Abstract
A 38-year-old male with no history of cardiac disease presented with chest pain typical of acute coronary syndrome. He assumed very-low-carbohydrate ketogenic diet (VLC-KD) 4 weeks prior to admission but no other lifestyle change. Workup showed dynamic ST-T changes on electrocardiogram, significantly elevated troponins, ketonuria, and mild rhabdomyolysis. Transthoracic echocardiogram revealed mild inferior wall hypokinesia and cardiac catheterization showed normal coronaries; hence, the diagnosis of type II myocardial infarction (MI) was established. Although the pathogenesis remains unclear, this temporal association between VLC-KD and type 2 MI raised some concerns about VLC-KD’s cardiovascular safety profile.
Introduction
Dietary CHO restriction in VLC-KD mediates significant and quick metabolic changes at the cellular level: glycogen stores become depleted, insulin levels drop, and free fatty acids (FFAs) are mobilized to the liver creating ketone bodies. The intensity of these physiologic and metabolic changes is proportionately related to the severity of CHO dietary restriction. 1 Several physiological outcomes of VLC-KD were reported to be beneficial and cardioprotective; however, recent literature and societal guidelines provided important perspectives about potential cardiovascular implications of VLC-KD. This report represents the first case in literature where VLC-KD had a strong temporal association with Type 2 myocardial infarction (MI) raising some concerns about VLC-KD’s cardiovascular safety profile.
Case Presentation
A 38-year-old Caucasian male with past medical history of obesity (body mass index [BMI] = 33 Kg/m2) and seasonal asthma presented with severe, dull, continuous, retrosternal chest pain radiating to the neck, and upper back associated with shortness of breath and nausea that started 6 hours prior to presentation. No palpitations, lower extremity swelling, dizziness, loss of consciousness, fever, chills, or sweating. No recent respiratory symptoms, recent travel, trauma, overexertion, or contact with sick individuals. He does not use tobacco products, drink alcohol, or use illicit drugs. He exercises regularly (walks 1-2 miles/day). No new over the counter or prescribed medications, and he does not have family history of premature cardiovascular disease (CVD).
Four weeks prior to admission, he initiated a very-low-carbohydrate ketogenic diet (VLC-KD) of 15 grams (g) carbohydrates/day and lost 25 lbs since then. One week prior to the onset of chest pain, he further decreased daily carbohydrate (CHO) intake to 10 g CHO/day. Vital signs were stable, with normal blood pressure and pulse rate, and cardiovascular examination was unremarkable.
Apart from a slightly decreased serum bicarbonate level (21 mmol/L), complete metabolic panel (CMP) was normal [blood urea nitrogen 15 mg/dl, creatinine 1.0 mg/dl, sodium 137 mmol/L, potassium 3.6 mmol/L, chloride 102 mmol/L, calcium 9.0 mg/dl, serum albumin 4.1 G/dl, and serum aspartate aminotransferase/alanine aminotransferase/alkaline phosphatase were 52/30/73 U/L respectively]. Venous blood gas was done and showed pH: 7.42 and pCO2: 34 mmHg. Complete Blood Count, lactic acid, and C-reactive protein (CRP) were normal. Blood alcohol level was undetectable and urine drug screen was negative. Hemoglobin A1c was 5.4% and lipid panel was normal except for low-density lipoprotein cholesterol (LDL-C) of 142 mg/dl. Urinalysis revealed ketonuria (80 mg/dl) and urine specific gravity > 1.050 but no casts or urinary tract infection (UTI). Creatinine kinase was 539 U/L. Q-3-hour Troponin T trend was 720, 745, and 878 ng/L (normal: 0-21 ng/L). Chest computed topography with angiogram was negative for aortic dissection, pulmonary embolism, pneumothorax, or pleural effusion. Transthoracic echocardiogram (TTE) showed inferior wall mildly hypokinetic with reduced wall thickening, otherwise normal systolic and diastolic functions, no pericardial effusion. While in-hospital, he developed another episode of typical chest pain. Electrocardiogram (EKG) at the time showed subtle (1-mm) ST-elevation in anterolateral leads (EKG was normal on admission) (Figure 1). Left cardiac catheterization showed normal coronary arteries.

Dynamic ST-segment changes in electrocardiogram (EKG). (A) normal EKG upon admission. (B) 1- mm ST elevation in anterolateral leads,1 day after admission (blue circles).
The patient was initially treated with aspirin, carvedilol, atorvastatin, morphine, sublingual nitroglycerin, and heparin infusion. Chest pain was continuous for 6 hours prior to admission and resolved completely after the administration of sublingual nitroglycerin in the emergency department. Once coronary atherothrombosis was ruled out (by cardiac catheterization), heparin infusion was then discontinued and the patient was discharged on aspirin and moderate-intensity statin therapy for primary CAD prevention. 2
Discussion
The diagnosis of Type II MI in this patient was based on the Fourth Universal Definition of MI supported by the Joint ESC/ACC/AHA/WHF Task Force: typical chest pain, elevated cardiac troponin levels, dynamic EKG changes, and evidence of wall motion abnormalities in TTE in the absence of coronary atherothrombosis. 3 As shown in Table 1, there are myriad of etiologies that can lead to cardiac troponin elevation; all of which were ruled out in this patient leaving type 2 MI as the lead diagnosis. Dynamic EKG changes raised the suspicion of coronary vasospasm and pericarditis; however, these EKG changes were so subtle to be diagnostic of either and rather support the diagnosis of type 2 NSTEMI. Elevated creatinine Kinase (CK) levels and ketonuria are likely caused by rhabdomyolysis and nutritional ketosis but they are not high enough to attribute to myocardial injury solely. Additionally, normal CRP level put pericarditis further down the differential diagnosis list.
Common Etiologies of Elevated Cardiac Troponins; Classified by Myocardial Ischemia vs. Injury.
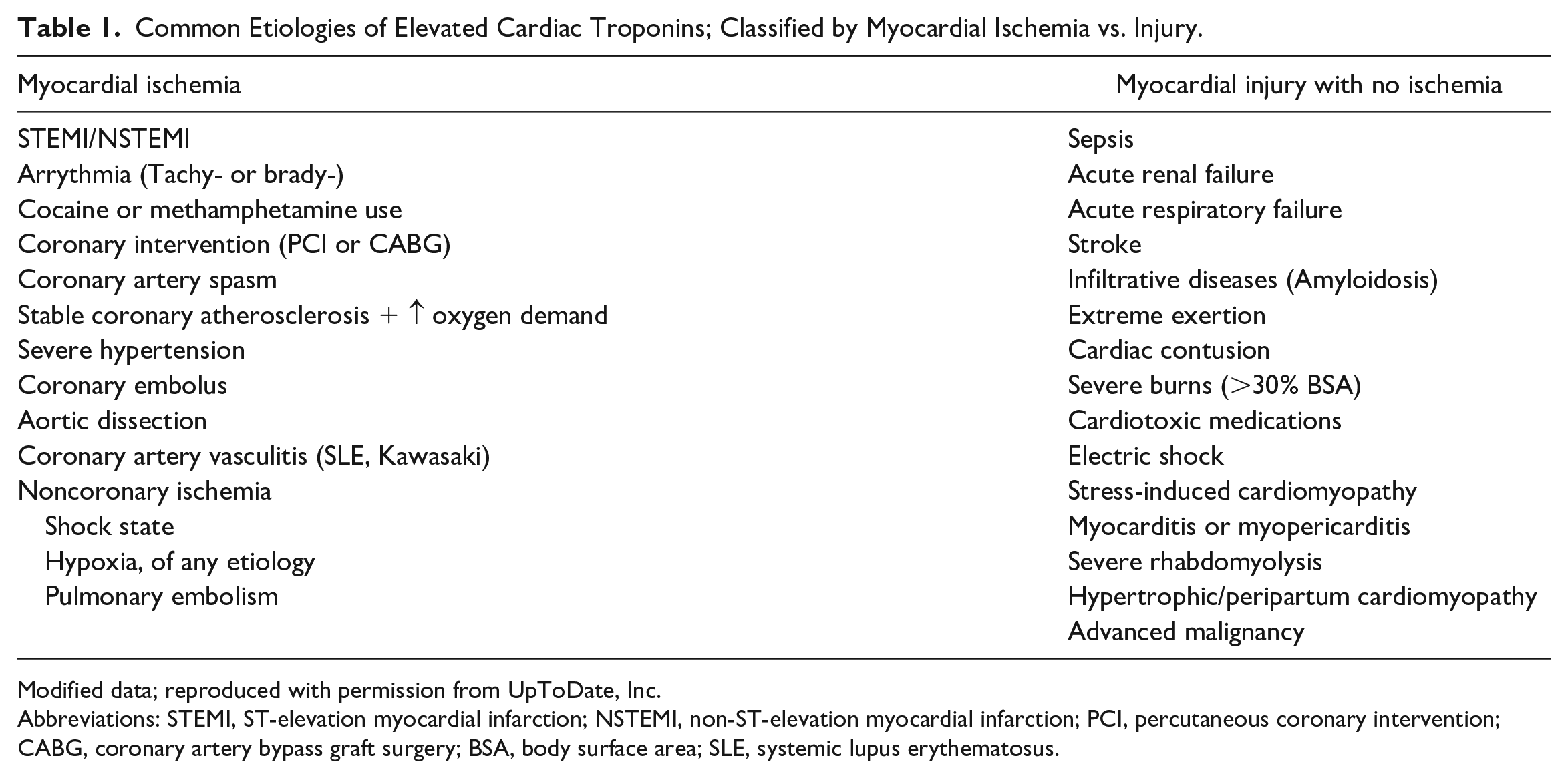
Modified data; reproduced with permission from UpToDate, Inc.
Abbreviations: STEMI, ST-elevation myocardial infarction; NSTEMI, non-ST-elevation myocardial infarction; PCI, percutaneous coronary intervention; CABG, coronary artery bypass graft surgery; BSA, body surface area; SLE, systemic lupus erythematosus.
Dietary macronutrients (fats, CHO, and proteins) were noted to significantly affect CVD risk factors. Evidence-based recommendations advise restricting saturated fatty acids, refined CHO, and animal-sourced proteins. Nutritional and cardiovascular societal guidelines also support replacement of these with unsaturated fats, unprocessed CHO, and plant-sourced proteins. 4 However, a class of therapeutic diets focusing primarily on macronutrients has grown in popularity over the past decades. These diets restrict CHO intake, to varying degrees, liberalize fat intake, and circumvent many of the evidence-based dietary recommendations above. The most extreme of these is the very-low-CHO ketogenic diet (VLC-KD).
Notably, dietary CHO restriction in VLC-KD mediates a significant and quick shift in metabolism within days to weeks. Glycogen stores deplete, insulin levels fall, and free fatty acids (FFA) are mobilized from adipose tissues. As demonstrated in Figure 2, mobilized FFAs then undergo mitochondrial β-oxidation in the liver to form aceto-acetyl-CoA and, eventually, the ketone body β-hydroxybutyrate. When released into the bloodstream, β-hydroxybutyrate acts as the prime source of energy for vital organs, particularly the brain and the heart, where it is turned back into acetyl-Co-A and enters the citric acid cycle. 5 Apparently, the intensity of these physiologic and metabolic changes is proportionately related to the severity of CHO dietary restriction and percentage intake of saturated fatty acids. 6

Summary of metabolic interactions and cardiovascular effects of very low carbohydrate ketogenic diet.
Despite VLC-KD widespread use, its cardiovascular impacts are not fully understood. Doubtless, several physiological outcomes of VLC-KD are beneficial and prevent CVD. It decreases body mass index (BMI) and visceral fat density, lowers triglyceride (TG) levels, and improves blood glucose levels in type 2 diabetics and prediabetics. However, recent literature and societal guidelines raised multiple concerns about VLC-KD and its potential CVD risks. Due to high dietary saturated fatty acids (SFA) and cholesterol intake, VLC-KD increases LDL-C levels. 7 VLC-KD also restricts cardioprotective food elements such as whole grains, beans, starchy vegetables, and most fruits and liberalizes animal protein intake (beef, pork, processed meats, and dairy products), both known to be associated with increased CVD risk. 8 Though elevated in our patient, it is unlikely that high LDL-C could have contributed to his acute cardiac event because (1) obstructive coronary atherosclerosis was ruled out in cardiac catheterization and (2) elevated LDL-C must be present for several years (along with other cardiovascular comorbidities) before leading to MI, which is not the case in here.
Marked nutritional ketosis and muscle degradation related to VLC-KD were evident in our patient. Oxidative stress in VLC-KD caused by nutritional ketosis, antioxidant deficiencies, and increased fat consumption eventually lead to the formation of reactive oxygen species (ROS). These ROS ultimately cause a significant reduction in nitric oxide, at the cellular level, leading to vascular endothelial damage. 9 Moreover, some literature also noted that ROS influx can negatively affect myocardial ion exchange which will also lead to cardiac arrhythmias and erratic cardiac remodeling/hypertrophy. 10 Bank et al. reported Torsade de Pointes in 1 patient and QT prolongation in another, both were associated with VLC-KD. 11 In another case series, Best et al., also reported a QT prolongation associated with VLC-KD. 12
Unfortunately, there are no large-scale studies to dissect the cardiovascular effects of VLC-KD on overall mortality; Seidelmann et al. is the largest epidemiological cohort to date that correlated dietary CHO intake and mortality. This cohort concluded that low CHO dietary patterns favoring animal-derived protein and fat sources (typical of VLC-KD) were associated with higher mortality; a discouraging finding against undertaking a long-term VLC-KD.13,14 Actually, this case is the first in the literature to report a strong temporal association between VLC-KD and type 2 MI. Although the etiopathogenetic mechanisms of type 2 MI in VLC-KD still remain unclear, oxidative stress, nutritional ketosis, and cardiac muscle degradation seem to play a role; however, further studies are needed to explore the VLC-KD cardiovascular safety profile.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
