Abstract
An 89-year-old Caucasian female with a recent diagnosis of endometrial adenocarcinoma status post hysterectomy and acute deep vein thrombosis on anticoagulation presented with hematochezia. Colonoscopy revealed sigmoid colon stricture with the biopsy findings of metastatic endometrial adenocarcinoma based on positive immunohistochemistry staining for cytokeratin 7, paired box gene 8, and estrogen receptor. The oncologist referral was given to the patient for consideration of chemotherapy, but she decided to go with palliative care. Thus far, only 2 similar cases have been published in the literature. Our case exemplifies the potential for an unconventional pattern of metastasis of primary endometrial adenocarcinoma to the colon.
Introduction
Endometrial cancer is the fourth most common cancer in females, and the mortality rate has increased over the past 20 years. 1 Local and distant recurrences continue to be a major concern in high-risk patients.2,3 Well-recognized sites of metastasis are the pelvic lymph nodes, paraaortic lymph nodes, vagina, peritoneum, and lungs, while atypical sites include the intraabdominal organs, bones, brain, abdominal wall, and muscle. 4 In this article, we present a rare case of metastatic endometrial cancer to the sigmoid colon.
Case Presentation
An 89-year-old Caucasian female with a recent history of endometrial cancer and deep vein thrombosis presented to the hospital emergency department with complaint of hematochezia. She underwent total laparoscopic hysterectomy and lymph node dissection 1 month prior to this current presentation. The endometrial cancer was characterized as a high-grade, poorly differentiated adenocarcinoma stage 3b, strongly positive for estrogen receptor with a wild type of p53. Three weeks after the surgery, she was diagnosed with proximal deep vein thrombosis of the right lower extremity and was started on anticoagulation with apixaban. Within a week of beginning the anticoagulation, she developed bleeding per rectum. She was hemodynamically stable at the time of admission with a hemoglobin of 8 g/dL. The physical examination was significant for right lower extremity swelling and bright red stool on rectal examination. Esophagogastroduodenoscopy was unremarkable; however, the colonoscopy revealed severe stricture in the distal sigmoid colon (Figure 1), which was subsequently biopsied. The serum carcinoembryonic antigen (CEA) levels were within the normal range. Colon biopsy demonstrated metastatic endometrial adenocarcinoma, with immunohistochemistry (IHC) staining positive for cytokeratin 7 (CK7), paired box gene 8 (Pax 8), and estrogen receptor (ER; Figure 2). The oncologist referral was given to the patient for consideration of chemotherapy, but she decided to go with palliative care.
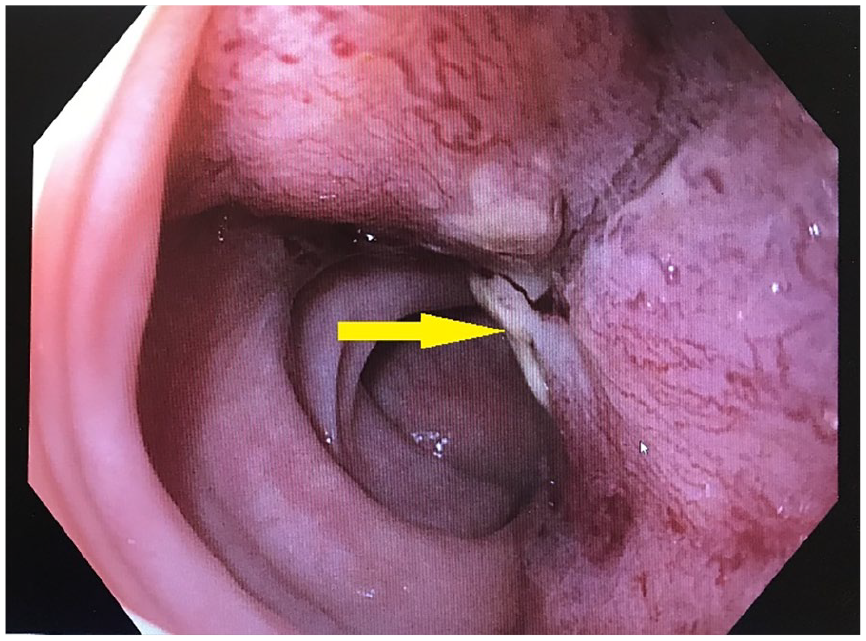
Colonoscopic images demonstrating severe stricture in the distal sigmoid colon (yellow arrow).

(A) Cytokeratin (CK) 7 positive in endometrial tumors (arrow). (B) CK20 positive in colon glands normal below (arrow), negative in tumor (arrow head). (C) Paired box gene (PAX) 8 positive in gynecological primaries (arrow), negative in normal colon (arrow head). (D) Metastatic endometrial carcinoma.
Discussion
Our case exemplifies the potential for an unconventional pattern of metastasis of primary endometrial adenocarcinoma to the colon. Thus far, only 3 similar cases have been published in the literature.2,3,5 Endometrial adenocarcinoma is the most common histologic subtype of endometrial cancer. The metastasis is mainly via lymphatic and hematogenous modes. 6 Hematogenous spread is commonly associated with metastasis to the lungs, brain, and bones. 7
Colon metastasis from endometrial adenocarcinoma is very rare, and it is most commonly due to direct spread and peritoneal seeding. The risk factors for colonic metastasis from endometrial adenocarcinoma include histologic grade 3, myometrial invasion >50%, age >60 years, lymphovascular invasion, and lower uterine involvement. 2 Endometrial and colorectal adenocarcinoma exhibit morphological similarities but have different immunohistochemical profiles. In poorly differentiated carcinomas, IHC helps identify the primary site of malignant tumors. In endometrial adenocarcinoma, IHC stains are positive for CK7 and ER, whereas negative for CK20. On the other hand, colonic adenocarcinoma IHC stains are positive for CK20 and negative for CK7 and ER. 8 Thus, the immunohistochemical phenotype pattern of CK20−/CK7+ favored the diagnosis of endometrial metastasis in our case.
In the case published by Hubers and Soni, a 75-year-old female with a remote history of endometrial cancer was found to have a sigmoid and proximal jejunal mass that was resected by exploratory laparotomy. 2 The tumors in the small bowel and sigmoid colon were histologically confirmed to be metastatic endometrioid adenocarcinoma. She went into remission with chemotherapy. Molnar et al published a case of a 71-year-old female with a past history of endometrial adenocarcinoma status postsurgical resection who presented with anemia and bowel obstruction symptoms 2 years after the surgery. 5 By imaging and endoscopic evaluation, she was found to have a tumor invading the left colon as well as spleen and gastric antrum. She underwent left hemicolectomy, splenectomy, and distal gastrectomy. The histopathology confirmed the diagnosis of metastatic serous endometrial cancer. There have been previous reports of malignant transformation of colonic endometriosis into endometrial cancer, which typically arises from the serous layer of the colon. 9 In our patient, there was no history or pathological findings of endometriosis on the biopsy.
Conclusion
Our case signifies the importance of considering the colon as a metastatic site, opposed to an independent primary malignancy, in the setting of primary endometrial adenocarcinoma. We could not ascertain the pathogenic mechanism of colonic metastasis in our case, especially in the absence of peritoneal carcinosis or endometriosis. Recognizing this rare but possible presentation will allow for appropriate diagnosis and treatment of these patients.
Footnotes
Acknowledgements
We would like to thank Mr Daniel Berry (Texas Tech Health Sciences Center El Paso) for assisting with the literature review for this case presentation.
Author Contributions
All the authors have substantial contributions to the manuscript.
Alexia Jauregui: Conception and drafting the manuscript.
Mahesh Gajendran: Conception, drafting the manuscript, and critical revision of the important intellectual content.
Priyadarshini Loganathan: Critical revision of the important intellectual content.
Angelica Padilla: Critical revision of the important intellectual content and pathology images.
Jesse Qiao: Interpretation of data, critical revision of the important intellectual content, and pathology images.
Sherif Elhanafi: Conception, critical revision of the important intellectual content, and supervision.
All authors approved the final version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for their anonymized information to be published in this article.
