Abstract
Patients with acquired immunodeficiency syndrome (AIDS) are at an increased susceptibility to pathogens and associated malignancies which can present with a unique constellation of symptoms. In this article, we describe a case of Castleman disease in a patient with AIDS, nonadherent with antiretroviral therapy (ART), who presented with fevers, constant abdominal pain, nausea, and vomiting. After an extensive work up, a lymph node biopsy confirmed a diagnosis of human herpesvirus-8 (HHV-8)-associated multicentric Castleman disease. Patients presenting with AIDS and fever have broad differential diagnoses; therefore, reaching a diagnosis as rare as Castleman disease can be challenging. HHV-8 has a propensity to CD20 positive B cells, which allows rituximab to be an effect treatment.
Introduction
Human herpesvirus-8 (HHV-8) is commonly associated with Kaposi sarcomas (KS), defined as an acquired immunodeficiency syndrome (AIDS) defining illness by the Centers for Disease Control and Prevention (CDC) guidelines. Prior to the introduction of antiretroviral therapy (ART), KS were seen 20 000 times more in patients with AIDS over the general population. 1 KS often presents as multiple cutaneous pigmented lesions on the limbs, trunk, and face. Mucosal lesions are noted as well in 20% of patients and visceral involvement in 15% of patients. As CD4+ reduces, the risk of KS increases.
HHV-8’s ability to infect B cells is also relevant in its association to HHV-8-associated Castleman disease (CD). Multicentric Castleman disease (MCD) is a lymphoproliferative disease that can affect one or more large lymph nodes in multiple regions of the body at the same time. About 6500 to 7700 new cases of CD are diagnosed each year in the United States, 75% of them being associated with HHV-8. Furthermore, all patients with human immunodeficiency virus (HIV) and MCD were found to have HHV-8 association. 2 Clinical presentation includes fever in 100% of cases, lymphadenopathy in 96% of cases, splenomegaly in 86% of cases, hepatomegaly in 63% of cases, pulmonary involvement in 35% of cases, edema in 29% of cases, and ascites in 6% of cases. 3
In HIV patients, the incidence of CD has increased with the introduction of ART, while KS also associated with HHV-8 has decreased. It is thought that the immune dysregulation caused by HIV may play a greater influence in CD than the immunosuppression caused by HIV. 4 The incidence of unicentric CD (UCD) is 16 per million person years occurring at every age. 5 HHV-8 multicentric prevalence is estimated at 30 000 to 100 000. 6 Idiopathic MCD incidence is estimated at 5 per million person years. 5
Case Presentation
A 28-year-old Latinx male with known HIV, diagnosed 6 years prior and nonadherent with his ART for over 2 years presented to the emergency department with persistent fevers, bilateral flank pain, nausea, and vomiting for 5 days. Social history revealed a male who has sex with male both insertive and receptive and significant alcohol abuse. He denied any travel outside of the United States or exposure to any animals.
Upon further questioning, the patient revealed a similar constellation of symptoms of fever and abdominal pain 3 years ago, was found to have pancytopenia and generalized lymphadenopathy, and admitted to an outside facility. At that time, a left axillary lymph node biopsy was performed which revealed benign lymphoid hyperplasia with no malignancy and a right inguinal lymph node excisional biopsy that revealed fibroadipose tissue with focal lymphoid hyperplasia. Flow cytometry of the samples showed many lymphoid cells but no evidence of monoclonality in B cell population. A bone marrow biopsy showed slightly hypocellular marrow with no evidence of lymphoma or myelodysplastic syndrome. His symptoms improved after he became adherent to his ART.
Upon this presentation, vital signs were significant for a temperature of 38.8°C, blood pressure of 161/96 mm Hg, heart rate of 145 beats per minute, and respiratory rate of 22 breaths per minute. Examination showed hepatosplenomegaly, and nontender axillary and inguinal lymphadenopathy.
Laboratory work up revealed pancytopenia, high HIV viral load, elevated sedimentation rate, positive cytomegalovirus (CMV) immunoglobulin G (IgG) and ParvoB19 IgG, and negative CMV and Parvo B19 DNA polymerase chain reaction (Table 1). Patient was empirically started on broad spectrum antibiotics but continued to have fevers. A decision to discontinue antibiotics was made when all cultures continued to remain negative. Imagings were significant for bilateral diffuse axillary and inguinal lymphadenopathy and hepatosplenomegaly (Figures 1-4).
Labs and Cultures.

Abbreviations: Ab, antibody; Ag, antigen; CMV, cytomegalovirus; CSF, cerebrospinal fluid; GMS, Grocott methenamine silver; HHV-8, human herpesvirus-8; HIV, human immunodeficiency virus; HSV, herpes simplex virus; IgG, immunoglobulin G; IgM, immunoglobulin M; PCR, polymerase chain reaction; rRNA, ribosomal RNA; WBC, white blood cell.

Chest X-ray PA upright showing bilateral hilar lymphadenopathy.

CT chest coronal plane showing bilateral axillary, mediastinal and hilar adenopathy.

CT abdomen/pelvis coronal plane showing diffuse adenopathy in the region of the hilum of spleen, retroperitoneum, and iliac chain. Also showing splenomegaly (17 x 17 cm) and hepatomegaly (20 cm in the cephalocaudal span).
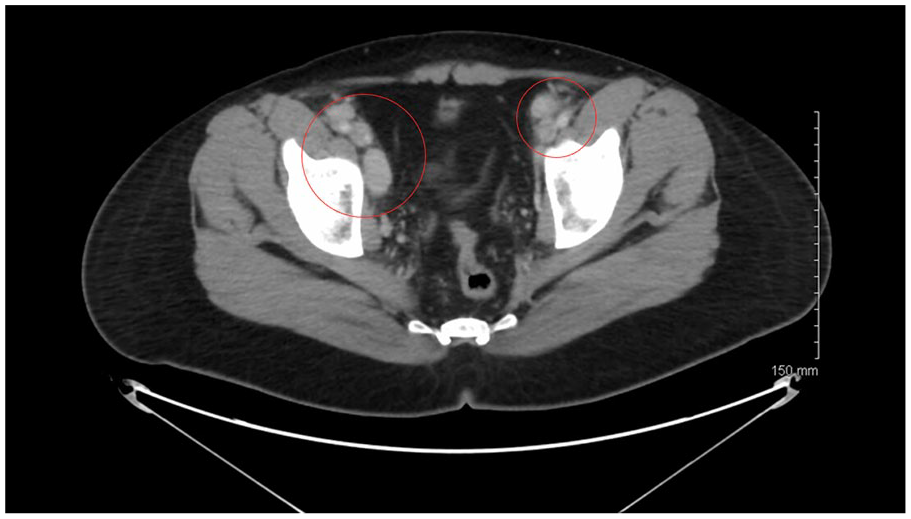
CT abdomen/pelvis axial plane showing adenopathy in the bilateral inguinal region.
Differential diagnosis at this time remained very broad from infectious etiologies including viral, fungal, mycobacterial, or parasitic on one side, and AIDS-associated malignancies such as lymphoma on the other side.
His CD4 cell count came back as 27 cells/µL (6%) and HIV viral load of 98 200 copies/mL. HIV genotype results showed no transmissible resistance, so he was restarted on his previous ART regimen with Bictegravir/Emtricitabine/Tenofovir AF (Biktarvy).
Bone marrow biopsy and aspirate showed normocellular marrow with megakaryocytic hyperplasia, decreased iron, no evidence of acid-fast or fungal organisms, pancytopenia, and no evidence of parvovirus or malignancy. CT-guided right inguinal lymph node core biopsy resulted in scant fragments of lymphoid and fibrous tissue and was nondiagnostic.
His hospital course was complicated with clostridioides difficile colitis with diarrhea, which resolved with oral vancomycin therapy.
After a nonrevealing exhausting work up, the decision was made to perform a right inguinal excisional biopsy. Histopathology showed interfollicular plasma cells and germinal centers with vascularization and “onion-skin” hyaline vascular appearance. HHV-8 immunostaining showed presence of HHV-8 (Figures 5–7). Serum revealed an elevated level of HHV-8 of 2 288 277 copies/mL (nmL <1000). Therefore, a diagnosis of HHV-8-associated MCD was reached.

H&E stain of right inguinal lymph node at 4× magnification showing reactive plasma cell populations in the lymph node consistent with plasma cell variant of Castleman disease.

H&E stain of right inguinal lymph node at 20× magnification showing reactive plasma cell populations in the lymph node consistent with plasma cell variant of Castleman disease.

HHV-8 immunostaining of right inguinal lymph node at 20× magnification showing focally positive HHV-8 staining consistent with Castleman disease.
The patient was started on 6 cycle of rituximab chemotherapy and transitioned to outpatient therapy. He improved clinically. Positron emission tomography (PET) scan showed decreased splenomegaly from 17 to 15 cm in axial dimension and diminished his diffuse lymphadenopathy. He also responded to ART and his HIV viral load dropped to 40 copies/mL.
Three months later, he became nonadherent to his ART due to heavy alcohol abuse. He was readmitted due to jaundice and fever and found to have elevated bilirubin and transaminases with negative work up for biliary obstruction and bacterial cholangitis. Due to fever, recurrence of pancytopenia and high index of suspicious for recurrence of MCD, he was restarted on another 8 cycles of weekly rituximab followed by doxorubicin liposomal. He was also placed back on his previous ART.
Discussion
Castleman disease encompasses a spectrum of lymphoproliferative manifestations defined by histology and presence of human herpes virus-8. Unicentric Castleman disease (UCD) is defined by the presence of a localized mass, most often in the mediastinum but at times extrathoracic in the abdomen or neck. UCD is further divided by histology into hyaline vascular type and plasma cell type. Hyaline vascular UCD presents as a localized mass without constitutional symptoms. On histology, atretic follicular centers are seen with radially penetrating vessels, usually ensheathed by collagenous hyalinization. Plasma cell UCD presents with fever, night sweats, fatigue, weight loss, splenomegaly, anemia, and hypergammaglobulinemia, and a solitary mass. On histology, lymph nodes are characterized by sheets of mature polytypic plasma cells in the interfollicular areas. 7
Multicentric Castleman disease (MCD) is characterized by multiple sites of lymphadenopathy and presents with fever, sweats, fatigue, cachexia, and splenomegaly. Labs usually reveal cytopenia and hypoalbuminemia. MCD is further divided by the presence of HIV and HHV-8. All patients with HIV and MCD have HHV-8. Fewer are HIV negative with HHV-8-associated MCD. In this case, the patient was HIV positive, with HHV-8-associated MCD. 7 HHV-8 viral loads can distinguish between MCD progression and remission. HHV-8 viral loads in progression have been found to vary between 2700 and 400 000 copies/mL with a median of 27 000 copies/mL, while in MCD remission HHV-8 viral load are 0 to 1950 copies/mL with a median of less than 50 copies/mL. 8
HHV-8 is a linear double-stranded DNA virus that gains entry to endothelial cells through glycoprotein-receptor interaction. Entry into the host cell is thought to be through integrins, cystine-glutamate transporter, heparan sulfate, or the tyrosine protein kinase receptor EPHA2. Once inside, the virus uncoats and delivers its genome into the nucleus where it remains latent or undergoes sporadic bouts of lytic reactivation. HHV-8’s ability to infect endothelial cells is consistent with KS clinical presentation of cutaneous lesions. However, it has also been found to infect other cells like B cells, epithelial cells, dendritic cells, monocytes, and fibroblasts. 1
Specific to HHV-8-associated MCD, HHV-8’s ability to infect B cells and plasmablasts is consistent with the marked plasmacytosis in the lymph nodes, and the clinical presentation of B symptoms. On a molecular level, HHV-8’s primary effect is by upregulating the interleukin-6 (IL-6) receptor signaling pathway via viral IL-6 (vIL-6) and LANA-1, promoting lymphoproliferation and angiogenesis. 9 Furthermore, infected cells were found to co-express X-box binding protein-1 (XBP-1), a protein found in maturing B cells, which helps explain why lytic viral proteins are seen without lytic replication being observed. 9 Other lytic phase viral proteins include vMIR-1 which down regulates MHC-I helping to escape apoptosis, vMIP-II and vMIP-III which chemoattracts TH2-type lymphocytes, and replication and transcriptional activator (RTA) which promotes lytic gene expressions. Latent viral proteins include LANA-1 which also inhibits apoptosis and maintain a latent phase, and Kaposins which stabilize cytokine transcripts and activate the STAT3 pathways. 7 On histology, immunohistochemical staining for latency-associated nuclear antigen-1 (LANA-1) would stain positively for CD20 cells often found in the mantle zone of the lymphoid cells in lymph nodes. 10
With its rarity, a gold standard therapy for MCD is still yet to be established. Some options include starting ART, rituximab-based chemotherapy, and IL-6 inhibitors (Tocilizumab, Siltuximab). Rituximab’s efficacy is related to its targeted effect of the monoclonal antibody against CD20 B cells inducing apoptosis via natural killer cells. Using rituximab to deplete CD20 cells has been an effective therapy for MCD. In a clinical study, patients treated with rituximab showed a 94% survival rate at 2 years and 90% survival rate at 5 years compared with the untreated group with a survival rate of 42% in 2 years and 33% at 5 years. 11 This targeted therapy has also demonstrated potential for a protective factor against non-Hodgkin’s lymphoma (NHL) due to its depletion of infected and overactive B cells. 12 Studies have showed increasing success using liposomal doxorubicin which induces regression of lymphadenopathy in MCD and decreases HHV-8 load. 13
Conclusion
The etiology behind fever of unknown origin is difficult and even more challenging in a patient with AIDS. Work up usually is extensive and prolonged, and a multidisciplinary approach is paramount. In cases with constellation of intermittent fevers, hepatosplenomegaly, and diffuse lymphadenopathy, HHV-8 infection and associated diseases should be included in the differential diagnosis.
Before the introduction of ART, Castleman disease had a poor prognosis due to increased risk of developing lymphoma. Today this had improved to a 2-year survival rate more than 90%.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval to report this case was obtained from the Kern Medical Institutional Review Board; approval ID: 21040. Informed consent for patient information to be published in this article was obtained.
Informed Consent
Informed consent was obtained from the patient for his anonymized information to be published in this article.
