Abstract
BK polyomavirus nephropathy (BKVN) is a common cause of nephropathy in kidney transplant patients and is typically seen within the first year after transplantation. BK polyomavirus nephropathy can occur in the native kidneys of patients with nonrenal solid-organ transplants (NRSOT). However, this is rare, especially outside the early post-transplant period, and BKVN is not usually considered in the differential diagnosis for acute kidney injury in NRSOT patients. We present a case of a 75-year-old man who had undergone orthotopic heart transplant 13 years prior with stable allograft function who developed progressive renal dysfunction in the setting of recent unilateral obstructive nephrolithiasis requiring ureteral stenting. Kidney biopsy demonstrated evidence of polyomavirus nephritis. Serum BK viral load was elevated. Despite reducing immunosuppression and initiating leflunomide, viral clearance was never achieved. The patient experienced progressive failure to thrive before ultimately transitioning to hospice care and dying. The intensity of immunosuppression is a well-known risk factor for viral replication; ureteral stenting has also been associated with BKVN. However, since clinical manifestations of BK viral infections often include a genitourinary (GU) tract pathology, it is important for clinicians to consider BKVN in patients with NRSOT with progressive renal dysfunction, especially in the clinical context of known GU disease.
Introduction
BK polyomavirus nephropathy (BKVN) is the most common form of polyomavirus nephropathy in kidney transplant patients and is typically seen within the first year after transplantation. 1 BK polyomavirus nephropathy can occur in the native kidneys of patients with nonrenal solid-organ transplants (NRSOTs). However, this is rare, and BKVN is not usually considered in the differential diagnosis for acute kidney injury (AKI) in NRSOT patients.2-5 Here, we report a case of BKVN involving the native kidneys of a patient who had undergone recent ureteral stenting 13 years after orthotopic heart transplantation (OHT).
Clinical Background
A 75-year-old man with stage G3bA2 chronic kidney disease (CKD) presented for evaluation of AKI. He had a history of ischemic cardiomyopathy for which he had received OHT 13 years ago. Since the OHT, he has had stable cardiac allograft function, as well as stable CKD with a baseline serum creatinine (SCr) of 1.6 mg/dL. His other medical history included type 2 diabetes mellitus (recent hemoglobin A1c 6.8%) and hypertension. Maintenance immunosuppression consisted of extended-release tacrolimus (0.75 mg daily), sirolimus (0.5 mg daily), and mycophenolate mofetil (540 mg twice daily). There had not been any previous documented episodes of acute rejection. No routine monitoring for BK polyomavirus had previously been performed.
Four months before the current presentation, he was found to have an asymptomatic AKI with SCr elevation to 2.9 mg/dL and was admitted to the hospital for evaluation. Further evaluation revealed right-sided obstructive ureterolithiasis at the ureteropelvic junction. Tacrolimus and sirolimus levels at that time were above their therapeutic target with trough levels of 6.4 ng/mL and 7.8 ng/mL, respectively (the combined trough goal was 7-11 ng/mL). The patient was treated with trimethoprim-sulfamethoxazole for a complicated urinary tract infection, and a ureteral stent was also placed. A week later, the SCr improved to 1.6 mg/dL. After 1 month, he underwent uncomplicated laser lithotripsy and ureteral stent removal for definitive treatment of nephrolithiasis.
Now, 4 months after the initial AKI, he was found to have a new persistent AKI with SCr of 2.6 mg/dL. Despite dose reduction, serum levels of immunosuppressive agents remained mildly above their combined goal, with tacrolimus and sirolimus trough levels at 5.0 ng/mL and 6.7 ng/mL, respectively. Further evaluation revealed sub-nephrotic-range proteinuria with a urine protein-creatinine ratio of 2.8 g/g, along with glucosuria (100 mg/dL), pyuria (44 white blood cells/high-power field [HPF]), and microscopic hematuria (19 red blood cells/HPF). Kidney ultrasonography demonstrated bilateral normal-sized kidneys with normal echogenicity and without hydronephrosis. Supplementary diagnostic evaluation was unremarkable and is summarized in Table 1. A timeline of the case report is shown in Figure 1.
Additional Laboratory and Imaging Evaluation.
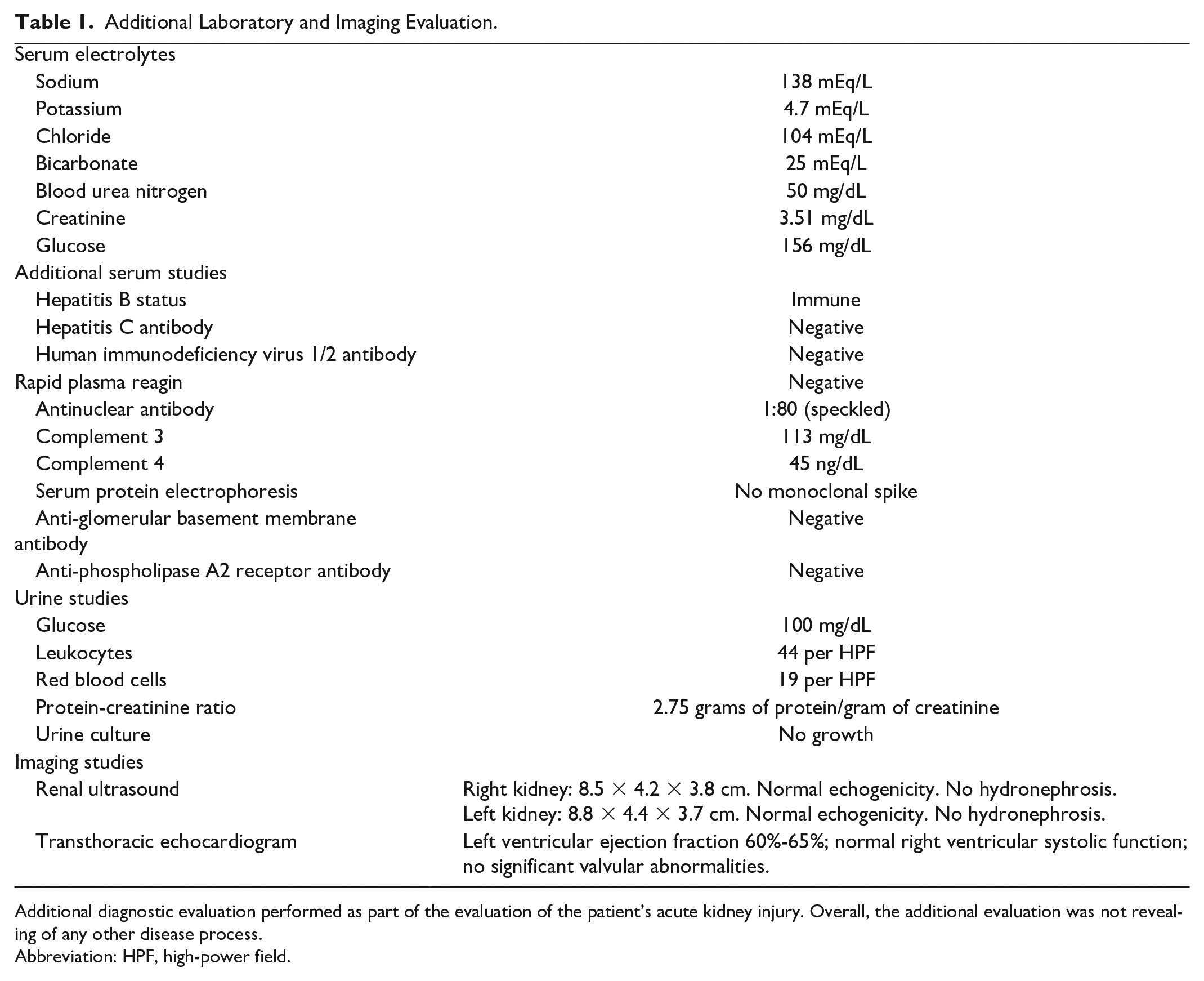
Additional diagnostic evaluation performed as part of the evaluation of the patient’s acute kidney injury. Overall, the additional evaluation was not revealing of any other disease process.
Abbreviation: HPF, high-power field.

Timeline of the case report.
A kidney biopsy was performed 1 month after the diagnosis of the recurrent AKI and revealed acute tubular injury with focal areas of viral cytopathic atypia and positive simian virus 40 immunohistochemical staining for BK/polyomavirus within tubular epithelial cells. These findings were consistent with polyomavirus nephritis, class I (Figure 2). At that time, serum and urine BK virus PCR levels were 362 000 copies/mL and >100 000 000 copies/mL, respectively.
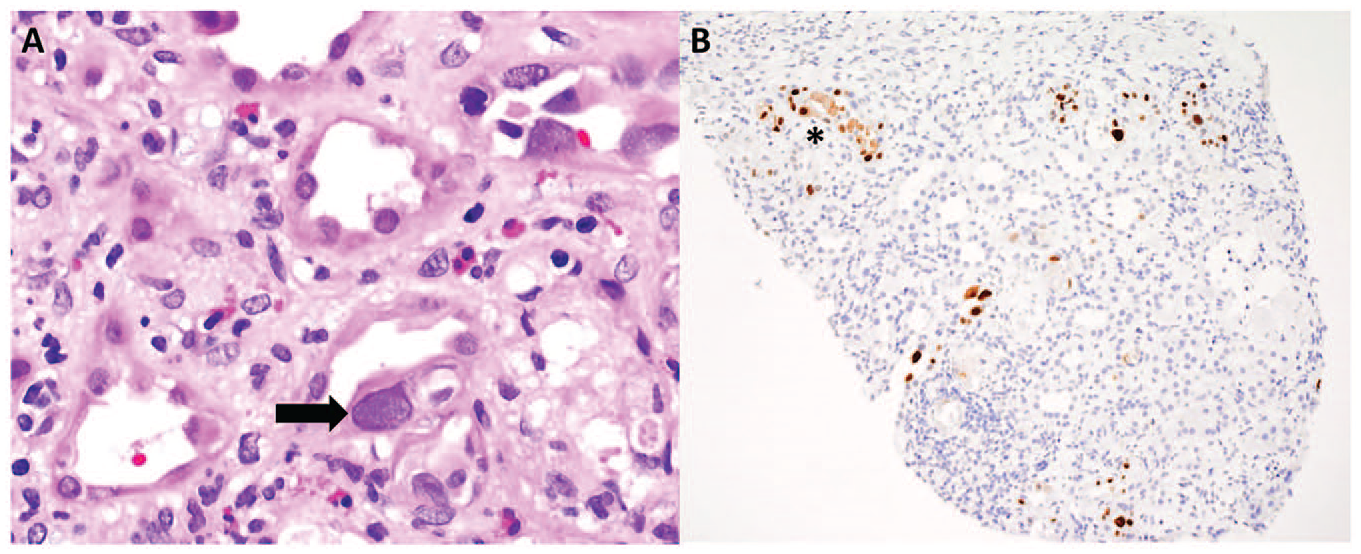
(A) Renal biopsy showing acute tubular injury with tubulointerstitial inflammation and viral cytopathic effect (arrow) (hematoxylin and eosin stain, original magnification 400×). (B) Renal biopsy showing tubular epithelial cells with focal positive simian virus 40 immunohistochemical stain for polyomavirus. An area of focal positive staining is denoted with an “*” (original magnification 100×).
Immediately upon the diagnosis of polyomavirus nephritis, immunosuppression was further decreased to extended-release tacrolimus 0.75 mg daily and sirolimus 0.5 mg every other day with a cumulative trough goal of 6 ng/mL. In addition, mycophenolate mofetil was stopped. Leflunomide was started for the treatment of BKVN because of its potential antiviral properties. The serum creatinine level was 3.5 mg/dL at the start of treatment but continued to worsen to 4.3 mg/dL 1 month later. Consequently, he was admitted to the hospital. Further evaluation at that time revealed mild bilateral hydronephrosis of unclear etiology with evidence of delayed right-sided flow on retrograde pyelogram. He subsequently underwent repeat right ureteral stenting. During the admission, he was also diagnosed with locally invasive squamous cell carcinoma of the scalp without evidence of metastases. Sirolimus was held 2 weeks preceding wide local excision to prevent impaired wound healing.
Over the 2 months following the diagnosis of BK nephropathy, serum BK viral levels and renal function fluctuated. The serum creatinine level peaked at 4.5 mg/dL, followed by subsequent improvement to a nadir of 2.9 mg/dL. BK viral load improved but remained detectable, dropping to 13 000 copies/mL 3 months after the initial reduction of immunosuppression and initiation of leflunomide (Figure 3). Despite these therapeutic efforts, the patient’s overall health continued to decline with persistent and progressive fatigue, physical deconditioning, and weight loss. In light of his deteriorating health and quality of life, he elected to enter hospice care and died from his progressive failure to thrive 4 months after the diagnosis of BK nephropathy and initiation of treatment.
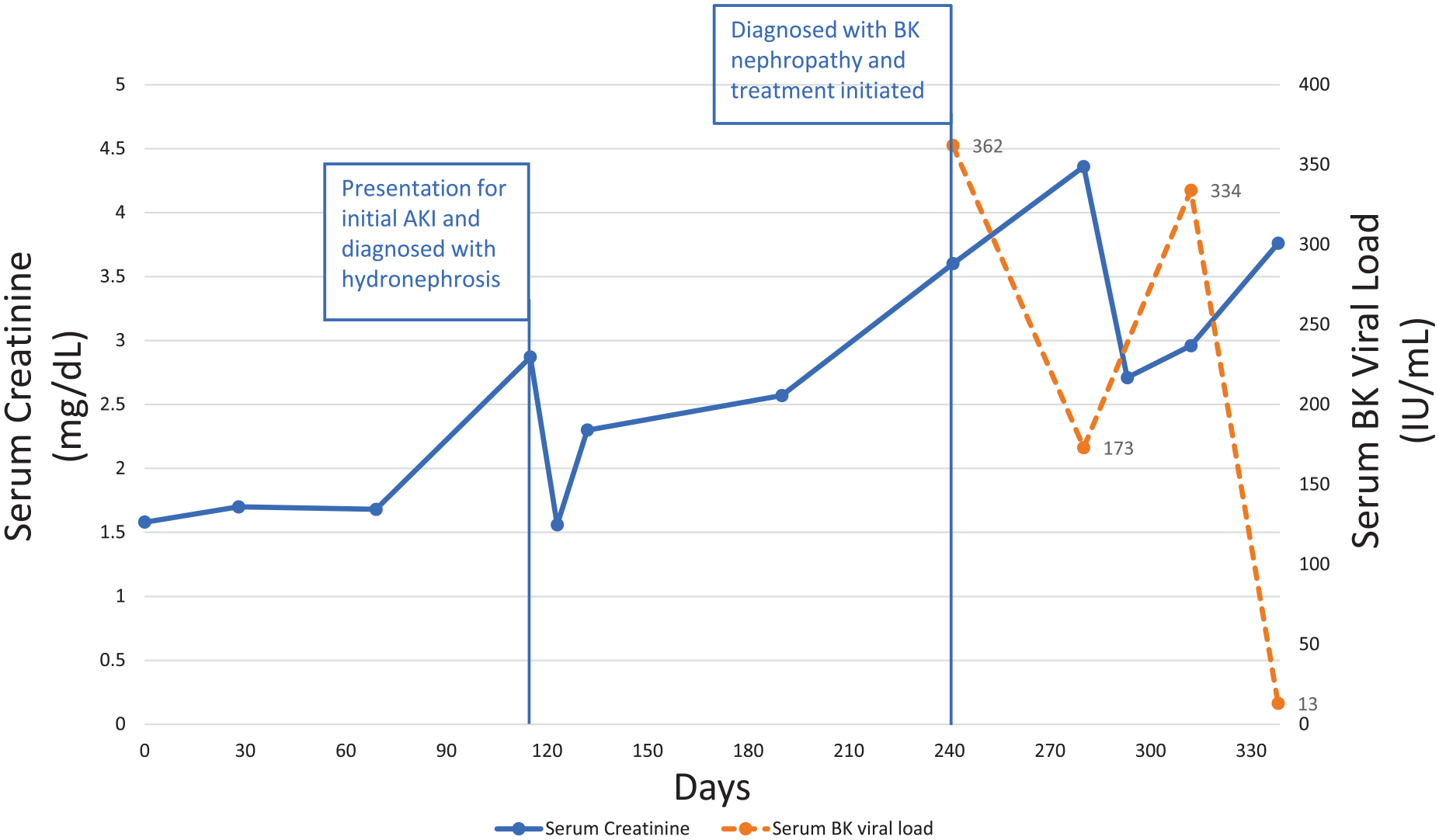
Trend of the patient’s serum creatinine and BK viral load over time during the preceding year. Day 0 represents baseline creatinine. The patient was diagnosed with unilateral hydronephrosis on day 115 and underwent unilateral ureteral stenting at that time. The patient underwent kidney biopsy and was diagnosed with BK nephropathy on day 240. Treatment was initiated at that time. Despite a reduction in immunosuppression and initiation of leflunomide, the viral load remained detectable with a nadir of 13 000 IU/mL, and the patient continued to have deteriorating renal function.
Discussion
Infection with BK virus is common worldwide. Seropositivity is up to 80% in some populations, with initial infection typically occurring in childhood.6,7 The virus can subsequently live in a dormant fashion in the renal tubules with transient episodes of asymptomatic replication and viruria over an individual’s lifetime.8,9 Despite the high rate of BK virus seropositivity in the general population, BKVN is seen almost exclusively in kidney transplant patients. 10 The degree of immunosuppression is a well-known risk factor for viral replication. 11 Despite the high intensity of immunosuppression in many NRSOT patients, native kidney BKVN is uncommon. 12 The preferential effect on kidney transplant patients is hypothesized to be related to the renal injury that occurs in the setting of renal transplant. 13 The combination of ischemia-reperfusion and immune-related injury is thought to act as a stimulus for viral reactivation. 13
In a meta-analysis evaluating the frequency and risk factors for BK viruria and viremia in NRSOT patients, Viswesh et al found a relatively high rate of viruria (8%-52%) but infrequent progression to viremia (3%-7%) and BKVN (1 biopsy-proven case in an OHT recipient). 12 Among those NRSOT patients who did have progression to viremia and BKVN, OHT patients represented the majority of cases. In one study, the median time to onset of BK viremia in OHT occurred in the 30 days immediately after the transplant or following the antirejection treatment. 14 While early BK viremia is associated with progression to BKVN in renal transplant patients, the clinical significance of early BK viremia in cardiac transplant patients remains unclear.15,16
Several other studies have also observed that BKVN is more common in OHT patients than other forms of NRSOT.5,12,17 This finding might be due to the proposed “double-hit” hypothesis, which suggests that the cumulative insult of immunosuppression and renal hypoperfusion secondary to cardiac allograft dysfunction causes clinical progression to BKVN. 18 However, in our case, the patient’s cardiac transplantation occurred over a decade ago. In addition, our patient did not have any evidence of cardiac allograft dysfunction that would lead to renal hypoperfusion; it is unclear what may have provoked viral reactivation other than immunosuppression.
Ureteral stenting has also been observed to be associated with increased risk of BKVN, likely due to ureteral trauma and subsequent increase in BK viruria.1,11,19-21 Conversely, there is also evidence that infection by BK virus itself can induce genitourinary tract pathologies such as ureteral strictures and hemorrhagic cystitis.11,19 In the case of our patient, the development of ureteral strictures could represent either a risk factor for BKVN due to manipulation of the genitourinary tract or a symptom of BKVN itself. Therefore, despite the rarity of BKVN in NRSOT patients, it is vital for clinicians to maintain a high level of suspicion for the condition if NRSOT patients have a known genitourinary tract disease.
Because native-kidney BKVN occurs quite infrequently, most knowledge regarding treatment principles is derived from experience with BKVN in kidney allograft recipients. However, despite BKVN being the most common cause of kidney allograft dysfunction within the first year after transplantation, advances in the management of polyomavirus-associated diseases are limited by a lack of randomized controlled trials of different therapeutic approaches. 22
In almost all instances, the primary step in the treatment of BKVN in the absence of concurrent acute organ rejection is a reduction of therapeutic immunosuppression. This approach is thought to allow the individual’s own immune system to combat the infection. A meta-analysis and several robust observational studies have supported this widely used strategy. 15 Clinical response to adjustment of immunosuppression is typically evaluated with repeated measurements of the plasma BK viral load and SCr over time. Various strategies have been tried regarding how to adjust immunosuppression in the face of infection. The most common approaches include a reduction of the calcineurin inhibitor and antiproliferative agents by 25% to 50%, combined with a reduction in the steroid dose. 22 Other strategies involve changing the type of calcineurin inhibitor or switching to a mammalian target of rapamycin inhibitor.22,23 A similar approach of immunosuppression reduction is used to treat another polyomavirus infection, JC virus–associated progressive multifocal leukoencephalopathy, in which the goal of treatment is also to restore immune function. 24 In our patient, despite a reduction in immunosuppression, viremia persisted, resulting in a progressive decline in kidney function.
We are not aware of any randomized controlled trials that support additional therapies for the treatment of BKVN if clinical improvement is not achieved with the reduction of immunosuppression. However, additional therapeutic agents that have been used in the treatment of BKVN include intravenous immunoglobulin (IVIG), leflunomide (a pyrimidine synthesis inhibitor most commonly used in the treatment of rheumatoid arthritis that has in vitro activity against BK virus), cidofovir (an antiviral agent), brincidofovir (a prodrug of cidofovir), and fluoroquinolone antibiotics.1,15
In some studies, IVIG has been shown to assist with more rapid clearance of BK viremia.25,26 Small studies have suggested some benefits with the use of cidofovir. 27 However, cidofovir itself is nephrotoxic and, therefore, must be used carefully. Brincidofovir, a prodrug of cidofovir, is less nephrotoxic and could be advantageous; however, data on its use are limited. Fluoroquinolone antibiotics have been shown to have in vitro activity against the BK virus; however, multiple randomized control trials and metanalyses have not demonstrated any benefits.28-30
Conclusion
Native-kidney BKVN is uncommon in NRSOT patients, especially after several years following transplantation. Within the NRSOT population, cardiac transplant recipients are at the highest risk for developing BKVN. The augmentation of immunosuppression is a well-documented risk factor for the development of BKVN in kidney allograft patients. Ureteral stenting might be another risk factor for the condition. Although rare, it is important for clinicians to consider BKVN in extra-renal solid-organ transplant patients with progressively worsening kidney function, especially in the clinical context of known genitourinary tract disease or recent ureteral stenting, since delays in treatment can lead to permanent kidney dysfunction. In addition, there is a significant gap in knowledge regarding the treatment of BKVN in NRSOT, and further investigation is warranted.
Footnotes
Authors’ Note
This care was presented as an abstract at the American Society of Nephrology Kidney Week 2022 Conference, Orlando, FL, on November 5, 2022.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Informed consent for patient information to be published in this article was not obtained because the patient died prior to being able to obtain informed consent, and the next of kin were unable to be reached despite multiple attempts. Only anonymized patient information is included in this article.
