Abstract
Diffuse carcinomatosis of the bone marrow (DCBM) is a rare clinical condition characterized by diffuse bone marrow involvement with hematological changes. This case study concerns a patient who presented with DCBM secondary to colon cancer with diffuse intravascular coagulation. This is a rare presentation of DCBM in colon cancer. The case study also elaborates on clinical features, pathogenesis, and therapy of this unique presentation.
Keywords
Introduction
Disseminated carcinomatosis of the bone marrow (DCBM) is a unique clinical condition distinct from bone and bone marrow metastasis. It is characterized by diffuse infiltrative growth with hematological abnormalities, including cytopenias, disseminated intravascular coagulation (DIC), or microangiopathic hemolytic anemia. DCBM is a rare occurrence. Common cancers that present as DCBM include stomach, breast, prostate, and lung cancers. Other cancers also reported include pancreatic, esophageal, neuroblastoma, and unknown primary. However, DCBM is a very rare occurrence in colon cancer. DIC, while infrequent in solid tumors, is a frequent but not universal occurrence in DCBM. Pathogenesis and clinical correlations associated with DIC in the setting of DCBM are not well defined. DCBM is often associated with evident bone metastasis on imaging; however, this may not be present in all cases.1,2 This case study highlights the pathogenesis, clinical presentation, and therapeutic challenges posed by this presentation.
Case Presentation
A 75-year-old White female with history of stage IIIB (PT4a,PN2b,M0; 25/26 lymph nodes positive) poorly differentiated mucinous colon cancer (Figure 1) was admitted for back pain. Six months prior, she had a colectomy, followed by one cycle of adjuvant chemotherapy. She refused further chemotherapy on account of toxicities encountered with first cycle. She presented with back pain, which had worsened over 2 weeks along with hematuria, increased bruising, nausea, and vomiting. She was evaluated in emergency department 10 days earlier and treated for an urinary tract infection with ciprofloxacin and azithromycin added on account of ground-glass changes in lung bases. Her oncologist evaluated her 2 days later with persistent back pain and resolving hematuria. Examination revealed bruising in the right flank. Computed tomography (CT) scans did not reveal any progression; however, CEA level was increased at 143 ng/mL. Examination was notable for extensive ecchymosis noted in both the upper extremities and abdomen, but less prominent in the lower limbs.

Poorly differentiated mucinous adenocarcinoma (black arrows) invading through the muscularis propria.
Laboratory Evaluation
Complete blood count revealed mild leukocytosis and neutrophilia (11.4 × 103/µL and 8.0 × 103/µL), normocytic anemia (10.3g/dL), and low platelets (120 × 103/µL). The metabolic panel was normal except for mildly elevated alkaline phosphatase (ALP; 143 U/L; range 38-126). Lactate dehydrogenase (LDH) level was not obtained. Peripheral smear revealed neither significant red cell fragmentation, nor any leucoerythroblastic features. Mild poikilocytosis and increased polychromasia were reported. Coagulation abnormalities noted included prolonged prothrombin time (23.6 seconds; 11.4-14.8) and partial thromboplastin time (45.1 seconds; 22.6-37), increased D-dimer (>20 µg/mL FEU [fibrinogen equivalent units; 0-0.48]) and low fibrinogen (60 mg/dL [207-442]), all consistent with DIC.
Bone Marrow Aspirate and Biopsy
Metastatic carcinoma was noted on bone marrow biopsy involving approximately 30% of the marrow space. The malignant epithelioid infiltrate showed expression of CK20 and CDX2 and lack of CK7 expression, consistent with metastatic colorectal carcinoma (Figure 2).
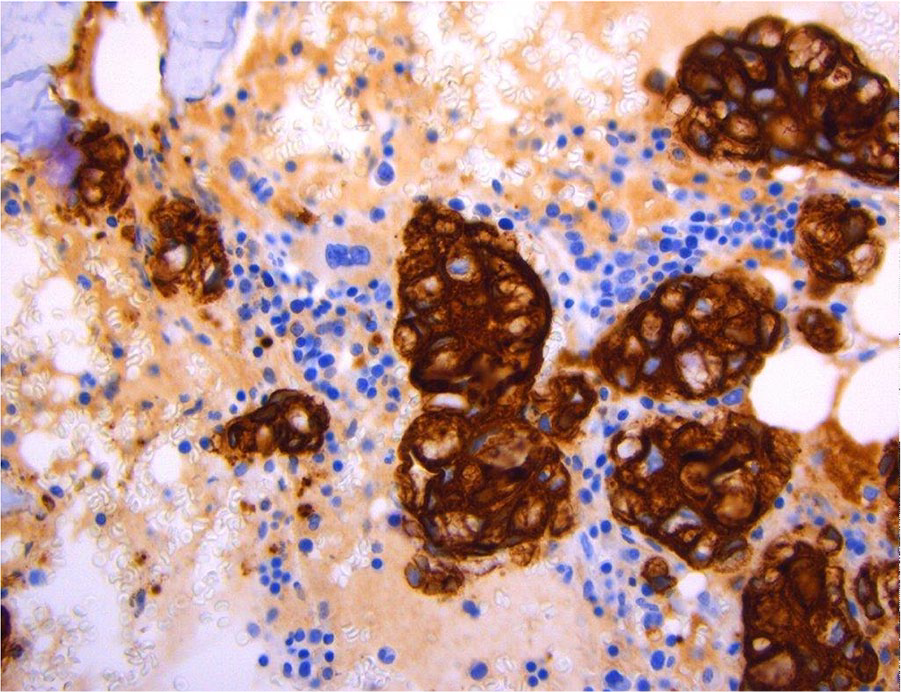
Cytokeratin staining of the bone marrow, positive for CK20 as well as CDX2 and negative for CK7 suggesting metastatic carcinoma.
Treatment
After the diagnosis of disseminated colorectal carcinoma of bone marrow with associated DIC was confirmed by bone marrow evaluation, the patient was initiated on 5-flurouracil, leucovorin, and oxaliplatin (modified FOLFOX6 regimen) for colorectal cancer along with supportive therapy for DIC with fresh frozen plasma and cryoprecipitate.
The patient had significant improvement in pain. Her hematuria resolved and coagulation abnormalities showed improvement; international normalized ratio improved from 2.5 to 1.4 and fibrinogen from 60 mg/dL to 143 mg/dL. Unfortunately, the patient refused further therapy and died in hospice care 2 months later.
Discussion
Skeletal metastasis is rare in patients with colorectal cancer (1% to 10%). In addition, bone marrow metastases occur in only a fraction (around 1%), and DCBM is also exceedingly rare.3,4 Increasing back pain in conjunction with hematological changes (DIC and cytopenias), increasing ALP, and increasing LDH should increase suspicion for DCBM. DIC was noted early on in the presentation of the patients as suggested by the easy bruising, thrombocyotpenia, elevated D-dimer, and low fibrinogen. Elevated ALP reflects bone metastases, which often accompany DCBM. The patient did not show a significantly elevated ALP level due to lack of skeletal metastasis. LDH level was not evaluated in our patient, though LDH level may have been a better reflection of bone marrow involvement. The CT scan was normal in our patient, despite significant back pain being the prominent symptom. Positron emission tomography-CT may be a better study for evaluating bone marrow involvement. 1
Gastric cancer is the leading cause of DCBM, and thus, the most studied. In a gastric cancer study involving a series of 27 patients, signet-ring cell carcinoma and poorly differentiated carcinoma predominated (25/26). The distribution of histology was similar in a smaller number of colorectal cancer patients, and rectal cancers accounted for more than half of the patients compiled in the study. 3 The subject of this case study was reported to have poorly differentiated, mucinous type cancer in the cecum. On bone marrow biopsy during admission, signet ring cells were noted suggesting a correlation between this histology and DCBM.
DIC is a very common occurrence in DCBM, although not universal. Twenty-three of 27 patients with DCBM from the gastric cancer study had laboratory features of DIC. Increased incidence of DIC in DCBM may be secondary to mucinous histology, which is known to be associated with increased incidence of thrombosis, and also with increase in circulating tumor cells associated with DCBM. Tissue factor (TF) expression by tumor cells plays a central role in tumor-induced thrombosis; moreover, TF contributes to tumor growth, metastasis, and angiogenesis. Incidence of thrombosis correlates with the level of TF expression. TF expression on microparticles in addition to tumor cells also contributes to DIC. Independent of TF, mucinous adenocarcinomas can also predispose to thrombosis via mucin-selectin interactions with leukocytes and platelets.1,5-7
Treatment of DIC is mainly based on reversing the underlying process. Outcomes are dependent on the response of the underlying cancer to the proposed therapy. Prompt initiation of chemotherapy in conjunction with other supportive measures can reverse DIC and cytopenias. In addition to reversing DIC, chemotherapy also decreases mortality. Advancements in chemotherapy have been associated with improved survival.1,8
Treatment of underlying cancer may not effectively improve DIC immediately, and supportive care with blood products and antithrombotic measures may be necessary. It is important to utilize blood products only when clinically necessary on account of active bleeding, and not based on coagulation parameters alone. In addition to guidelines-based administration of blood products (platelets, fresh frozen plasma, and cyroprecipitate), patients should be started on unfractionated or low molecular weight heparin to address excessive thrombin production.1,9,10 Thrombomodulin, a glycoprotein expressed on the luminal surface of vascular endothelium, is a multifaceted anticoagulant protein. In addition to anticoagulant action, it exhibits cytoprotective and anti-inflammatory properties. Recombinant thrombomodulin has been approved in Japan for treatment of DIC from sepsis. Thrombomodulin has also been used in treatment of DIC from hematologic malignancies and DIC observed in solid tumors. 8
Despite DCBM exhibiting diffuse infiltration, osteoclast activation is thought to be important in the pathogenesis. RANKL expression has been demonstrated by immunostaining on infiltrating gastric cancer cells. Denusomab, a human monoclonal antibody, which binds to RANKL and is commonly used to treat bone metastasis from solid tumors, is also used in treatment of DCBM. However, the utility in DCBM has not been proven through clinical studies. 8
DCBM is a rare diagnosis in colorectal cancer that imparts poor prognosis. Reversal of DIC and cytopenia is accomplished by prompt treatment of the underlying malignancy with chemotherapy. Case reports indicate that chemotherapy also improves survival. 8 At this time, chemotherapy and supportive care are the mainstays of treatment. It has yet to be seen what role other modalities, including targeted therapy or immunotherapy, will have in the treatment of DCBM with DIC.
Footnotes
Acknowledgements
We would like to acknowledge our pathologist Dr Deborah Johnson for her help and guidance in the diagnosis of our patient.
Author Contributions
Sai Prasad Desikan (guarantor) wrote the initial draft and submitted the report. Raman Desikan (guarantor) edited the draft and provided oncology input. Nathan Mclaughlin participated in the care of the patient and provided input in the case presentation. Charles McClain provided radiology input.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases.
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
