Abstract
Mediastinal masses are relatively uncommon. Surgical approach is often needed to diagnose mediastinal masses. Using endobronchial ultrasound transbronchial needle aspiration (EBUS-TBNA) toward approaching a mediastinal mass has yet to be established. Here, we present a case of successful diagnosis of thymic cancer with the use of EBUS-TBNA.
Introduction
Mediastinal tumors are rare. These tumors are classified into anterior, middle, and posterior tumors in association with the compartments of the mediastinum. Imaging studies combined with clinical presentation and attention to age and sex are critical in establishing the presumptive diagnosis. 1
The causes of mediastinal lesions are vast, ranging from congenital to infectious to malignant, warranting extensive evaluation. Multiple modalities for sampling the mediastinal lesions have been described in the literature. 2 Although various techniques exist, numerous critical structures in the mediastinum may present a challenge. 3 In this case report, we preferred to initiate the evaluation of our patient with endobronchial ultrasound transbronchial needle aspiration (EBUS-TBNA) as he was deemed high risk for postsurgical complications. This led to a successful diagnosis of thymic cancer.
Method
Approval was obtained from the Institutional Review Board of Kern Medical. A retrospective review of the patient’s record was performed. A literature search was conducted on PubMed, Google Scholar, and Centers for Disease Control and Prevention (CDC). The following search terms were applied: mediastinal mass, endobronchial ultrasound, transbronchial needle aspiration, and thymic cancer.
Case Presentation
Here, we present a 71-year-old man with chronic kidney disease (CKD), hypertension, hyperlipidemia, diabetes mellitus, prostate adenocarcinoma status postradiation therapy, and 30-pack-year smoking history. Two weeks prior to presentation, he was evaluated for new-onset hoarseness for which flexible nasopharyngoscopy was performed. Left vocal cord paralysis was diagnosed.
Given his extensive past medical history inclusive of prostate cancer, persistent elevated prostate-specific antigen (PSA), and history of tobacco use, presence of unilateral vocal cord paralysis malignancy was included in the differential diagnosis. Due to his CKD, the decision was made to obtain a magnetic resonance imaging (MRI) without contrast of the head, neck, and chest. The MRI revealed a 7.9 × 5.5 × 6.7 cm mass in the left upper mediastinum engulfing the origin of great vessels and displacing the trachea to the right (Figure 1). A subsequent positron emission tomography–computed tomography (PET-CT) scan was then performed which confirmed the high radiotracer uptake of the mass in middle and the anterior mediastinum. The mass was found to completely engulf the great vessels at the level of the aortic arch and the thoracic inlet, with maximum standardized uptake value (SUV) being 6.2 cm. Notable evidence of small bilateral hilar lymphadenopathy with emphysema and without pulmonary infiltrates was also demonstrated (Figures 2 and 3).

MRI T2 chest without contrast demonstrating a 7.9 × 5.5 × 6.7 cm in the left upper mediastinum mass engulfing the origin of great vessels.
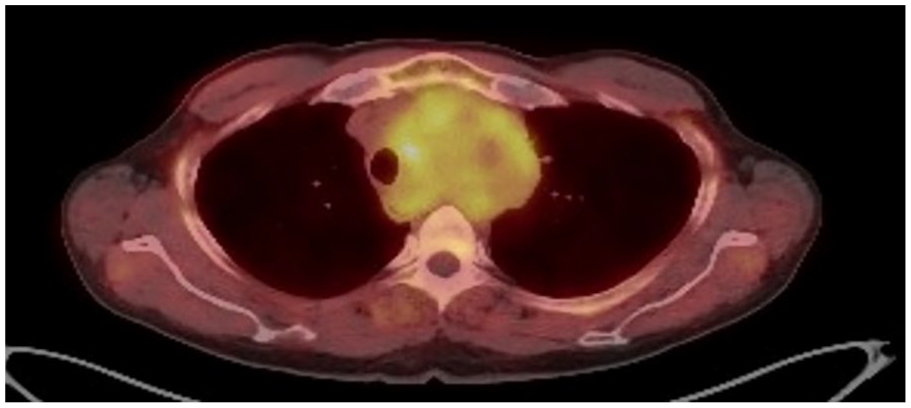
PET scan shows the high radiotracer uptake of the mass in the middle and the anterior mediastinum.

Sagittal and coronal computed tomography of the chest showing the mass completely engulfing the great vessels.
Due to several medical conditions of this patient, encasing the mediastinal great vessels and resection was not feasible. In addition, the patient expressed the use of a less invasive method for diagnosis. Hence, bronchoscopy with EBUS-TBNA technique was performed without complications instead of mediastinoscopy. Histopathology revealed the expression of pancytokeratin and cytokeratin 7 with co-expression of CD5 on the cell’s surface consistent with the diagnosis of epithelial thymic carcinoma (Figures 4 and 5). Subsequently, the patient was discharged home with the plan of initiating chemotherapy as outpatient therapy.

Histopathology is consistent with the diagnosis of epithelial thymic carcinoma.

Histopathology demonstrated the expression of pancytokeratin and cytokeratin 7 with co-expression of CD5 on the cell’s surface consistent with the diagnosis of epithelial thymic carcinoma.
Discussion
Thymic epithelial tumors are heterogeneous in nature and demonstrate variable histological characteristics, including thymoma and thymic carcinoma. For instance, thymomas have been classified by World Health Organization (WHO) into 6 groups using the morphology of the epithelial cells and the ratio between epithelial cells and lymphocytes. Although thymic carcinomas are rare, they are typically aggressive tumors with distant and lymphadenopathic metastatization upon presentation. This is what makes them to have worse prognosis compared with thymomas. The pathological diagnosis can be made by the malignant features and by using variable immunohistochemical and genetic stains. 4
Although various techniques exist for sampling of the mediastinum, numerous critical structures in the mediastinum and tissue sampling may present a challenge. Surgical pathology is often considered to be the gold standard for diagnosing mediastinal mass. Fine needle aspiration or core biopsy either through bronchoscopy or percutaneously guided by imaging is usually the first approach for such tumors. 2 Failure to diagnose using the above methods may warrant proceeding with more invasive procedures like surgical mediastinoscopy that carries a miniscule but higher risk when compared with nonsurgical techniques. For instance, establishing the diagnosis of lymphoma sometimes requires surgical excision of the entire lymph node. 3
The first reported case of sampling a mediastinal tumor via flexible bronchoscopy was published in 1978 by Yang et al. 5 Conventional transbronchial needle aspiration (C-TBNA) has transformed gaining access to the mediastinal and hilar masses for evaluation. This transformation has led to the development of endobronchial ultrasound (EBUS) to overcome the limitations of C-TBNA by increasing the yield and decreasing complications and assuring better patient safety.6-8 This test can be performed under local or general anesthesia, depending on the operator’s and the patient’s preference. The targeted lymph nodes or masses are identified using bronchoscopic visualization and ultrasound imaging. A needle extended from the bronchoscope through the bronchial wall is used to puncture the lesion and aspirate the tissue.9,10 The EBUS-TBNA is superior to conventional TBNA as the needle can be guided using the ultrasound imaging while aspirating the lesions. Since then, EBUS-TBNA has been used widely in the staging of mediastinal tumors. In addition, extensive literature data such as the ASTER trial have supported widely the use of EBUS-TBNA in diagnosing and staging of lung cancers. 11
Comparing tissue sampling using mediastinoscopy versus EBUS-TBNA in evaluating mediastinal lymph nodes among non–small cell carcinoma has shown comparable specificity and positive predictive value in both the techniques, which eventually led the American College of Chest Physicians (ACCP) to recommend EBUS-TBNA as the test of choice for the initial staging of patients with lung cancers.12-14 The approach of EBUS-TBNA is not only minimally invasive when compared with mediastinoscopy but can be performed under local anesthesia which further decreases the risk of any complications. 15 This makes EBUS-TBNA more advantageous for patients who are unable to tolerate invasive procedures and general anesthesia. The diagnostic advantage mentioned above in diagnosing and staging non–small cell carcinoma has not yet been established in other etiologies of mediastinal tumors.
One retrospective study had compared different modalities in approaching mediastinal masses, including EBUS-TBNA, surgery and/or clinical follow up. 16 The EBUS-TBNA was diagnostic in 93.6% of the patients, including malignant and benign diagnosis. In addition, in 67.1% of the patients EBUS-TBNA was also helpful in directing subsequent workup and therapies without complications. 16 A total of 100 benign and 40 malignant mediastinal masses were found.
In conclusion, EBUS-TBNA is effective in providing relevant results for diagnosis of primary tumors and mediastinal staging of tumors along with being a minimally invasive procedure.16,17 It has shown to be the preferred modality in lung cancer with mediastinal involvement and, in some studies, better than mediastinoscopy for the invasive staging of lung cancer. It is more cost-effective than mediastinoscopy and is currently recommended as the first test of choice for invasive mediastinal lymph node staging in lung cancer patients. 18 However, further randomized control studies are needed to evaluate EBUS-TBNA in establishing the diagnosis of various types of mediastinal tumors.
EBUS-TBNA should be considered a useful technique for the diagnosis of mediastinal masses as this technique is safe and highly accurate. This test is also highly appropriate for tumors that are not feasible to resect as claimed by imaging. Continuing efforts to enhance the quality and operative training as well as to make EBUS bronchoscopes more available throughout the institutions will likely improve the yield, decrease complications, and improve overall outcomes. Efforts to improve the quality, overall availability, and operative EBUS-TBNA training should be expanded. In addition, further comparative studies to help providers with decision-making regarding the best approach are also recommended. In our case, we were able to provide accurate diagnosis of thymic carcinoma with a less invasive method, satisfying the patient’s preference and avoiding unnecessary hospital stay or any postoperative complications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Kern Medical Center Institutional Board Review approved this study as minimal risk (Study No 20055).
Informed Consent
Informed consent from the patient for this publication was obtained.
