Abstract
Neonatal lupus is a rare entity, secondary to placental transfer of antibodies from mothers with immune-mediated conditions. While pulmonary involvement is common in lupus, its incidence in neonates is extremely rare, with very few cases being reported in the literature. The authors report a case of a neonate whose mother was diagnosed with systemic lupus erythematosus, with a prenatal diagnosis of third-degree atrioventricular block. While initially admitted in the neonatal intensive care unit with no need for organ support, he presented progressive respiratory failure, initially attributed to sepsis. Favorable clinical progression after pacemaker placement allowed ventilatory weaning, but respiratory failure was again apparent. Chest computer tomography revealed areas of ground-glass lesions, raising the suspicion for lupus pneumonitis. He was started on immunoglobulin and corticosteroids, with clinical improvement. The authors consider this case to be relevant due to the rarity of acute lupus pneumonitis in neonates, alerting that it must be considered a differential diagnosis in neonates with prolonged ventilator dependency.
Introduction
Neonatal lupus (NL) is a rare entity, secondary to placental transfer of antibodies from mothers with immune-mediated conditions, including systemic lupus erythematosus (SLE), Sjögren’s syndrome, antiphospholipid syndrome, and others. 1 The most commonly responsible antibodies are anti-Sjogren’s-syndrome type A/Ro (anti-SSa/Ro), anti-Sjogren’s-syndrome type B/La (anti-SSb/La) and/or anti-ribonuclear protein (anti-U1RNP).1-3 The reported incidence of NL in the presence of positive maternal antibodies is 1% to 5%.4,5
This condition is most commonly diagnosed through skin changes in the neonate’s first weeks of life, but it may also be diagnosed in utero when congenital heart block (CHB) is evident, beginning in the second semester.1,6 CHB is the most severe manifestation of NL, with a mortality rates reaching 20% and pacemaker implantation rates above 60%.4,6 While extracardiac involvement is usually benign and self-limited, complete heart block is considered permanent. 7 Its severity and incidence correlate with higher antibody titers.8,9 Cardiac and dermatological manifestations are the most common, but multisystemic involvement may occur, including cytopenia, liver enzyme elevation, hepatomegaly, splenomegaly, and central nervous system involvement.4,10 Organic involvement appears to be dependent on the type of antibody passing through the placenta. In all, 52/60-kD anti-SSa/Ro and 48-kD anti-SSb/La induce an inflammatory response that leads to scaring of the heart’s conducting system while anti-U1RNP and 50-kD anti-SSb7La have been associated with cutaneous lesions.3,8,9 While pulmonary involvement is common in lupus, 11 its incidence in NL is extremely rare, with very few cases being reported in the literature.12,13 The authors report a case of lupus pneumonitis (LP) in a neonate with NL with CHB and a complex clinical progression.
Case Report
We present the case of a newborn with a prenatal diagnosis of a third-degree atrioventricular block. The mother had been previously diagnosed with SLE, had positive anti-SSa and anti-SSb antibodies and was treated with hydroxychloroquine and prednisolone. At 33 weeks, growth restriction and oligohydramnios were evident. Birth was induced at 34 weeks, through cesarean section. The male newborn presented no need for immediate reanimation, with an Apgar score at 1st, 5th, and 10th minutes of 9,9,9, respectively. The heart rate remained below 100 beats per minute (bpm), but consistently above 60 bpm. The physical examination, including the skin, was normal. He was admitted to the neonatal intensive care unit (NICU) and parenteral feeding was started, with no organ support. Echocardiography revealed a restrictive ventricular septal defect, a wide patent foramen ovale, and a small atrial septal defect (ostium secundum) with left-right shunt.
Neonatal antibody titration at D14 revealed positive antinuclear antibodies (>1/1000), 52-kD anti-SSa/Ro and anti-SSb/La. Beginning at the 11th day of life, he presented progressive respiratory failure requiring oxygen therapy, bronchodilators, and diuretics. Chest computerized tomography (CT) on DOY 36 revealed bilateral areas of disperse ground glass and air bronchogram (Figure 1). Invasive mechanical ventilation was started on D39 due to acute pulmonary oedema. Clinical sepsis was evident, with fever and elevated C-reactive protein (CRP) and he was treated with antibiotics for 14 days. Blood cultures identified methicillin susceptible Staphylococcus aureus and urine culture revealed Escherichia coli. A definitive pacemaker was placed on D50 due to worsening of clinical status. Two days later, fever and high CRP arose. Due to suspected sepsis, he received antibiotics for 1 week, cultural studies were negative. He presented favorable clinical evolution, allowing weaning to non-invasive ventilatory support and high-flow oxygen cannula (HFOC), ending on D64.

Chest computerized tomography with bilateral areas of disperse ground glass and air bronchogram.
Respiratory failure was again evident less than 24 hours after HFOC suspension, requiring invasive ventilatory support. Antibiotics were also restarted, but cultural studies remained negative, as did inflammatory markers. Chest CT was repeated, revealing worsened lesions, with larger areas of ground glass opacities, which supported a diagnostic suspicion of LP (Figure 2). Antibiotics were also restarted, but cultural studies remained negative. He was treated with intravenous immunoglobulin at immunomodulating doses (500 mg/kg for 4 days) while cultures were pending, followed by methylprednisolone pulses (30 mg/kg/day for 3 days) and latter with prednisolone 1 mg/kg/day followed by a weaning scheme of 10% weekly reduction. Favorable clinical evolution was apparent, allowing suspension of ventilatory support after the methylprednisolone pulses and no need for oxygen therapy after 2 weeks.
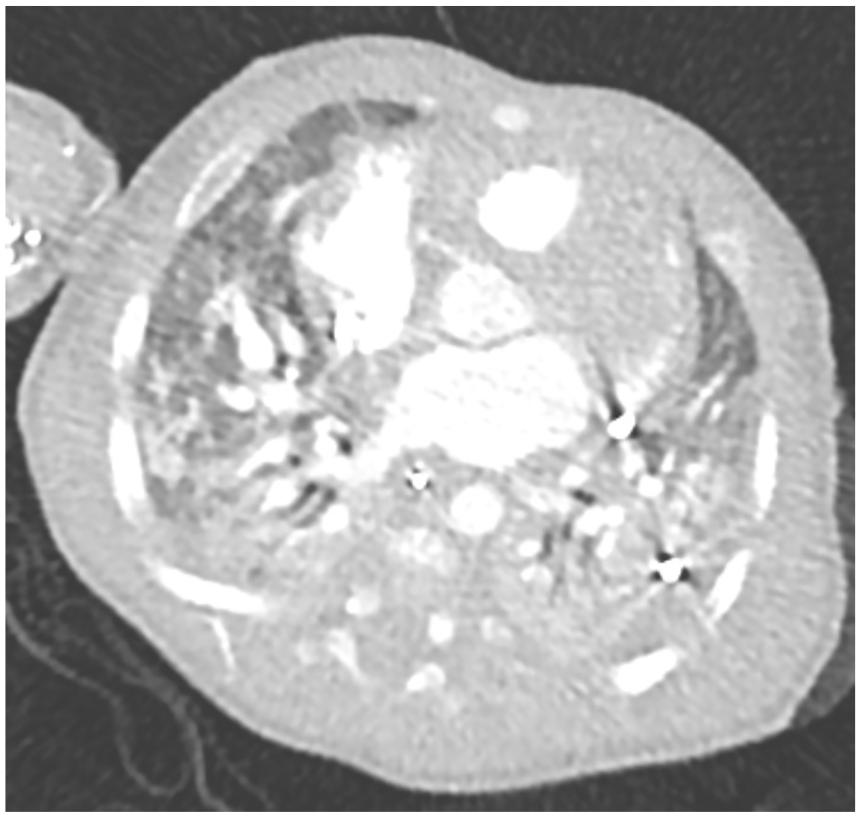
Chest computerized tomography with worsened lesions and larger areas of ground glass.
He was discharged from NICU at D92 and is currently monitored in outpatient multidisciplinary consult, with adequate neurodevelopment, feeding tolerance, and no infectious, cardiac, or respiratory complications. All antibodies were negative. He maintains the definitive pacemaker.
Discussion
With this case report, the authors present an infant with one of the most characteristic manifestations of NL, CHB, who also presented and exceedingly rare pathology in this age group.
Cardiac sequels of NL include CHB, endocardial fibroelastosis and cardiomyopathy.8,14 Our patient presented complete congenital heart block (CCHB) caused by lesion of the AV node with secondary complete atrioventricular dissociation, resulting in significant fetal bradycardia. Isolated CCHB (ie, not associated with congenital heart disease) amounts for 58% to 86% of all cases, and its most common cause (91%) is exposure to maternal autoantibodies, which is the case of this infant. 15 In all, 52-kD anti-SSa/Ro autoantibodies where present in the newborn, as well as anti-SSb/La. According to the literature, these are the most consistently associated with cardiac lesion. 14 CCHB is the most common degree of heart block in NL and more than 2/3 require permanent pacemaker implantation. 7
Pulmonary involvement of lupus is common in adult patients (50%–70%) and it includes pleural disease, parenchymal lung disease, and pulmonary vascular disease such as pulmonary hypertension and diffuse alveolar hemorrhage. 16 It is, however, extremely rare in neonates, with very few cases reported.12,17,18 While interstitial lung disease usually progresses slowly over time, presenting decades after SLE diagnosis, LP, another lupus-related parenchymal disease, is an acute disease that may occur as the initial presentation of SLE. 16 It is one of the rarest pulmonary manifestations in the adult population and case series describe clinical presentation as similar to a respiratory infection, with fever, cough, dyspnea, hypoxemia and pleuritic chest pain, it may also be challenging to distinguish from severe infection and acute respiratory distress. 19
This challenge was apparent in our case, since one of the main difficulties in the diagnostic and treatment approaches in this neonate was the clinical juxtaposition of the possible causes for respiratory failure and ventilation dependency. Our patient presented fever, elevated inflammatory markers, and positive cultural studies, leading to an initial suspicion of infection as the main cause. Favorable clinical progression after pacemaker placement also suggested improved heart function likely to due to a more favorable heart-lung interaction.
The neonate presented worsening ground glass opacities in the chest CT., these are non-specific findings and may also be found in infection or acute pulmonary oedema but are also described in acute LP. 19 Chest radiographs may present unilateral or bilateral infiltrates, also present in this neonate. 16 The diagnosis of pneumonitis was considered more likely, however, due to the rapid improvement after immunoglobulin and methylprednisolone administration and the lack of improvement with antibiotics and diuretics. This is consistent with case reports describing rapid improvement in neonates and children with similar clinical courses after being administered corticosteroids and/or immunomodulators.12,20
The authors consider this case to be relevant due to the rarity of acute LP in neonates, alerting that it must be considered a differential diagnosis in neonates with NL with prolonged ventilator dependency.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
