Abstract
Idiopathic intracranial hypertension (IIH) is a rare cause of headache and visual disturbance with no known association with sickle cell disease (SCD). We present a patient with SCD with gradual onset of a severe headache and visual changes. Brain magnetic resonance imaging, angiography, and venography were nondiagnostic. Lumbar puncture established a diagnosis of IIH, and the patient had rapid improvement with large-volume lumbar puncture and acetazolamide. To our knowledge, this is the first case of IIH in a nonobese adult with SCD taking hydroxyurea, suggesting an association between SCD or hydroxyurea use and IIH. Furthermore, her clinical course demonstrates the complexities of treating IIH in patients with SCD.
Background
Headache is a common complaint in individuals with sickle cell disease (SCD), with more than one-third reporting headache at least weekly; headache types in SCD mirror the general population, with tension and migraine being the most common. 1 In addition, persons with SCD may experience headache secondary to obstructive sleep apnea, bone infarction, and medication overuse, as well as higher acuity causes such as cerebral infarction, dural venous sinus thrombosis, intracranial hemorrhage, posterior reversible encephalopathy syndrome, and moyamoya disease. 2
Idiopathic intracranial hypertension (IIH) is a rare cause of headache and visual disturbance in the general population, and there is no known association with SCD. The most well-recognized risk factor for IIH is obesity; in fact, IIH is exceedingly rare in nonobese individuals, with >90% of cases occurring in those with a body mass index ≥30 kg/m2.3,4 To date, 4 case reports of IIH involving 8 patients with SCD (7 pediatric and 1 obese adult) have been published.5-8 This report represents the first case, to our knowledge, of intracranial hypertension in a nonobese adult with SCD.
Case Presentation
Our patient was a 22-year-old woman with a history of hemoglobin SS SCD and well-controlled migraine headaches, who presented with gradual progression of headache over 3 days. Previous complications from her SCD included vaso-occlusive pain episodes, avascular necrosis of the bilateral hips and shoulders, and gallstones. She had never had a stroke or other central nervous system manifestation of her SCD. Her only medications were hydroxyurea 1500 mg daily and folic acid 1 mg daily. Her headache was intermittent, holocranial, exacerbated by positional changes, and not typical of her previous migraines. She experienced intermittent blurred vision and tinnitus concomitant with headache. Her symptoms became unremitting and increased in severity, prompting her to present to the emergency department. Table 1 includes a detailed timeline of her case.
Case Timeline.

Clinical Findings
The patient was afebrile with normal vital signs. Her body mass index was 27.28 kg/m2. She did not appear in any distress. Neurologic examination did not reveal any neurologic deficits. Dilated fundoscopic examination revealed bilateral papilledema, which raised concern for increased intracranial pressure (ICP).
Diagnostic Assessment and Therapeutic Intervention
Computed tomography (CT) of the brain did not reveal any obstructing mass lesions. Given concern for elevated ICP, a lumbar puncture (LP) was performed; the opening pressure was elevated at 45 cm H2O. The remainder of cerebrospinal fluid (CSF) studies were within normal limits and are shown in Table 2. This prompted magnetic resonance imaging (MRI) of the brain without contrast to rule out structural causes of increased ICP. The MRI revealed a partially empty sella turcica, prominent sheath space surrounding the bilateral optic nerves, and mild narrowing of the distal transverse sinuses bilaterally, which were nonspecific findings that are seen in a setting of increased ICP. Magnetic resonance angiogram of the head and neck without contrast showed no large vessel occlusion or hemodynamically significant stenosis. Magnetic resonance venography of the head with and without contrast revealed no dural venous sinus thrombosis.
CSF Results [Normal Values].
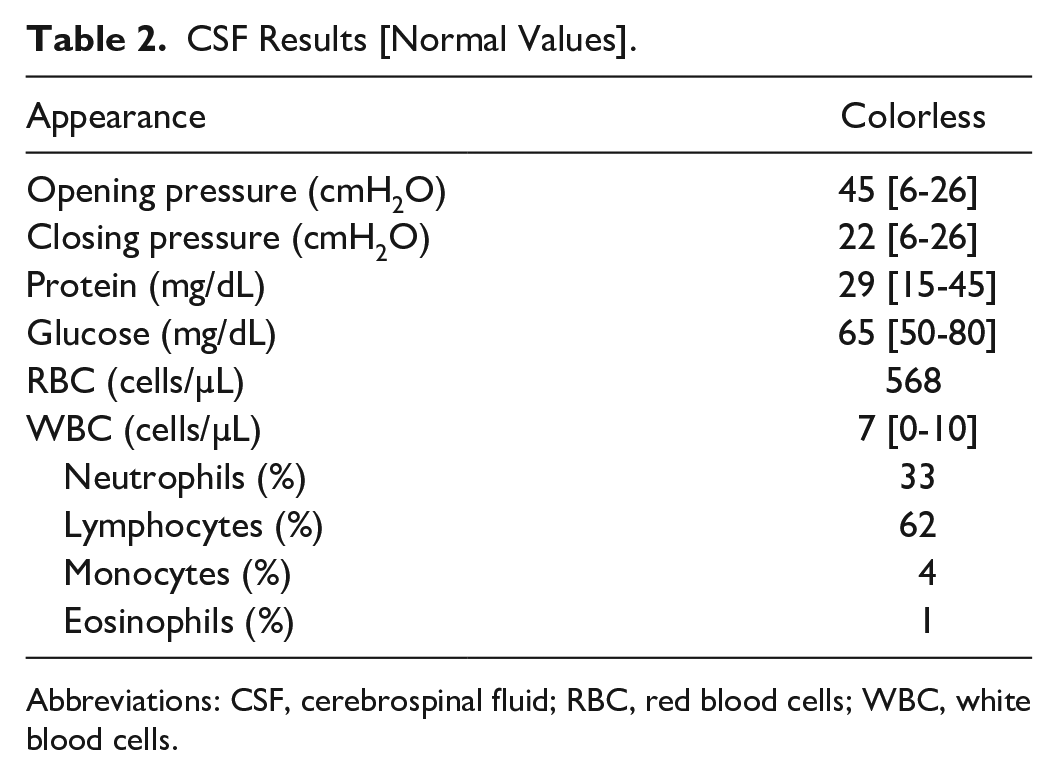
Abbreviations: CSF, cerebrospinal fluid; RBC, red blood cells; WBC, white blood cells.
As no secondary causes of intracranial hypertension were identified on neuroimaging or CSF studies, a diagnosis of IIH was made. Acetazolamide was initiated at 250 mg twice a day and the dose was increased to 500 mg twice a day. Symptoms improved but did not fully remit after starting acetazolamide, so topiramate was added as an adjunctive therapy. Topiramate was initiated at 25 mg daily and the dose was increased to 50 mg twice a day as an outpatient.
Follow-up and Outcomes
The patient followed up with ophthalmology, neurology, and hematology. Her optic disc edema resolved. Her IIH remained in remission on acetazolamide. The patient adjusted her regimen to 1 week on, 1 week off acetazolamide (500 mg twice a day) due to an increase in vaso-occlusive pain episodes while on medication. She continued topiramate 50 mg twice a day.
Discussion
In this case, we discuss a nonobese adult female with SCD taking hydroxyurea who presented with headache, tinnitus, vision changes, and was diagnosed with IIH. The diagnosis of IIH is made according to the updated modified Dandy criteria, which require an elevated ICP on LP with normal CSF composition and no structural etiologies for elevated ICP on neuroimaging. 3 Many risk factors for IIH have been established, including medication use (tetracycline antibiotics, retinoids) and medical disease (endocrinopathies, leukemia, iron deficiency anemia), but the link between SCD or hydroxyurea use and IIH remains unclear. 9 Among all previously published cases of IIH in patients with SCD, the patients have been either prepubertal or obese.5-8
The pathophysiology of IIH is poorly understood but is thought to involve decreased blood flow through and subsequent absorption of CSF into the arachnoid granulations. 4 Any disease process that disrupts this normal flow through arachnoid granulations can result in increased ICP. The propensity of sickled cells to aggregate and cause vascular damage offers a theoretical link between IIH and SCD. 10 In addition, hydroxyurea increases red blood cell mass and blood viscosity, which may predispose to IIH. 5 Whether SCD or hydroxyurea use should be considered risk factors for IIH has not yet been established.
Carbonic anhydrase inhibitors (CAIs) such as acetazolamide remain the cornerstone of treatment for IIH. Carbonic anhydrase inhibitors are thought to exert their effect by decreasing CSF production in the choroidal plexus. 11 The optimal dose of acetazolamide in IIH is a total daily dose (TDD) of 2 to 4 g. 12 Consequences of CAI use include volume depletion and metabolic acidosis, which have been linked to an increased risk for vaso-occlusive episodes in SCD.13,14 Our patient had to reduce her dose of acetazolamide due to an increase in vaso-occlusive episodes once starting the medication, resulting in a TDD lower than the 2-4 g target. 12 While off-label use of alternative CAIs such as topiramate and methazolamide in IIH is common, evidence for their safety and efficacy in the SCD population is lacking.3,13,14
While IIH is an uncommon diagnosis in those with SCD, all previously reported cases of IIH in SCD adults occurred in those with comorbid obesity. The diagnosis of IIH in a nonobese SCD adult taking hydroxyurea raises the possibility that SCD or associated hydroxyurea use may be risk factors for IIH. Moreover, treatment of IIH in patients with SCD may be complicated by CAI-induced volume depletion and acidosis, leading to an increase in vaso-occlusive episodes. If unrecognized or untreated, IIH can have devastating consequences including permanent vision loss. 3 Treatment should be guided by a multidisciplinary team of neurologists, hematologists, and ophthalmologists to maximize therapeutic benefit while reducing impact on SCD symptom burden.
Footnotes
Acknowledgements
The authors wish to thank Sherri Bogard, MD; Jasmah Hanna, MS; and Manpreet Malik, MD for their support and assistance. The authors also wish to acknowledge the Emory University Division of Hospital Medicine Open Access Publishing Fund for supporting dissemination of this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: No specific funding was received for the clinical conduct or writing of this work. The Emory University Division of Hospital Medicine Open Access Publishing Fund provided funding for open access publication.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for her anonymized information to be published in this article.
