Abstract
Primary hepatic angiosarcoma (PHA) is a rare and aggressive mesenchymal liver tumor with a poor prognosis and high mortality. Treatment options are limited to palliative chemotherapy with surgical resection reserved for the few cases that present early. We present a case of a patient who presented with jaundice and elevated liver enzymes. Imaging identified a diffusely heterogeneous liver consistent with cirrhosis, findings of portal hypertension, and 2 ill-defined liver lesions. Biopsy results confirmed PHA. Primary hepatic angiosarcoma does not have a typical presentation but should be considered for any patient presenting with an infiltrative liver mass.
Introduction
Primary hepatic angiosarcoma (PHA) is a rare and aggressive mesenchymal liver tumor with a poor prognosis and high mortality.1,2 Diagnosing PHA is challenging as it accounts for only 0.1% to 2% of primary malignant liver tumors. 3 Given its poor prognosis, treatment options are often limited to palliative chemotherapy with surgical resection only reserved for the few cases that present early.2,4
Case Report
An 84-year-old man with a history of prostate cancer presented to his primary care clinic with jaundice. He reported pruritus and anorexia for several weeks. He had no history of liver disease or heavy alcohol use. His initial workup revealed aspartate aminotransferase (AST) 113 U/L, alanine aminotransferase (ALT) 59 U/L, alkaline phosphatase (ALP) 196 U/L, total bilirubin 5.3 mg/dL, direct bilirubin 2.9 mg/dL, and international normalized ratio (INR) of 1.5. Testing for hepatitis B and C was negative. A computed tomography (CT) scan showed diffuse heterogeneity of the liver without a discrete mass. The liver had a nodular appearing contour and ascites was identified. Spleen was normal. A follow-up magnetic resonance imaging (MRI) revealed a diffusely heterogeneous liver with a cirrhotic morphology, small amount of ascites, and 2 ill-defined liver lesions (6.3 × 5.9 cm and 3.3 × 1.1 cm). These areas were isointense to the surrounding liver on delayed phase enhancement. There was no evidence of portal vein occlusion or intra-abdominal metastasis. Alpha-fetoprotein and carbohydrate antigen 19-9 were normal. Urgent biopsy of the mass was recommended; however, biopsy was delayed due to patient hesitancy and coordination at a local facility per the patient preference. Bilirubin rose to 12.8 mg/dL on interval labs and a repeat MRI about a month after the first MRI revealed similar sized lesions with an additional lesion noted (Figure 1). There was a small area focal biliary dilation around the largest lesion, but otherwise no large bile duct obstruction to explain the rising bilirubin.
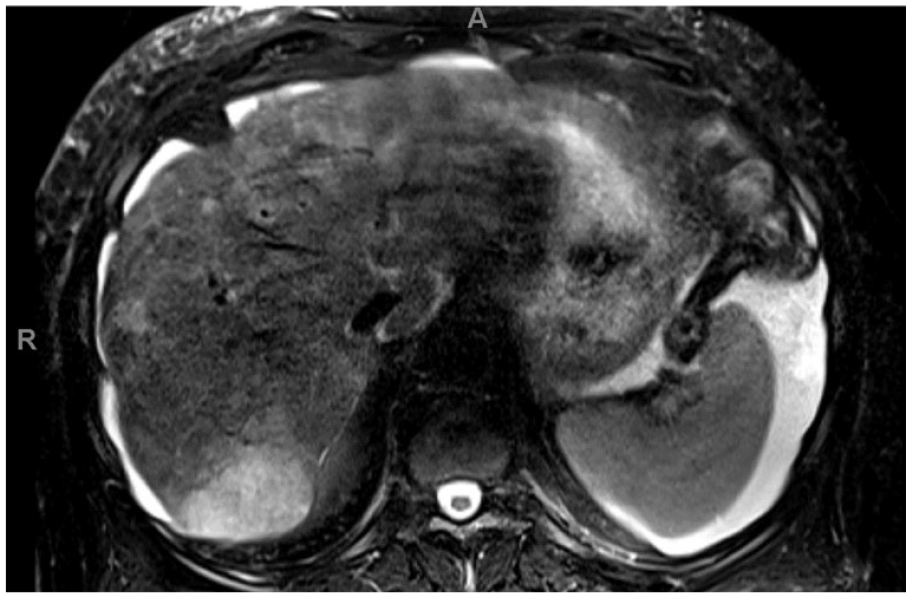
T2 axial magnetic resonance images of the abdomen, showing enhancing hepatic lesions (white arrows) favoring neoplasm (LI-RADS M: probably or definitely malignant, not necessarily hepatocellular carcinoma).
The patient was subsequently hospitalized for worsening fatigue, vomiting, abdominal distention, and lower extremity edema. Physical examination was remarkable for jaundice and a distended abdomen with a positive fluid wave. Liver enzymes had increased, notable for AST 171 U/L, ALT 79 U/L, ALP 268 U/L, total bilirubin 24.2 mg/dL, and direct bilirubin 20.8 mg/dL. The INR was now 2.6. Diagnostic paracentesis was consistent with portal hypertension and negative for infection.
He underwent CT-guided percutaneous liver biopsy. Pathology results were consistent with angiosarcoma. Atypical cells were positive for ERG and CD31, further supporting the diagnosis. Background liver elements were highlighted by HEP1, CK7, and CK20 (weak). Glypican 3, NKX3.1, and CAMTA1 immunostain were negative (Figure 2A-C). Given the diagnosis of PHA with extensive disease, abnormal liver function testing, and his overall frailty, he was not a candidate for surgical resection and was a poor candidate for palliative chemotherapy. The patient declined further aggressive care. He was ultimately discharged with home hospice and passed away 1 week later.

(A) Liver biopsy specimen showing tumor composed of infiltrative, anastomosing vascular channels lined by atypical pleomorphic cells with hyperchromatic nuclei (hematoxylin and eosin stain, 10× magnification). (B) Liver biopsy specimen showing tumor composed of infiltrative, anastomosing vascular channels lined by atypical pleomorphic cells with hyperchromatic nuclei (hematoxylin and eosin stain, 20× magnification). (C) Tumor sample with ERG immunostain highlighting tumor cells (10× magnification).
Discussion
Angiosarcoma is a rare malignant neoplasia accounting for about 3% of all sarcomas. The PHA accounts for less than 5% of all angiosarcoma cases and 0.1% to 2% of primary malignant liver tumors.3,5,6 In most cases, PHA is idiopathic or due to exposure to certain chemical agents (vinyl chloride, arsenic, thorium dioxide, and inorganic copper).
The PHA has variable clinical presentations and commonly presents with nonspecific manifestations such as abdominal pain, distention, fatigue, and weight loss. Jaundice and ascites are common signs in PHA, similar to our patient’s initial presentation.2,7 Laboratory findings are nonspecific and include elevated liver enzymes, hyperbilirubinemia, thrombocytopenia, and elevated INR as seen in our case. The tumor markers, alpha-fetoprotein, and carbohydrate antigen 19-9 can be mildly elevated; however, these are also nonspecific.8,9
Imaging studies, primarily CT and MRI, can help identify the location and extent of hepatic lesions, but no specific findings definitively diagnose PHA as it presents with various morphological features such as nodules, masses, or infiltrative lesions. Evidence of rapid lesion progression and growth on serial imaging is suggestive for PHA as was seen in our patient.6,7,10
Definitive diagnosis can only be achieved with liver biopsy and the vascular nature of the tumor can increase the risk of bleeding with the procedure. Important immunochemistry stains for PHA include CD31, CD34, von Willebrand factor, vascular endothelial growth factor, and Ulex europaeus agglutinin.2,5,7 Histological findings show vascular channels formed by spindle-shaped cells. 3 Of these, CD31 and von Willebrand factor are most sensitive in about 90% of PHA. 11 Our histological analysis showed atypical cells with immunohistochemical staining positive for CD31.
There are no current guidelines for PHA treatment; however, multiple treatment options are available in cases that are discovered early. The PHA is an absolute contraindication for liver transplant as it is associated with high recurrence rate posttransplant. 12 Zheng et al 8 showed that surgical resection alone or in combination with chemotherapy is associated with a 76.7% survival rate of more than 6 months, but median survival is only 17 months. Unfortunately, most cases are discovered at advanced stages and thus few patients undergo surgical resection. 13
Few studies have been conducted on the effectiveness of chemotherapy for PHA. Dannaher et al 14 reported improved survival using Adriamycin, cytotoxin, and methotrexate compared with Adriamycin alone. In another study by Kim et al, 15 a potential benefit of palliative chemotherapy in metastatic PHA was shown. Transcatheter arterial chemoembolization can be also used as palliative chemotherapy if local excision is not an option. 16 In addition, Huang et al 17 reported one patient who survived 24 months after receiving transcatheter arterial embolization followed by hepatic arterial infusion chemotherapy. In general, PHA patients have a poor prognosis as most are diagnosed with metastatic disease at the time of presentation with a mortality rate up to 50% in 6 months.6,18
In summary, PHA is a rare neoplasia associated with poor prognosis. Due to the aggressive nature of the tumor, patients present with advanced disease-limiting treatment options. The PHA does not have a distinct presentation, requires a pathological diagnosis, and should be on the differential for any patient presenting with jaundice and enlarging infiltrative liver masses.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
