid and adrenal testing in patients undergoing cancer treatment with pembrolizumab or other ICIs. Physicians should also educate patients on potential irAEs and their presenting symptoms before starting treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article
