Abstract
Extrahepatic metastasis of hepatocellular carcinoma (HCC) to the head and neck is unusual, especially the oral cavity/maxillofacial region. Metastatic HCC to the hard palate, however, is particularly rare. The most common site of HCC metastasis is the lung, followed by lymph nodes, bone, and adrenal gland. Importantly, oral cavity metastatic HCC may be misdiagnosed as a primary malignancy, such as a salivary gland carcinoma. In this article, we describe a young woman with metastatic HCC to the hard palate that was initially diagnosed as an acinic cell carcinoma.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is the most common primary liver malignancy, comprising approximately 70% to 90% of hepatocellular malignancies. Hepatocellular carcinoma is much more common in men than in women. In men, HCC is the second leading cause of cancer death worldwide and in developing countries. In developed countries, it is the sixth leading cause of cancer death among men. 1 Common causes include Hepatitis B or C, metabolic disorders, and alcohol, which can each lead to cirrhosis and thus increase the risk for HCC. 2 Extrahepatic metastasis occurs in approximately 30% to 50% of HCC patients. Although extrahepatic metastasis of HCC is relatively common, extrahepatic metastasis as an initial presentation is uncommon, and it is recognized as an advanced disease with poor prognosis.
Case Presentation
A 32-year-old woman was referred to the oral surgery department at our institution for a painful hard palate mass (Figure 1A). Her pertinent past medical history included arthritis, asthma, and congestive heart failure. Her social history included tobacco use and mild alcohol consumption. She initially presented to her primary care physician approximately 1 month prior for complaints of nasal congestion and sinus pressure and cough. She was prescribed amoxicillin / clavulanic acid for suspected frontal sinusitis. Two weeks later, she noticed a small ulcer on her hard palate for which she sought care at the local dentist who recommended close observation. She was subsequently referred to otolaryngology where an exophytic hard palate lesion was noted and was biopsied. The initial pathology returned as acinic cell carcinoma. Laboratory testing on presentation showed the complete blood count was unremarkable, apart from a mildly low platelet count of 140 × 109/L. The complete metabolic panel demonstrated a mild hypokalemia of 3.1 mEq/L, a calcium level of 10.6 mg/dL, a bilirubin of 1.3 mg/dL, and an albumin of 3.3 g/dL. The AST (aspartate aminotransferase) and ALT (alanine transaminase) were unremarkable. Alkaline phosphatase was 113 IU/L. A maxillofacial computed tomography (CT) showed a 3.6 × 3.2 cm2 heterogeneous mass centered along the anterior aspect of the hard palate with penetration into the left nasal cavity (Figure 2A). Subsequent positron emission tomography (PET) CT skull to mid-thigh showed additional findings including cirrhotic appearing liver with ascites and multiple hypermetabolic hepatic masses (Figure 2B).
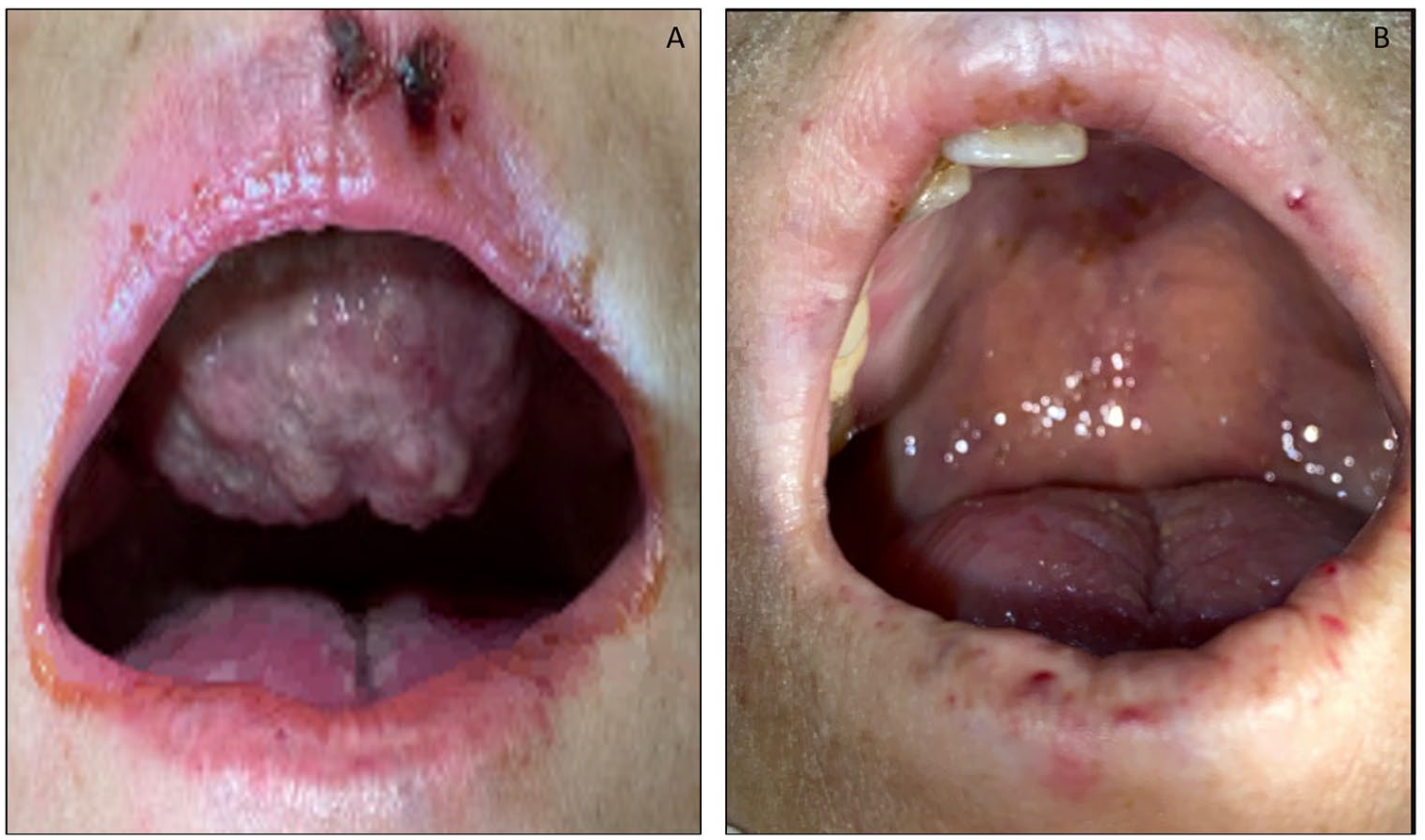
Photographs of patient before treatment demonstrating exophytic hard palate mass (A); after treatment the mass is no longer grossly visible (B).

(A) CT maxillofacial coronal image demonstrating tumor extending into the left aspect of the nasal cavity from the palate (white arrow). (B) PET scan demonstrating multifocal hypermetabolic masses within the cirrhotic liver. Ascites is present (white arrow).
Our pathology department received biopsies from the liver and pelvic masses. Both biopsies demonstrated a proliferation of tumor cells with eosinophilic to clear cytoplasm in trabecular and nesting patterns (Figure 3E, 3F, 3H). The palate mass biopsy from the outside institution was reviewed and showed similar findings (Figure 3A-3B). The tumor cells were diffusely moderately to strongly positive for pankeratin AE1/AE3, keratin OSCAR, and CAM5.2; patchy weakly positive for arginase-1 (Figure 3D) and CK7; and negative for PAS-diastase, HepPar 1, glypican3, synaptophysin, chromogranin, DOG-1, S100, and p63. Albumin in situ hybridization (ISH) was strongly diffusely positive within the tumor cells (Figure 3C). Together with the morphology, immunohistochemistry, and the ISH findings, the diagnosis of moderately to poorly differentiated HCC was rendered. The background liver showed cirrhosis with mild steatohepatitis and focal cholestasis (Figure 3G). Hepatitis B surface antigen test was negative.
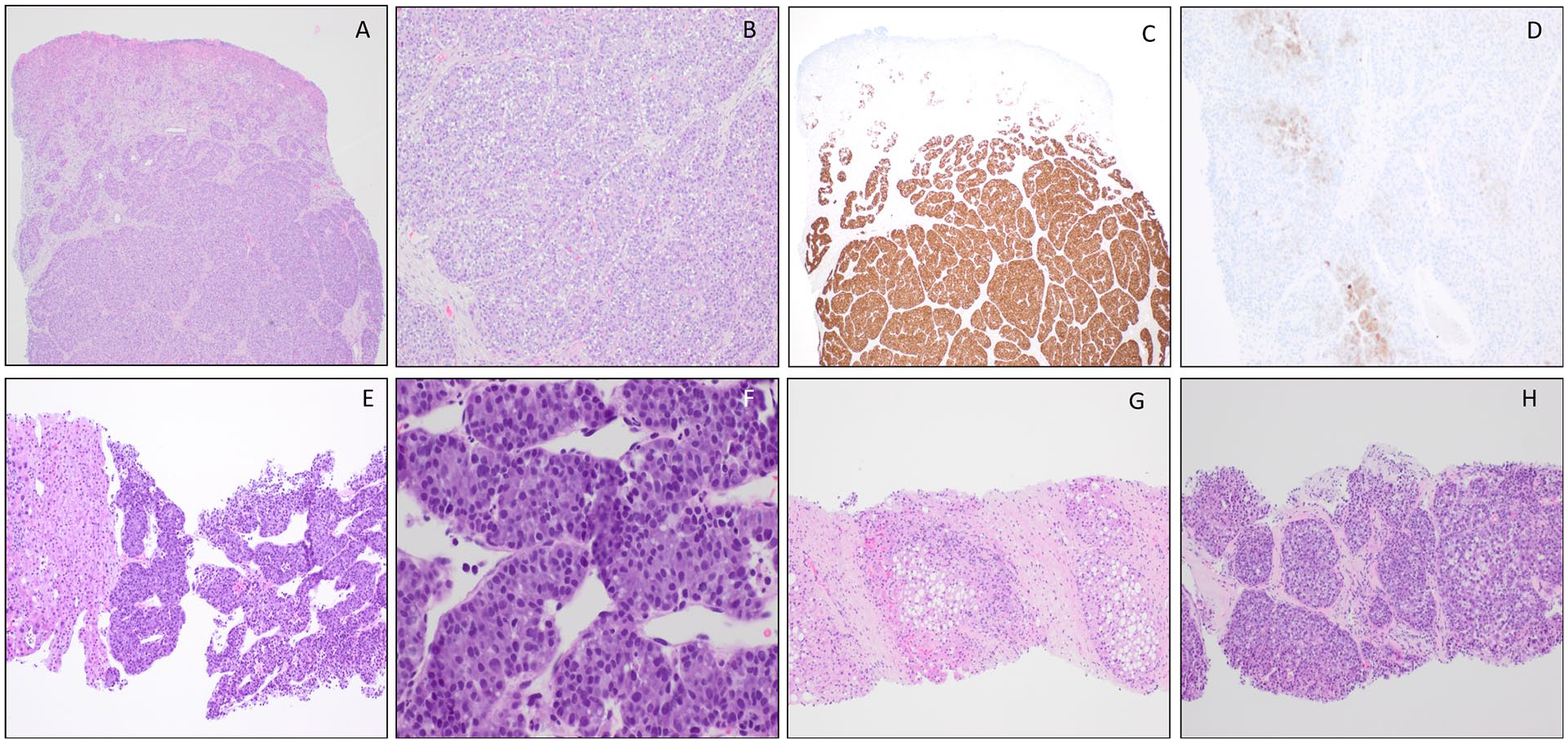
(A) Ulcerated hard palate metastatic tumor (hematoxylin-eosin, 40×). (B) Hard palate tumor with trabecular and nesting patterns (hematoxylin-eosin, 100×). (C) Hard palate tumor diffusely strongly positive for albumin in situ hybridization (40×). (D) Hard palate tumor focally weakly positive for arginase immunostain (100×). (E) Hepatocellular carcinoma with adjacent liver tissue (hematoxylin-eosin, 100×). (F) Hepatocellular carcinoma with trabecular pattern (hematoxylin-eosin, 400×). (G) Background liver with cirrhosis (hematoxylin-eosin, 100×). (H) Metastatic pelvic tumor (hematoxylin-eosin, 100×).
In preparation for radiation therapy, she had significant bleeding after extraction of a single tooth and received 2 units of packed red blood cells over 2 days. She received local radiation therapy. She received concurrent atezolizumab (Tecentriq) beginning with her radiotherapy which continued after radiation was completed. During the course of her treatment, she had episodes of bleeding from the tumor requiring transfusion. On follow-up 1 month after radiotherapy was completed, she reported palliative benefit and demonstrated local tumor control along the hard palate where the tumor was no longer visible (Figure 1B). Nevertheless, she also had significant ascites, which continued to accumulate despite multiple paracentesis procedures, suggestive of portal vein thrombosis. Her condition declined rapidly, and she agreed to hospice. While on hospice, the oral cavity tumor recurred and partially obstructed her airway. After 2 weeks in hospice, she passed away.
Discussion
Hepatocellular carcinoma is the most common primary liver cancer with a relatively high rate of metastasis. Globally, it is ranked as the sixth in terms of incidence and fourth in terms of mortality. Hepatocellular carcinoma is classically found in the background of cirrhosis, although it can arise in any form of liver disease. Extrahepatic metastasis of HCC is relatively common with lung, bone, and abdominal lymph nodes being the most common sites of distant metastasis. Metastasis to the head and neck is unusual and even fewer cases metastasize to the oral cavity.3,4
The oral cavity is indeed an uncommon site for metastatic tumors in general but is usually indicative of widespread disease. A study of metastatic tumors by Hirshberg et al 5 showed that oral metastases were the first sign of metastatic spread in 25% of cases, and in 23% they were the first indications of undiscovered malignancies at distant sites. Metastasis can be found anywhere in the oral cavity; however, the jaw is twice as common as the oral mucosa/soft tissue. Within the jawbones, the mandible is more frequently involved than the maxilla, with the molar area being the most frequent site. 6 In the oral soft tissue, the gingiva is the most commonly affected site followed by the tongue.4-7 The study of 673 metastatic tumors to the oral cavity by Hirshberg et al found the following: for the jaw and surrounding soft tissue, lung cancer was the most common metastatic tumor in men; however, breast cancer was the most common metastatic tumor in women. Conversely, studies by Chen et al 3 and Lim et al 7 stated that the liver cancer was the most common tumor to metastasize to the oral soft tissue. The clinical presentation of metastatic lesions differs depending on the sites in the oral region. In the jawbones, most patients complain of swelling, pain, and paresthesia which can progress rapidly. For metastasis to the oral soft tissue, patients can present with an ulcer or a painful, bleeding lesion that can mimic hyperplastic/reactive lesions such as pyogenic granuloma and peripheral giant cell granuloma, or benign tumors such as hemangioma and peripheral fibroma.3,5
In the limited cases of oral cavity metastatic HCC, the most common sites are the mandible and gingiva. There are 2 possible pathways for the oral cavity metastatic HCC. The first possible pathway is through the hepatic artery or portal vein in which dissemination to the lung would occur first and then the oral cavity region. The second possible pathway is through vertebral venous plexus (Batson’s plexus) which would bypass the lungs. 8 Jaw HCC metastasis is more common in men with average age of approximately 57 years old. 9 Mandibular metastasis can involve the angle, premolar, and condylar regions. It is proposed that it may be related to the branching of the blood vessels within the bone or to local slowing of blood flow. 8 Gingival metastatic HCC is more likely to involve the upper gingiva, possibly due to the anatomical characteristic of the arteries supplying the blood to the gingiva. 4
Although oral cavity metastatic HCC is quite rare, it is not infrequently the initial presentation of HCC in previously reported studies, from 25% to 72%.3,4,10 In our case, however, the patient initially presented with a metastatic HCC to the hard palate, which is exceptionally rare. To the best of our knowledge, only one metastatic HCC to the hard palate has been reported in the literature. 11 The previously reported metastatic HCC to the hard palate case was a 50-year-old man with chronic alcoholic and hepatitis B with cirrhosis. The metastatic tumor also involved the left maxillary alveolar process, and the patient passed away 1 week after the diagnosis due to hepatic and renal failure. The diagnosis in our case was slightly more elusive due to the young age and lack of liver disease history, as well as initial diagnosis of acinic cell carcinoma on the first biopsy from the hard palate. The diagnosis of metastatic HCC was confirmed with positivity of tumor with arginase immunostain and albumin ISH. As a matter of comparison, there is some morphologic overlap between HCC and salivary gland tumors, specifically acinic cell carcinoma which has variable morphologic features including tumor cells with eosinophilic and clear cytoplasm as in our case. This is especially poignant given the limited sampling and/or limited immunohistochemical panels. Of note, there was 1 reported case of previous metastatic HCC to the parotid gland which was also initially diagnosed as acinic cell carcinoma. 12 There are several markers of hepatocellular differentiation with varying sensitivity and specificity, including arginase-1, hepatocyte paraffin 1 (HepPar 1), polyclonal CEA (carcinoembryonic antigen), and CD10. Among these immunostains, arginase is the most sensitive marker of HCC and shows high sensitivity even in poorly differentiated HCC and scirrhous HCC. 13 Albumin ISH is a marker of primary hepatic tumors, including hepatocellular and cholangiocarcinomas. 14 Metastatic HCCs to the oral cavity in the literature vary from well differentiated to undifferentiated, but is more commonly moderately differentiated.4,8
Hepatocellular carcinoma in young adults is much less common than in older adults. It is often discovered at an advanced stage due to lack of follow-up and relatively good liver function in young adults. The cause of HCC in young adults is most commonly due to chronic hepatitis B infection. 15 Most of the reported oral cavity metastatic HCC had pre-existing hepatic disease; however, due to limited number of cases, the most common cause of the HCC with this metastatic location still cannot be determined. 4 The cause of the underlying cirrhosis in our case was unknown.
Oral cavity metastatic HCC has a poor prognosis. The median survival time following diagnosis is 4 to 12 months.2,9 Although local radiation therapy and systemic control with chemotherapy or immunotherapy may result in improved quality of survival, in most cases, the management of oral cavity metastatic HCC is mainly focused on pain relief and management of complications such as bleeding. 8
Due to the rarity of oral cavity metastatic HCC, the diagnosis is challenging for both the clinician and the pathologist. In any case where the clinical presentation is unusual such as a persistent or rapid growth of the lesion and/or a previous history of malignancy, consideration of a metastasis as part of the work-up is prudent.
Conclusion
Metastatic HCC to the oral cavity/maxillofacial region represents a rare occurrence. It has significant morbidity and mortality with a low survival rate. Therefore, it should remain within the differential for patients presenting with persistent or rapidly growing lesions within the oral cavity, as well as a thorough review of systems and underlying conditions. A thorough examination including imaging, biopsy, and immunohistochemical staining are essential in diagnosis.
Footnotes
Acknowledgements
We thank Dr Hee Eun Lee, MD, PhD, at Mayo Clinic in Rochester, MN, for her expertise in consultation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical/IRB approval for reporting individual cases.
Informed Consent
Informed consent for patient information to be published in this article was not obtained as the patient is deceased and contact has been lost with the patient’s family.
