Abstract
We report a case of a fetus with a prenatal diagnosis of classical congenital adrenal hyperplasia (CAH) due to 21-hydroxylase deficiency. Although CAH is typically assessed postnatally, this fetal case had multiple prenatal clinical assessments made feasible by an interdisciplinary CAH center. The approach facilitated the development and delivery of comprehensive and earlier care for the fetus, and the family living with this complex, congenital condition, with perinatology, endocrinology, genetic counseling, psychology, and urology involvement. As well, the addition of fetal MRI to standard ultrasound revealed significant deficits in the biparietal diameter, occipitofrontal diameter, and total intracranial volume of the fetal CAH brain. These early anomalies in the brain suggest that neurological comorbidities observed in older children and adults with CAH should be studied as early as prenatally, with the addition of fetal MRI to ultrasound potentially being useful for identifying and understanding prenatal anomalies in CAH.
Case Report
A 32-year-old female (gravida 4 para 3) presented to her obstetrician with her next pregnancy following the birth of a previous female child with classical congenital adrenal hyperplasia (CAH). The estimated fetal age was 15 weeks and 4 days at the time of presentation based on initial sonographic assessments.
At 19.42 gestational weeks (GWs), ultrasound revealed genitalia that were female-appearing but ambiguous or atypical. A karyotype indicated the fetus was 46,XX with no chromosomal abnormalities. CYP21A2 gene testing of DNA from amniotic fluid showed compound heterozygosity for an R356W mutation on one allele and a 30-kb deletion on the other allele, confirming a prenatal diagnosis of CAH due to 21-hydroxylase deficiency. Parental genetic testing confirmed the fetal genotype. The mother subsequently underwent multiple consultative and care appointments on a bi-weekly/monthly basis with endocrinology, genetic counseling, psychology, urology, and perinatology until delivery at the Fetal Maternal Center, and followed at our CAH Comprehensive Care Center. 1 This next pregnancy led to the surfacing of psychological issues for the proband sibling with regards to her external genitalia, and the related challenges the family experienced during her first year of life, prior to the establishment of our center. It was imperative to immediately address and resolve these issues for both the proband sibling and the family, prior to the arrival of the next affected female child, so that the experience for all family members would be much improved via therapy, education, and a supported effort by the care provider team for CAH.
At 29.14 GW, high-resolution prenatal ultrasound (General Electric, 5-9 MHz transducer) was performed and three serial 2D measurements of the biparietal diameter (71.1 mm; 20th percentile) of the fetus and two serial 2D measurements of the head circumference (258.1 mm; 3rd percentile) were averaged and compared with standard references using the Hadlock growth curves. 2
At 30.57 GW, fetal magnetic resonance imaging (MRI; Philips 1.5T) was performed to better evaluate clinically for other abnormalities as MRI can provide information regarding certain conditions like white matter abnormalities and temporal lobe atrophy that cannot be obtained by ultrasound alone. 3 2-D and 3-D MRI measurements of fetal brain structures were computed (Figure 1) and shown in Table 1. 2-D measurements of brain biparietal diameter, occipitofrontal diameter, skull biparietal diameter, and occipitofrontal diameter, and calculated head circumference were recorded. 4 Significant deficits were noted for skull biparietal diameter and occipitofrontal diameter versus age-matched controls (Figure 2).5,6 Absolute biparietal diameter measurements were similar for ultrasound and MRI. 3-D measurements were obtained of the extra-axial cerebrospinal fluid (CSF) volume, supratentorial volume, cerebellar volume, cortical plate volume, and total lateral ventricles volume (Figure 3). 4 Total intracranial volume and deep gray matter volume, but not developing white matter, showed deficits versus controls (Table 1).5-7 A postnatal follow-up MRI of the patient on day 10 of life showed a total brain volume (300.82 cm3) that was below the 5th percentile for age-matched, healthy infants. 8

Example of computing a 3-D volume of the fetal brain. (A) Multiple 2D stacks were acquired in the three principal directions (axial, sagittal, and coronal). (B) A 3-D fetal brain magnetic resonance imaging (MRI) image was reconstructed, following motion-correction, from the 2D stacks. (C) Major brain-tissue types were manually delineated from the reconstructed 3-D brain volume (indicated in various colors).
2-D and 3-D Regional Fetal Brain Measurements on MRI.

Abbreviations: GA, gestational age; MRI, magnetic resonance imaging; CSF, cerebrospinal fluid.
Percentile comparisons are not reported because transient tissue types result in varying definition of tissue boundaries within these regions and preclude accurate comparisons.
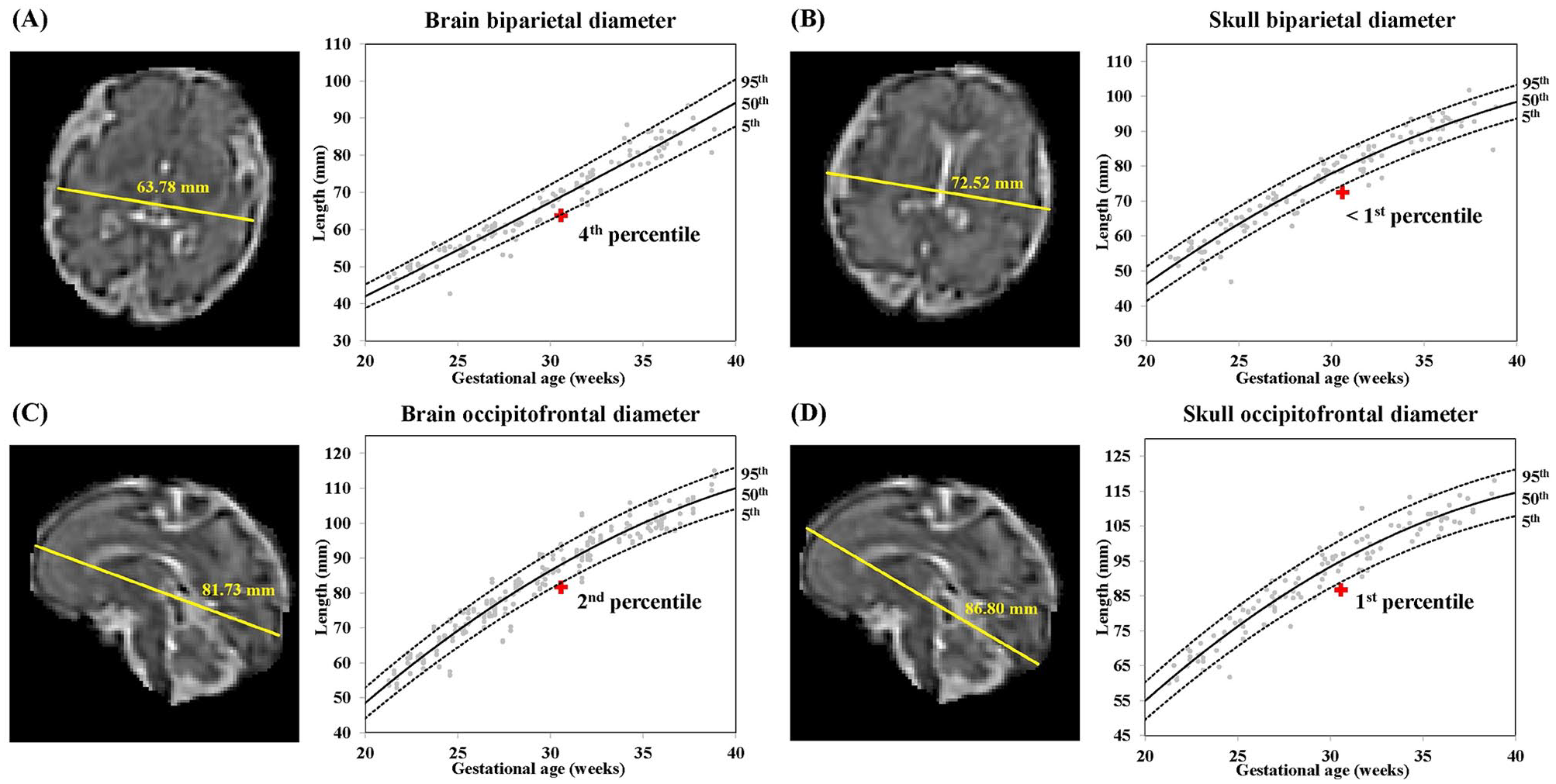
2D-MRI brain measurements of a fetus with congenital adrenal hyperplasia (CAH) at 30 weeks 4 days estimated gestational age. (A–D) Measurements of biparietal diameters and occipitofrontal diameters of the brain and skull (in mm). Red plus sign indicates percentile for measurements of the CAH fetus relative to normative fetal brain morphometry curves.

3D-MRI volumetric brain measurements of a fetus with congenital adrenal hyperplasia (CAH) at 30 weeks 4 days estimated gestational age. (A–E) Measurements of extra-axial cerebrospinal fluid, cortical plate, total lateral ventricles, cerebellum, and supratentorial brain tissue. Red plus sign indicates the percentile for the volumetric measurement in the CAH fetus relative to normative fetal brain volume curves. (F) Representative example of a sagittal brain slice showing the tissue regions measured (supratentorial brain tissue excluded).
The neonate was born at 39.14 GW, via uncomplicated vaginal delivery, and seen in the CAH clinic 3 days after birth. On physical examination, she was Prader stage 3, with clitoral width 1 cm, stretched clitoral length 1 cm, and noticeable hyperpigmentation and rugation of the labioscrotal folds. Her examination was otherwise unremarkable. She was a well-appearing neonate with no concerns for adrenal crisis. Hormone analytes were measured at the visit, prior to initiation of treatment, and included: 17-hydroxyprogesterone (17-OHP), androstenedione, and testosterone (LC-MS/MS for all; Quest Nichols Diagnostic Laboratory, San Juan Capistrano, CA). The neonate had an elevated serum 17-OHP of 11,500 ng/dL (normal range for term newborns within 12 h of birth: ≤460 ng/dL), androstenedione 4917 ng/dL (normal range not established for newborns; normal range for infants: 6–78 ng/dL), and testosterone 279 ng/dL (normal range of females 1–10 days old: ≤24 ng/dL).
Discussion
In pregnancies complicated by CAH, the delicate synchrony of fetal development is affected by early disruptions in the hormonal homeostasis of the intrauterine environment. Our main finding was this fetus with classical CAH due to 21-hydroxylase deficiency showed an immature brain compared to age-matched, typically developing fetuses, as observed by both ultrasound and 2D-MRI measurements. This finding holds relevance for patients with CAH, who have been shown to have altered neurodevelopment and adverse behavioral, cognitive, and psychological outcomes in adolescence and adulthood.3,9-15 Altered fetal brain development is known to influence the trajectory of brain development across the lifespan. Our findings raise the possibility that fetal brain immaturity could underlie neurodevelopmental differences observed in older youth and young adults with CAH such as leukariosis and temporal brain atrophy.3,9,16-21 However, large-scale, longitudinal studies are required to ascertain the link between our early findings and the various neurodevelopmental differences that have been reported in CAH.
The fetus had multiple prenatal clinical assessments that were interdisciplinary, affording the opportunity to identify a high-risk pregnancy and provide comprehensive and collaborative care during the prenatal period, beyond what would otherwise be limited to postnatal newborn screening (mandated in the United States). We have, for the first time, identified early brain abnormalities in a fetus diagnosed with CAH by leveraging recent advancements in acquisition and analysis of prenatal MRI and ultrasound. As these cutting-edge technologies continue to be utilized, prenatal imaging could enhance our understanding of the role of fetal programming in this screenable disorder, and in turn inform treatment of longer-term outcomes in these patients. We recognize that the prenatal diagnosis of CAH remains challenging in terms of large-scale investigations. However, newborn screening for CAH is universally mandated across the United States, providing a unique window of opportunity to study the etiology of neurological changes within this population beginning in the neonatal period. Longitudinal neurocognitive assessments, from infancy to early adolescence, will allow us to understand the neurocognitive correlates of the early brain changes observed in these children.
Thus far, most of the published literature on neurocognitive effects of CAH has been reported in late childhood, adolescence, or adulthood. These studies have established that these youth and adults with CAH have key brain morphometric differences compared with unaffected individuals as noted by MRI. However, brain development and outcomes in these age ranges are confounded by multiple factors such as varying approaches to postnatal medication dosage for hormone replacement therapy. 9 These findings regarding brain deficits in a fetus with CAH merit further study to understand if these delays in brain development persist into the neonatal period or are attenuated by hormone replacement therapy. Early life studies, particularly in neonates, will help to disentangle the effects of these confounders and better understand the direct effects of CAH and postnatal hormone replacement therapy on brain development. Establishing early markers of altered brain development would provide a roadmap for early identification and treatment of children with CAH who are at highest risk for adverse neurocognitive outcomes across the lifespan.
In summary, our report provides novel insight into early neurostructural differences in the fetus with an altered intrauterine hormonal environment due to CAH, as compared with healthy fetuses. These findings suggest that the in utero hormonal abnormalities inherent to this condition, involving cortisol deficiency and/or excessive androgen exposure, may affect fetal programming and contribute to key neurostructural differences and neurocognitive comorbidities in youth and adults with CAH, as seen in other conditions. Further study is merited of neonatal brain changes in CAH to better identify and treat affected patients and thereby improve long-term patient outcomes.
Footnotes
Acknowledgements
The authors gratefully thank our family for their participation in the study. The authors acknowledge Irene Klecha, RN, for her coordinating assistance in the CHLA FMC, the Children’s Hospital Los Angeles Human MRI core, and Dorit Kliemann, PhD, for her review of the manuscript.
Authors’ Contributions
V.R., L.N.O., W.S.K., and M.S.K. take responsibility for the integrity and accuracy of the data and drafted the manuscript. V.R., L.N.O., and W.S.K. performed imaging and statistical analyses. All authors were involved with the critical revision of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.E.G. receives research support from Novo Nordisk; consultant fees from Adrenas Therapeutics, Daiichi Sankyo, Ferring, Millendo Therapeutics, Neurocrine Biosciences, Novo Nordisk, Nutritional Growth Solutions, Pfizer, and Tolma; and royalties from McGraw-Hill and UpToDate; and serves on data safety monitoring boards for Ascendis, Millendo, and Tolmar. M.S.K. receives research support from Neurocrine Biosciences, Spruce Biosciences, and Adrenas Therapeutics. The other authors have no competing interests to declare.
Ethics Approval
Ethical approval to report this case was obtained from Children’s Hospital Los Angeles Institutional Review Board (CCI-12-00020).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank the Saban Research Institute at CHLA for funding this project via the Imaging Core Pilot Award (to M.S.K., V.R., J.L.W.). The authors thank the Abell Foundation (to M.E.G.) and CARES Foundation (to M.E.G. and M.S.K.) for their ongoing support of the CHLA Comprehensive Care Center. The preparation of the manuscript was supported by grant awards K23HD084735 and R03HD101718-01 (NIH/NICHD) to M.S.K., and K23HD099309 (NIH/NICHD) to J.L.W., K01HL153942 (NIH/NHLBI) and the Saban Research Institute to V.R.
Informed Consent
Written informed consent was obtained from a legally authorized representative for anonymized patient information to be published in this article.
