Abstract
Mpox was an emerging outbreak in California in 2022, primarily in major metropolitan areas, as part of the global mpox outbreak declared by World Health Organization in July 2022. Community hospitals outside of major metropolitan areas have seen fewer cases to date, so they may be less equipped to diagnose and treat patients with mpox. They may have limited public health resources commensurate with the area’s population density. Mpox may also be superimposed on ongoing local outbreaks of other sexually transmitted infections. We present a case of a person with HIV who contracted mpox and also developed secondary syphilis. Early detection can be beneficial for prompt treatment, decreased burden of disease for the individual, and prevention of further spread of the infection.
Introduction
Mpox virus (formerly monkeypox), a non-variola orthopoxvirus, is the cause of a zoonotic pox disease that is endemic in several Central and West African countries. 1 It was first detected in 1958 in captive monkeys transported to Copenhagen, Denmark, from Africa for research. 2 “Monkeypox” as a name is actually misleading because the largest animal reservoir is small rodents that are hunted for food. 3 It is understood to be spread via direct contact with sores or scabs on infected individuals as well as large respiratory droplets or indirect and direct contact with bodily fluids, including saliva. It is assumed to enter the body through the respiratory tract, broken skin, or mucous membranes. 4
There are 2 clades, West African and Congo Basin, of which the latter causes more severe illness.1,5 Sporadic cases and outbreaks in the United States associated with returning travelers6,7 as well as with the import of small mammals from endemic countries 8 have been reported. In May 2022, several cases of mpox virus West African clade were discovered in the United Kingdom, the United States, 9 and Spain. 10 As part of the United States’s response to the emerging outbreak, rapid diagnostic testing with qualitative orthopoxvirus PCR was expanded to commercial labs in July 2022 through the Laboratory Response Network. 11 Since testing expansion, cases in the United States have now been reported in all 50 states, the District of Columbia, and 1 US territory, as well as over 90 other non-endemic countries. These new cases revealed new epidemiological patterns, including high prevalence among gay or bisexual men, or any men or transgender people who have sex with men or transgender people, and their sexual partners 12 ; higher rates of sexually transmitted infections, 13 fomite contamination,14,15 caregiver exposure, 16 health care exposure, 17 - 19 incarceration, 20 perinatal infection 21 ; as well as no known epidemiological risk factors.22,23 People at risk for severe disease include those with immunocompromising conditions and chronic skin disorders such as eczema. Two cases of mpox-associated encephalomyelitis without underlying immunocompromising conditions have been reported though. 24
Vaccination is useful for pre-exposure and post-exposure prophylaxis. JYNNEOS is a replication-deficient live vaccinia virus that is administered subcutaneously. ACAM2000 is a replication-competent live vaccinia-clonal derivative virus vaccine that is administered by skin scarification and is in active use by the military and laboratory personnel for smallpox. These were made available by the US Department of Health and Human Services as part of the National Mpox Vaccine Strategy for the mpox public health emergency 2022. 25 As JYNNEOS administration rate is limited by manufacturing supply, intradermal administration was authorized for emergency use by the US Centers for Disease Control and Prevention (CDC) to increase available supply, as the equivalent volume for 1 subcutaneous vaccine can provide 4 to 5 intradermal doses. Scheduled appointments to ensure utilization of each dose in a vial, generally, 4 to 5 at a time, are recommended to limit waste. Eczema vaccinatum akin to that from smallpox eradication days 26 has not been reported in the current mpox outbreak to date.
Tecovirimat, a novel orally bioavailable antiviral that targets the highly conserved orthopoxvirus VP37 protein to inhibit development and release of mature virions, is US Food and Drug Administration (FDA)–approved for the treatment of smallpox (variola). Tecovirimat has also been made available in the United States for the treatment of mpox through a protocol held by the CDC under the FDA-regulated Expanded Access Investigational New Drug mechanism. 27 Access to tecovirimat is improving nationally through the Strategic National Stockpile via the CDC.
Co-infection of mpox, HIV, and syphilis has only been reported 3 times to date. 28 - 30 Here we report a case of a person with uncomplicated well-controlled HIV who contracted mpox and secondary syphilis. The efforts made in a community hospital setting to diagnose and treat these infections in the setting of a public health emergency are discussed.
Case Presentation
A 35-year-old man with uncomplicated well-controlled HIV on bictegravir, emtricitabine, and tenofovir alafenamide presented to the emergency department with multiple facial lesions for 7 days. The first lesion was a small pustular “bump” on his lip. He subsequently developed 4 more lesions on his lips and chin before seeking an evaluation by a physician. Lesions were flesh-colored with a central concavity and small pustular discharge (Image 1). They were non-pruritic and non-tender. Concurrent with the onset of these lesions, he also developed mild right-sided submandibular pain associated with lymphadenopathy.

Monkeypox lesions at presentation, 7 days after symptom onset.
The patient identifies as bisexual with a history of oral and anal intercourse. The last sexual contact was 2 months prior. He then traveled to Hawaii 3 weeks prior to the index presentation and attended a large public gathering.
Lesions were swabbed for mpox virus using viral culture media and sent to a contracted commercial reference lab for qualitative PCR testing. He returned home to self-isolate. Results were available after 4 days. The reference lab notified the local public health department per Laboratory Response Network protocol. Public health officials informed the patient’s infectious disease physician who arranged a clinic visit following infection control precautions as recommended by the CDC.
At the clinic visit, his original facial lesions had increased in size and central crusting (Image 2). He had also developed a new diffuse maculopapular rash on the neck, chest, and torso with a herald patch on the right chest (Image 3). Early eczema versus secondary syphilis was the concern at this time. His lymphocyte CD4 count was 588 per cubic millimeter (reference range, 500-1500), and his HIV viral load was undetectable.

Monkeypox lesions with a maculopapular rash 10 days after symptom onset.
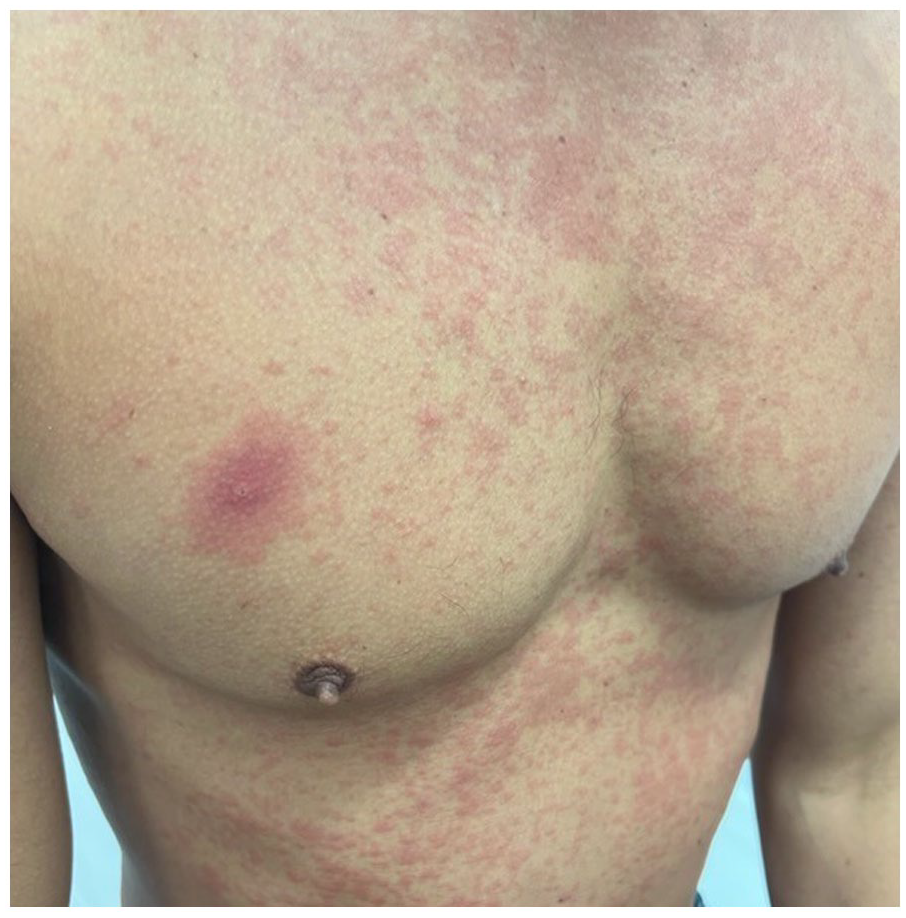
Maculopapular rash with herald patch on right chest.
After the patient was provided informed consent for use of tecovirimat for treatment of non-variola orthopox virus infection under CDC Expanded Access Investigational New Drug Protocol No. 116 039 (IRB No. 6402), he was started on tecovirimat 600 mg twice a day for 14 days. A small supply had already been arranged by the pharmacy department in coordination with the CDC, California Department of Public Health, and Kern County Public Health Department a few weeks in advance for possible local outbreaks.
Co-infection testing eventually revealed reactive syphilis antibody, reactive Fluorescent Treponemal Antibody Absorption test, and reactive Rapid Plasma Reagin test (RPR) with titer 1:1. Through California Reportable Disease Information Exchange, no prior treatment history for syphilis was found. Syphilis was staged as secondary syphilis. He was treated with 2.4 million units of benzathine penicillin G intramuscularly once. Urine gonorrhea and chlamydia nucleic acid amplification testing was negative.
At follow-up 1 week (Images 4 and 5) and 3 weeks (Images 6 and 7) after treatment initiation, he showed improvement and subsequent resolution of all symptoms. Isolation was lifted after all lesions had resolved, 4 weeks after symptom onset.

Monkeypox lesions 17 days after symptom onset, 7 days after treatment initiation.

Seven days after treatment initiation, maculopapular rash has completely resolved, and herald patch on right chest is improving.

Monkeypox lesions 1 week after treatment completion.
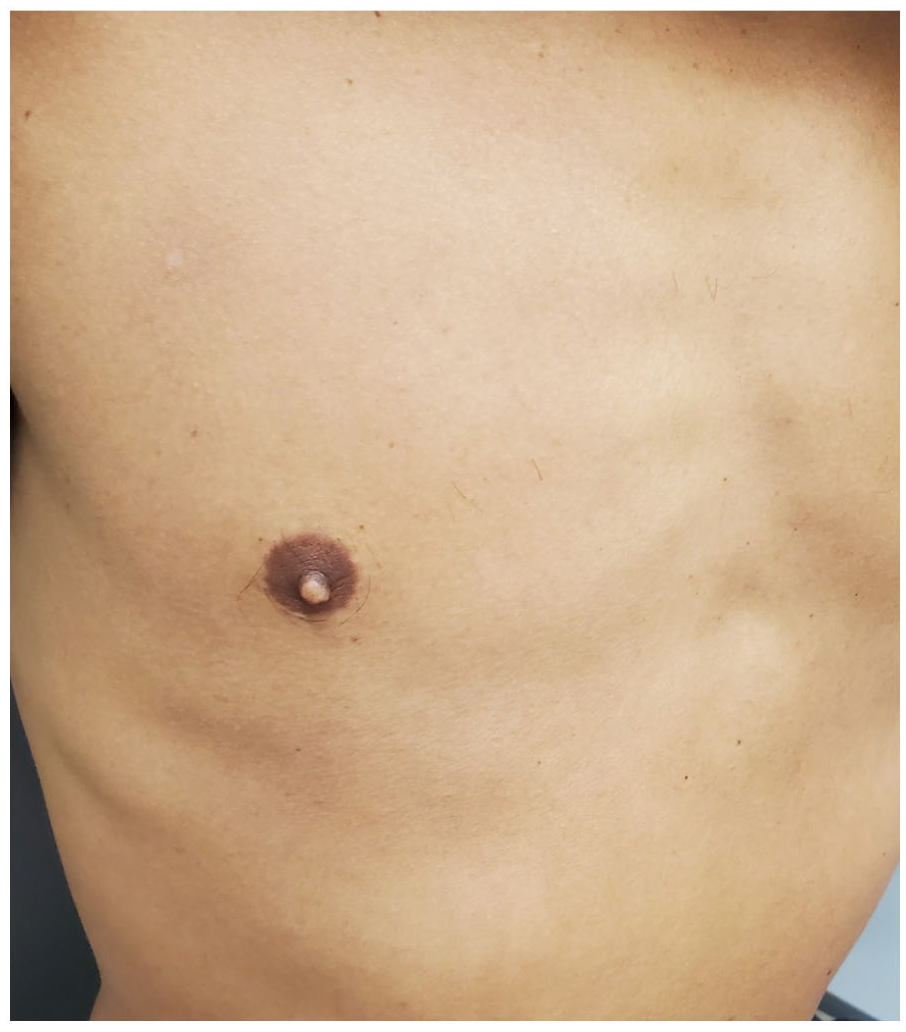
Herald patch is almost completely resolved 3 weeks after penicillin administration.
Discussion
This patient represents the first mpox diagnosis at our community institution, months after the mpox public health emergency had been identified at major metropolitan centers. This patient tested the readiness of the preparation plan for mpox at our institution and, in retrospect, offered a critique of the health care system at a local level.
Our patient would have benefited from receiving JYNNEOS as he was at high risk to contract mpox. However, for the months prior to his presentation, the supply of vaccines was still limited in this county and only available at certain public health vaccine clinics by appointment.
Our patient sought medical care for mpox within 1 week of symptom onset at an emergency department. Any earlier entry to care would require improved contact tracing to inform exposed contacts and offer post-exposure prophylaxis. He had not been contacted as part of contact tracing efforts. He denied close contact with others since returning from his trip to Hawaii.
The expansion of rapid diagnostic testing to commercial laboratories did help to diagnose him readily. As part of the expansion, testing at many points of initial health care contact, including urgent care clinics, emergency departments, as well as labor and delivery and primary provider offices is now possible. However, a 4-day turnaround for results leaves some room for improvement.
A strong clinical pharmacy team secured a supply of tecovirimat through the Strategic National Stockpile before our first patient arrived. This required coordination with the Kern County Public Health Department, the California Department of Public Health, and the CDC. There is a lack of data to date about the effectiveness of tecovirimat in people with mpox and which patients might benefit the most from tecovirimat. There is also a concern for the development of resistance to tecovirimat from selective pressure with widespread use given its relatively low barrier to resistance. The decision to treat with tecovirimat was based on his HIV status and the onset of a new maculopapular rash that could have been early eczema or secondary syphilis without further testing.
Regular in-person follow-up of patients with mpox is not routine; however, it may yield the most therapeutic benefit given the stigma attached to mpox may lead at-risk individuals to avoid seeking or receiving comprehensive care. Secondary syphilis was eventually diagnosed by physical examination on follow-up. Two additional in-person follow-ups helped alleviate concerns and provide moral support to an otherwise vulnerable patient.
The majority of California’s mpox experiences are in major metropolitan centers, such as Los Angeles, San Francisco, and San Diego, where public health resources are concentrated per capita. However, geospatial social determinants of health were found to contribute to health inequities as measured by Healthy Places Index, 31 and greater health inequities were associated with higher rates of sexually transmitted infections such as syphilis. 32 Central Valley of California scored low on the Healthy Places Index, which correlated with greater health inequities and is therefore at risk for more sexually transmitted infections, including syphilis. As such, during this mpox outbreak, co-infection testing of sexually transmitted infections is important, particularly in areas of greater health inequities.
Conclusion
Our case shows that patients diagnosed with mpox may need close follow-up to evaluate for new skin lesions and/or sexually transmitted co-infections that may manifest later. Preparation for the care of patients with mpox in a community hospital should follow that at the national level. It starts with the utilization of testing that is widely available now, physician education of signs and symptoms, and a ready supply of appropriate antivirals and personal protective equipment. Further studies need to be performed to validate whether individuals previously diagnosed with sexually transmitted infections such as HIV and syphilis are at higher risk for contracting the mpox virus. Such studies could lead to more effective identification of high-risk patients and more rapid intervention, treatment, and possibly decreased burden of disease.
Footnotes
Acknowledgements
The authors wish to acknowledge the Kern County Public Health Department for their efforts during the mpox outbreak in Kern County. This case is submitted to American Federation of Medical Research’s Western Conference, Carmel, CA, in January 2023.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval to report this case was obtained from the Kern Medical Institutional Review Board (IRB 22132).
Informed Consent
Written informed consent was obtained from the patient for their anonymized information and photography to be published in this article.
