Abstract
A healthy 11-year-old girl presents with epigastric abdominal pain, fever, weight loss, and decreased appetite for 1 month. On physical examination, she appears ill, dehydrated, and cachectic. Her abdominal examination is significant for large ascites with a fluid wave and is nontender to palpation. Her labs show leukocytosis with an eosinophilic-predominant granulocytosis and an absolute eosinophil count of 6800/mm3. She has elevated serum inflammatory markers, hypoalbuminemia, and lipase is 5000 U/L. Magnetic resonance cholangiopancreatography (MRCP) shows an irregular and dilated pancreatic duct, so she had an endoscopic retrograde cholangiopancreatography with pancreatic stent placement, paracentesis, and colonoscopy. Her peritoneal fluid was significant for an eosinophilic-predominant granulocytosis with no evidence of malignancy on flow cytometry. All other studies and cultures did not reveal an etiology. She initially showed improvement, 18 days later she developed a fever, night sweats, tachycardia, and abdominal distention. Empiric antibiotics were initiated due to concern for infected pancreatic necrosis versus spontaneous bacterial peritonitis. Repeat MRCP showed interval development of 2 peripancreatic fluid collections and re-accumulation of ascites. She continued to have daily fever ranging from 39°C to 40°C. Repeat paracentesis and evaluation of her peritoneal fluid showed resolution of eosinophilia with an elevated neutrophil count, negative Gram stain, and no growth on culture. She completed a 10-day course of antibiotics, however, remained febrile with elevated inflammatory markers and leukocytosis throughout her hospitalization. A genetic panel to evaluate for a hereditary cause of chronic pancreatitis was sent and returned positive for a mutation of the serine protease inhibitor Kazal type 1.
Case Description
A previously healthy 11-year-old girl presents with a 4-week history of postprandial abdominal pain. Outside hospital labs were significant for leukocytosis with predominant eosinophilia, hypoalbuminemia, and elevated lipase. Computed tomographic scan revealed large ascites and a mildly dilated pancreatic duct. Ultrasound showed neither gallstones nor dilated bile ducts. Due to her history of epigastric abdominal pain with elevated lipase level, she was diagnosed with acute pancreatitis complicated by ascites and was subsequently transferred to our institution for further evaluation and management.
Upon arrival, her physical examination was significant for an ill, dehydrated, and cachectic-appearing prepubescent white girl. She was found to have abdominal distention with fluid wave. She was nontender to palpation without peritoneal signs. Her history was significant for associated fever and decreased appetite with 10 lb weight loss since the onset of her abdominal pain. She had no significant past medial history with no known medication ingestion prior to presentation and no history of trauma. She had no known family history of GI diseases, malignancy, or pancreatitis. She had no history of alcohol use, smoking, or illicit drug use.
Her baseline labs at time of admission confirmed leukocytosis with white count 16 300/mm3 with 40% eosinophils and absolute eosinophil count of 6520/mm3. Her lipase was elevated at 862 U/L and albumin was low at 2.4 g/dL. All other labs were grossly within normal limits on admission.
Problem List
Our patient subsequently underwent a workup for eosinophilic-predominant pancreatitis of unknown etiology complicated by large ascites.
What Are Possible Underlying Causes for Pancreatitis With Associated Finding of Peripheral Eosinophilia?
In evaluation of this case, our team considered common causes of pancreatitis including viral infection, pancreatic duct obstruction or abnormality, hyperlipidemia, gallstones or biliary obstruction, hyperparathyroidism. Due to the associated finding of eosinophilia, we also discussed the possibility of invasive parasitic infection, malignancy, autoimmune pancreatitis, allergic reaction, eosinophilic gastroenteritis with associated pancreatitis, and drug-induced pancreatitis.
What Investigation Should Be Performed?
In addition to leukocytosis with eosinophilia, our patient’s initial evaluation was notable for lymphopenia with 1% lymphocytes with an absolute lymphocyte count of 163. Peripheral smear confirmed eosinophilia with no dysmorphology noted. She had mildly elevated inflammatory markers, erythrocyte sedimentation rate (ESR) 25 mm/h and C-reactive protein (CRP) 2.443 mg/dL. Her electrolyte panel was within normal limits, and liver function panel was notable only for hypoalbuminemia with albumin 2.4 g/dL. Liver enzymes and gamma-glutamyl transferase (GGT) were not elevated. Triglyceride level and cholesterol panel were performed and were within normal limits. Initial lipase level was 862 U/L and was trended throughout her hospital course. She had a respiratory pathogens panel performed to evaluate for viral illnesses, which was negative. Her urinalysis was unremarkable. She had blood and urine cultures performed that were finalized without growth.
Due to the finding of significant eosinophilia and unclear cause of pancreatitis, additional workup was performed to evaluate for invasive parasitic infection, autoimmune pancreatitis, and allergic pancreatitis. Due to a history of close exposure to cats and dogs, she was evaluated for invasive parasitic infection. Toxocara IgG antibody performed and was negative. Her stool was tested for ova and parasites with no significant findings. She also had a gastrointestinal (GI) panel that was negative and stool culture with no significant growth. Autoimmune pancreatitis was also considered, and her total IgG level was within normal limits (613 g/L) and antinuclear antibody (ANA) was normal (<1:40). She also had IgG subtype levels evaluated, all were within normal limits including IgG4. IgE levels were drawn to evaluate for allergic pancreatitis and were normal (6.77 g/L). She had a sweat chloride test to evaluate for cystic fibrosis, which was normal.
Due to the presence of large ascites at the time of her presentation, our patient underwent paracentesis with analysis of her abdominal fluid. Her fluid analysis was consistent with transudate secondary to pancreatitis. Her initial cell count contained 29 026 RBCs (red blood cells) and 3747 WBCs (white blood cells), with eosinophil as the predominant white cell type at 80%. Fluid total protein was <3.0 (g/dL), glucose 96, pH 7.98, and amylase level was 7952 (U/L). Her abdominal fluid was sent for culture with Gram stain, viral culture, and acid-fast bacterial culture. No organisms were visualized and all cultures were finalized without growth.
Magnetic resonance cholangiopancreatography (MRCP) was performed abnormality and was significant for an irregular, dilated pancreatic duct with concern for pancreatic duct obstruction. It also revealed no obstructive lesions including choledocholithiasis, mass effect, annular pancreas or pancreatic divisum. The parenchyma appeared consistent with acute pancreatitis without evidence of necrosis or peripancreatic fluid collections.
She also underwent colonoscopy and upper endoscopy and had biopsies of her esophagus, stomach, duodenum, terminal ileum, ascending, transverse, and rectosigmoid colon collected to evaluate for concern for associated inflammatory bowel disease. She had no abnormalities visualized during her endoscopies and her biopsies revealed no specific abnormalities. Her stomach biopsy was notably negative for Helicobacter pylori.
How Do We Manage Our Patient’s Pancreatitis With Associated Pancreatic Duct Obstruction?
On admission, our patient was made NPO and received aggressive IV fluid resuscitation. She underwent endoscopic retrograde cholangiopancreatography (ERCP), which was notable for a prominent major papilla (Figure 1) and an irregular, dilated, tortuous pancreatic duct. There was also noted extravasation of contrast from the tail of the pancreas, consistent with a pancreatic duct leak. A stent was subsequently placed into the main pancreatic duct.
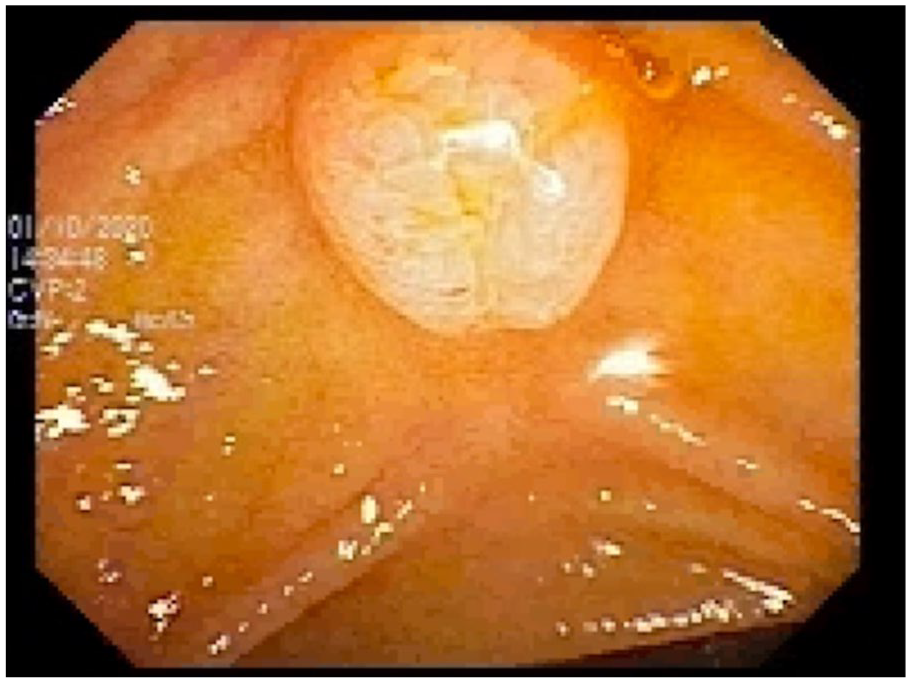
Prominent major papilla visualized on ERCP.
Following ERCP, her diet was advanced to a clear liquid diet. She was started on a proton pump inhibitor, and her lipase levels were monitored. Due to poor tolerance of a regular diet with worsened abdominal pain with up-trending lipase, she had an nasal duodenum tube placed and received continuous feeds. On hospital day 13, she developed a fever to 39.1°C. Infectious diseases were consulted to assist with antimicrobial management and broad-spectrum antibiotics were initiated due to concern for peritonitis. She tolerated a regular diet and completed a 7-day course of antibiotics prior to discharge with follow-up with pediatric gastroenterology and planned to evaluate for hereditary pancreatitis with genetic panel at her outpatient visit.
And the Case Continues . . . Antibiotics or No Antibiotics? That Is the Question
Following her discharge, our patient developed persistent fever with associated night sweats at home with worsening abdominal pain and increased abdominal distention and ascites and was subsequently readmitted. Examination at time of admission was significant for mild abdominal distention with epigastric tenderness. Initial labs showed leukocytosis with white count (16 000/mm3), ESR (120 mm/h), and CRP (22.427 mg/dL), all elevated compared with time of discharge. Due to her clinical worsening, high fevers, and concern for infected necrotizing pancreatitis vs peritonitis, she was started on empiric ampicillin, gentamicin, and cefoxitin. She had a repeat MRCP on the day following admission that was significant for patent pancreatic stent with 2 new, small peripancreatic fluid collections at the lesser sac and along the tail of the pancreas, suggestive of possible pseudocyst formation secondary to pancreatic leakage (Figure 2). There was no free air and no evidence of pancreatic necrosis.

Interval development of 2 peripancreatic fluid collections, one located at the splenic hilum and at the left pararenal space extending toward the pancreatic tail.
Following MRCP, our patient had ongoing high, daily fevers ranging from 39°C to 40.4°C. Due to re-accumulated ascites and concern for bacterial peritonitis, a repeat paracentesis was performed. Peritoneal fluid was significant for elevated polymorphonuclear (PMN) count of 3420/mm3, suggestive of bacterial peritonitis; however, Gram stain showed no organisms. Culture of fluid was finalized with no growth. She received a 10-day course of antibiotics, which were discontinued due to no growth from fluid cultures, despite continued fevers. Following discontinuation of antibiotics, our patient began to improve greatly with decreased fever curve and remained afebrile for 36 hours following discontinuation of antibiotics. Due to her improved clinical status, down-trending inflammatory markers and white count, she was discharged with follow-up with GI.
On follow-up with GI, she was noted to have been doing well since her hospital discharge. She reported having no abdominal pain, improved appetite with adherence to a low-fat diet with positive weight gain. She had remained afebrile since her discharge. During her follow-up appointment, she had a genetic chronic pancreatitis panel sent to evaluate for a hereditary cause of pancreatitis. Her result revealed a homozygous mutation identified in serine protease inhibitor Kazal type 1 (SPINK1), N34S.
Final Diagnosis
Chronic pancreatitis complicated by pancreatic pseudocyst with associated finding of one homozygous risk allele (N34S) identified in SPINK1.
In Summary
Our patient case highlights some interesting nuances to the evaluation and management of the pediatric patient with chronic pancreatitis. While pancreatitis is known to be an intensely inflammatory disease process due to autodigestion of the pancreas, 1 patients with severe pancreatitis are known to be at risk for significant morbidity and mortality if infected pancreatic necrosis develops. 2 Currently, prophylactic antibiotics are not recommended in pediatric patients with severe pancreatitis without signs of necrosis, although the role of antibiotics in pediatric patients with severe acute pancreatitis remains controversial. 3 Our patient had worsening clinical features with recurrent abdominal pain, poor appetite, elevation in her white count (peak of 22 000/mm3) and CRP (peak of 22.427 mg/L), recurrence of abdominal distention, re-accumulation of ascites, and new peripancreatic fluid collection visualized on repeat MRCP. Given her clinical decline and two possible foci for infection, her peritoneal fluid, and new fluid collections surrounding her pancreas, she was treated with a course of antibiotics. She, however, showed no improvement in her fever curve, lab values, or overall clinical condition until antibiotics were discontinued.
Second, our patient’s case highlights the broad differential diagnosis that is to be considered in the evaluation of a pediatric patient with idiopathic severe pancreatitis. While our initial evaluation did not yield an answer to the cause of our patient’s pancreatitis, she was later found to have a genetic mutation in SPINK1, a trypsin inhibitor, which has shown to cause an increased risk of developing chronic pancreatitis. 1 Most commonly, the SPINK1 gene is inherited in an autosomal recessive pattern. Patients typically are heterozygous and require other genetic and/or environmental factors to cause pancreatitis. This has led others to consider the mutation to be more of a disease modifier. 4 However, our patient had a homozygous mutation, which could contribute to the presentation. Several studies have reported a tendency for earlier onset of symptoms with the homozygous mutation, but due to the decreased incidence, it is not statistically significant. 5
In addition to the SPINK1 gene mutation, there have been several other pathogenic gene variants that have been related to pancreatitis. In a small study of 29 pediatric patients with idiopathic recurrent or chronic pancreatitis, 79% of the studied patients were found to have had at least one genetic mutation. 6 In a review on hereditary pancreatitis by Panchoo et al, several other genes have been identified. The most common gene mutation is in PRSS1, which is inherited in an autosomal dominant fashion. Its 3 main variants, R122H, N29I, and A16V, have been identified in >60% of familial cases of hereditary pancreatitis. Other genes that can be seen with hereditary pancreatitis are the R75Q variant of the CFTR receptor and the CTRC mutation. Both the CTRC and the R75Q mutations are autosomal recessive and are less commonly identified. 4 In the pediatric study, approximately 27% of the patients in this study were found to have the same mutation as our patient, the N34S mutation in SPINK1. 6
While the exact prevalence is unknown, chronic pancreatitis is rare in children. 6 Our patient underwent an extensive evaluation to elucidate the cause of her pancreatitis. In addition, her course was complicated by prolonged fever, which presented our team with a clinical conundrum regarding management, particularly with antimicrobial agents.
Footnotes
Acknowledgements
We thank the parents of the girl described for allowing us to share her details and we also thank Dr Sharma and Dr Arrouk for their expertise and knowledge that helped care for this patient.
Authors’ Note
Prior Presentation of Abstract Statement: Persistent Eosinophilia and Fever in Pancreatitis: A Clinical Conundrum was a published abstract in the 2021 Southern Medical Research Conference, Journal of Investigative Medicine, February of 2021.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
