Abstract
We report a rare case of a 32-year-old male who ingested 32.4 to 54 mg of colchicine and presented after 44 hours. He developed progressive multiple organ failure with shock, acute kidney failure, troponemia, pancytopenia, absolute neutropenia, disseminated intravascular coagulation, acute liver failure, rhabdomyolysis, and lactic acidosis. He also developed electrolyte abnormalities and refractory hypoglycemia. Initial treatment consisted of activated charcoal, fluids, and broad-spectrum antibiotics with supportive treatment of mechanical ventilation, hemodialysis, vasopressors, N-acetylcysteine, colony-stimulating factors, and blood products. Literature shows potential benefit of colchicine-specific Fab fragments for acute toxicity with limited studies and is not currently available in the United States. Further research for N-acetylcysteine protocol for acute liver failure in colchicine toxicity and potential use of colchicine-specific Fab fragments is needed. Our case demonstrates the importance of early use of activated charcoal for ingestion overdose with the incorporation of poison control into multidisciplinary team for coordinated patient care.
Background
Colchicine is used for treatment of gout, familial Mediterranean fever, Behcet’s disease, pericarditis, coronary artery disease, and other inflammatory conditions like osteoarthritis.1-4 In order to treat these disease processes, it does have various mechanism of actions. The primary mechanism of action is binding to tubulins blocking the production and lengthening of microtubules, which make up the skeleton of cells. 1 It, also, inhibits neutrophil chemotaxis and fibrosis. 1
Colchicine does have a narrow therapeutic index. Reported half-life of oral colchicine is 4.4 to 16 hours in therapeutic doses and can increase to 11 to 32 hours in patients with toxicity. 5 With any drug with a narrow therapeutic index, toxicity can be due to improper dosing or nonadherence to the dosing instruction, but on the other hand, toxicity can still happen with proper dosing and adherence.
Colchicine has minimal side effects of abdominal pain, nausea, vomiting, and diarrhea. 1 These minimal side effects often improve on its own or is limited in nature. For those with liver or kidney dysfunction, the dose of colchicine needs to be reduced. 1 High fatality rates were documented after ingestions >0.5 mg/kg, but the lowest reported lethal doses of oral colchicine have been 7 to 26 mg. 5 Based on a study done by Hirayama et al, 6 lethal dose of colchicine is considered to be 0.8 mg/kg. It is rapidly absorbed from the gastrointestinal (GI) tract reaching peak serum concentrations at about 0.5 to 3.0 hours. Therefore, the severity of side effects is not necessarily dose proportionate. Colchicine dosing for treatment purpose is closely linked with the timing of GI adverse effects severity which guides the dosing.
Case Presentation
A 32-year-old male with medical history of myocarditis/pericarditis on colchicine, asthma, and poly-substance use (reported prior use of marijuana, opioids, and cocaine) brought himself to the emergency department about 44 hours after ingestion of 54 pills of colchicine 0.6 mg (32.4 mg; 0.39 mg/kg) with severe, diffuse abdominal pain with persistent and frequent nausea, vomiting, and diarrhea. The patient stated that in the past he had taken extra amount of colchicine for chest pain related to his myocarditis, which helped but could not quantify, and he did not have any side effects like he did this time. His ingestion was an accidental overdose to self-medicate for his chest and abdominal pain but not as an intended overdose. A few days into his hospitalization, family reported potential ingestion to be 90 pills (54 mg; 0.65 mg/kg). There was a question of possible co-ingestion of unknown amount of hydroxyzine. Urine toxicology was positive for opiates, suboxone, cocaine, and marijuana. He was afebrile, normotensive, tachycardic, and tachypnea. He was alert and conscious with unremarkable physical examination except for diffuse abdominal pain that was out of proportion to examination on presentation.
Initial laboratory tests as shown in Table 1 showed leukocytosis, acute kidney injury (AKI), acidosis, elevated lactic acid (7.1 mmol/L), liver dysfunction, rhabdomyolysis, elevated lipase, and troponinemia. Chest X-ray and computed tomography abdomen and pelvis were unremarkable. Serum colchicine level, drawn on hospital day 2 (at least 68 hours after ingestion), was noted to be 9.6 ng/mL. He received activated charcoal, multiple boluses of intravenous fluids, broad-spectrum antibiotics of piperacillin/tazobactam and vancomycin, and was admitted to the intensive care unit after poison control was contacted. He was intubated secondary to tachypnea with respiratory rate of 50s and change in mental status on the evening of his first hospital day. He continued to have worsening acidosis requiring bicarbonate drip.
Laboratory Trend During the Course of Hospitalization.

Abbreviations: AST, aspartate aminotransferase; ALT, alanine aminotransferase; BUN, blood urea nitrogen; INR, international normalized ratio; PT, prothrombin time; WBC, white blood cell.
On hospital day 2, he developed progressive multiple organ failure (MOF) and required vasopressors of norepinephrine, vasopressin, and phenylephrine in addition to stress dose steroids for refractory hypotension. From his worsening AKI, he became oliguric requiring hemodialysis. He had progressive pancytopenia and disseminated intravascular coagulation as seen in Table 1. With his absolute neutropenia, he was given granulocyte colony–stimulating factor (filgrastim). He also required multiple units of fresh frozen plasma and platelets. With his acute liver failure, he received N-acetylcysteine based on the 21-hour dosing protocol for acetaminophen toxicity.
With his troponinemia, bedside echocardiography was done and showed cardiovascular compromise with reduced left ventricular function. He also developed many electrolyte abnormalities including refractory hypoglycemia present on admission, hypocalcemia (hospital day 2), hypomagnesemia (hospital day 2), hyperkalemia (hospital day 4), and hyperphosphatemia (hospital day 4). On physical examination, he developed significant anasarca, skin blisters with skin sloughing, blood oozing from oral mucosa, central venous catheters, and peripheral lines, and lower extremity mottling. His acute renal failure and acidosis were refractory to hemodialysis requiring multiple vasopressors. After N-acetylcysteine, his liver enzymes did normalize. However, due to the progressive decline in his clinical status, family requested withdrawal of care, and the patient expired on hospital day 7. Blood cultures were positive for Candida dubliniensis post mortem.
Discussion
Our patient did present with mostly GI complaints related to colchicine side effects since it is metabolized in the GI tract. Colchicine is rapidly absorbed from the GI tract and undergoes first pass metabolism through the liver where the metabolites enter the enterohepatic recirculation before being excreted in the bile and feces.3,7 Renal clearance also accounts for 10% to 20% of excretion and can be increased with hepatic failure.3,7 Therefore, if there is any liver or kidney impairment, colchicine cannot be excreted like it normally does. With our patient having both liver and kidneys impairment, this could have led to his progression of MOF.
Toxicity leading to adverse events is uncommon but well described and is a known risk when prescribing this medication. When taken in high doses, it has been shown to be associated with high mortality rate especially when the ingested dose exceeds 0.5 mg/kg. 5 Due to the unknown amount of ingestion our patient took, it is believed that he ingested anywhere from 0.39 mg/kg to 0.65 mg/kg. However, ranges ingested of colchicine leading to MOF varies. In a controlled study with colchicine mean daily dose of 0.5 mg to 4.8 mg, the adverse events they found were GI events but death, neuropathy, and myotoxicity were not reported. 2 In another study, a patient ingested 90 mg (1.38 mg/kg) colchicine with MOF in which activated charcoal, dialysis, N-acetylcysteine, and granulocyte colony–stimulating factor were started, and patient was discharged home a month later with discontinuation of dialysis. 8 However, of note, one patient recovered after ingestion of 1.38 mg/kg but presented 1 hour after ingestion. 8 Another patient recovered from a critical presentation after taking 15 mg (0.2 mg/kg). 6 Unfortunately, our patient expired secondary to his clinical course and family requesting comfort care.
Our patient did progress through the typical clinical course of colchicine toxicity (Figure 1). He presented in the first stage with severe abdominal pain, nausea, vomiting, diarrhea, and leukocytosis. The first stage is the GI phase, which is typically seen within 0 to 24 hours secondary to mucosal damage.3,5,7,8 He rapidly progressed into the second stage with his MOF of respiratory distress, cardiac failure, renal failure, liver failure, disseminated intravascular coagulation, bone marrow suppression, hemolysis, and metabolic/electrolyte derangement as discussed above. This occurs 1 to 7 days post ingestion, presenting also with cardiac arrhythmias, cardiac arrest, encephalopathy from brain edema, convulsions, myopathy, and neuropathy.3,5,7,8 Although his liver function normalized, his AKI was refractory to dialysis, and with withdrawal of care, he was unable to reach the third stage. This is the recovery phase, which occurs 7 to 21 days post ingestion with resolution of organ system derangements, rebound leukocytosis, and alopecia.3,5,7,8 Most deaths from acute oral colchicine poisoning result from hemodynamic collapse and cardiac arrhythmia instead of bone marrow suppression. 9
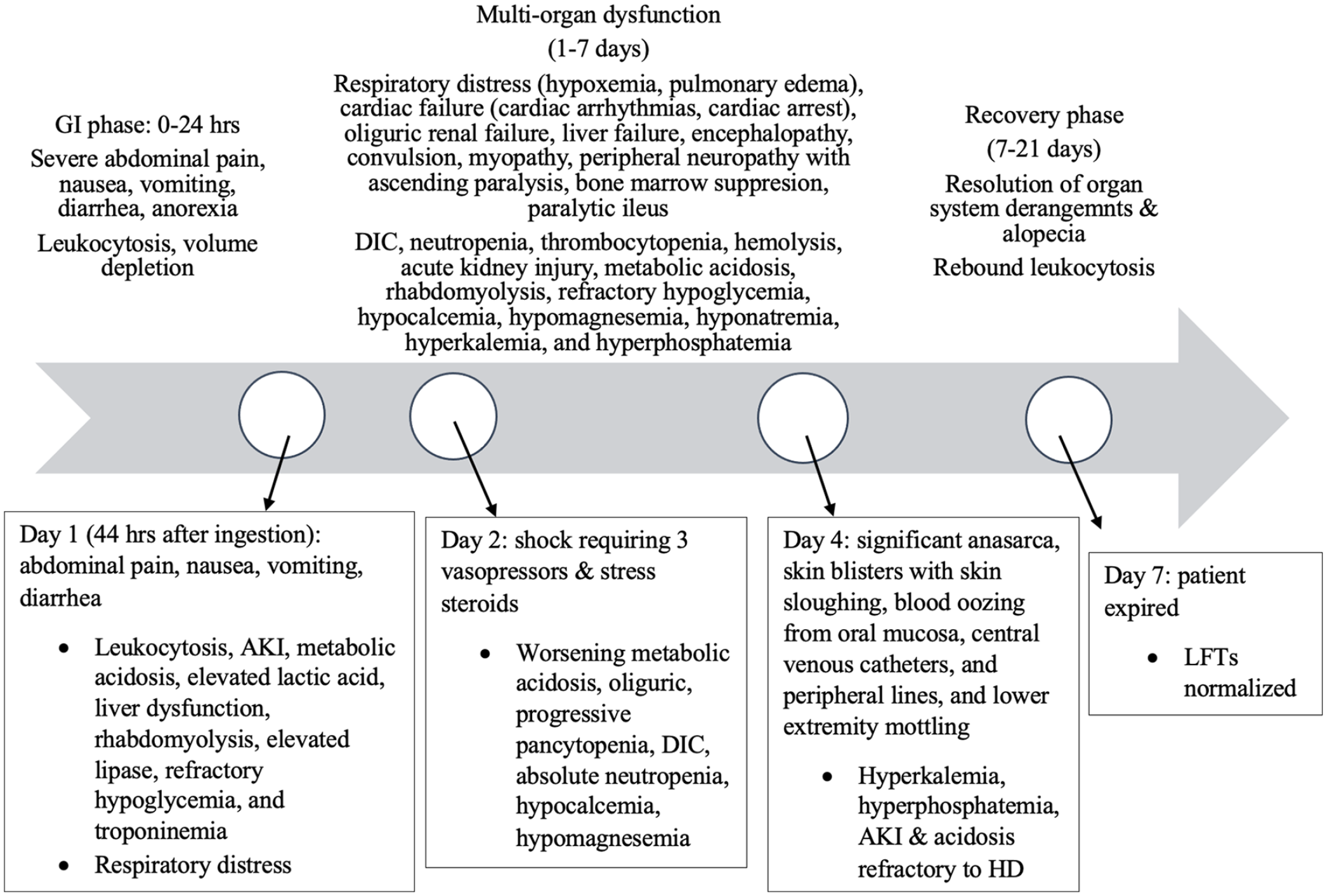
Timeline of colchicine toxicity.
There is currently no antidote or directed therapy available for colchicine overdose. We chose to treat our patient supportively trying to manage his clinical presentation. Management mostly consists of early GI decontamination with gastric lavage and activated charcoal with aggressive supportive care, which becomes difficult if MOF develops.3,5 Activated charcoal use in toxicity cases is often controversial. Poison control in our instance did recommend the use of multidose activated charcoal as long as the patient was able to protect his airway. Since it was a late presentation, gastric lavage would not be beneficial as it is best done within an hour of ingestion. Even within 24 hours after ingestion, large amounts of colchicine can remain in the stomach. 5 Since side effects of colchicine is closely linked to the GI system, the use of activated charcoal, which works in the GI system, can be beneficial.
There is no protocol in place for the use of N-acetylcysteine for colchicine-induced liver failure, but its use was described in prior case reports for non-Tylenol-induced acute liver failure.8,10 N-acetylcysteine is an antioxidant and free radical scavenging agent that increases intracellular glutathione. Glutathione is a major component of the pathways cells use as protection from oxidative stress. 11 There is a case where the patient made a full recovery from 1.39 mg/kg ingestion of colchicine with N-acetylcysteine based on their protocol for acetaminophen overdose. 8 For our patient, we decided to use the 21-hour dosing protocol for acetaminophen toxicity. Forty-eight hours after N-acetylcysteine administration, there was normalization of his liver enzymes, as seen in Table 1.
Although toxicity of colchicine is rare secondary to controlled doses for medical management, due to the quick progression of fatal events occurring with varied colchicine doses, treatment modalities are needed to counteract colchicine. For instance, colchicine-specific Fab fragments have been used for treatment of colchicine toxicity. Colchicine-specific active immunization in rabbits and passive immunization in mice with goat colchicine-specific antibodies and their Fab fragments were shown to have success in preventing or reversing acute colchicine poisoning by reactivating tubulin. 9 In a woman who presented 24 hours after ingesting 60 mg of colchicine, they administered colchicine-specific Fab fragments from the antiserum of goats immunized with a conjugate of colchicine and serum albumin, and serum colchicine was undetectable 8 days after leading to full recovery. 9 Another case of a recovery was seen with anti-colchicine Fab fragments in a porcine model. 4 There is still limited data showing efficacy of treatment in humans especially with it not being available in the United States or Europe for human studies, but it is being studied in animals.
In summary, we detailed a case of colchicine ingestion of 0.39 mg/kg to 0.65 mg/kg leading to progression of MOF leading to death. Our patient did receive supportive care with early administration of activated charcoal even though he presented late after ingestion. We did incorporate poison control into our discussion of management. This case prompts the need for further research into the benefit of N-acetylcysteine for acute liver failure in colchicine overdose whether it be the development of a protocol specific to colchicine since colchicine-specific Fab fragments is not currently available in the United States. A limitation seen is that critical adverse events occur vastly different depending on the dose of colchicine ingested, timing of presentation after ingestion, and if there is an underlying kidney or liver failure.
Footnotes
Authors’ Note
This case was presented as an abstract at the Critical Care Congress Conference, virtual, on January 31, 2021.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
