Abstract
Inflammatory fibroid polyps (IFPs) are rare mesenchymal lesions that can arise throughout the gastrointestinal tract. These tumors represent less than 0.1% of all gastric polypoid lesions and are frequently found incidentally on endoscopic evaluation. While presenting symptoms depend on the location of the tumor, gastric polyps commonly present with epigastric pain and early satiety. We hereby delineate the case of a middle-aged female who presented with chronic iron deficiency anemia and a positive fecal occult blood test. She underwent an upper endoscopy, which revealed an actively oozing umbilicated lesion in the gastric antrum. Endoscopic ultrasound divulged the submucosal origin of the lesion. It was subsequently excised using endoscopic mucosal resection. Pathologic examination of the resected specimen confirmed the diagnosis of gastric IFP. Furthermore, we conducted a systematic literature search of the MEDLINE database centered on gastric IFPs from January 2000 till March 2020. The data on patient demographics, clinical features, endoscopic findings, lesion site and size, and treatment approaches were collected and analyzed. This article illustrates the overarching need for clinicians to be vigilant of gastric IFPs presenting with initial clinical symptoms suggestive of occult upper gastrointestinal bleeding. Prompt diagnosis and management of gastric IFPs carry paramount importance to combat chronic unexplained iron deficiency anemia following occult bleeding in such patients. A concoction of endoscopy, biopsy, and immunohistochemical examination can be employed toward their prompt detection. Although gastric IFPs have conventionally been treated with surgery, endoscopic resection is now emerging as a safe and efficient therapeutic modality.
Keywords
Introduction
Gastric inflammatory fibroid polyp (IFP) is a rare clinicopathologic entity that represents 0.1% of all gastric polyps. 1 It usually afflicts adults over a wide age range, with a peak incidence in the sixth and seventh decades of life. 2 These lesions predominantly remain asymptomatic and are often incidentally detected on endoscopy or imaging performed for unrelated causes. Occasionally, they may elicit symptoms due to their large size, causing gastric outlet obstruction (GOO), or due to mucosal erosion presenting with overt or occult upper gastrointestinal bleeding. 3 On endoscopic ultrasound (EUS), gastric IFPs are noted to be located deep inside the mucosa, or submucosally. 4 They penetrate the lamina propria and appear as a macroscopic bulge in the mucosal layer. 4 We elucidate the case of a gastric IFP presenting with iron deficiency anemia (IDA) secondary to occult upper gastrointestinal bleeding, without other gastrointestinal symptoms or signs. Based on the endoscopic and pathologic findings, a gastric IFP was considered as an occult etiology of her IDA. This article highlights gastric IFP as a rare, though noteworthy, cause of occult gastrointestinal bleeding. Clinicians should be cognizant of the potential presence of these lesions in patients with symptomatic chronic IDA and a positive fecal occult blood test (FOBT). Additionally, we conducted a systematic literature review of recent data pertaining to the patient characteristics, presentation patterns, endoscopic findings, clinical associations, lesion size, and management options of gastric IFPs to further substantiate our findings.
Case Presentation
A 50-year-old female presented to our endoscopy suite for an open-access esophagogastroduodenoscopy (EGD) and colonoscopy requested by her primary care physician for evaluation of chronic symptomatic IDA. Her past medical history was significant for unprovoked pulmonary embolism, and she had been on rivaroxaban for the last 6 months. She reported no history of abdominal pain, melena, hematochezia, hematemesis, menorrhagia, weight loss, and nonsteroidal anti-inflammatory drug use. She denied prior liver disease or gastrointestinal surgery. Her family history was negative for gastrointestinal bleeding and cancer. At presentation, she was hemodynamically stable and her physical examination was unremarkable.
Initial hemoglobin obtained as a part of baseline laboratory testing was 8.1 g/dL (12.0-15.5 g/dL), with a mean corpuscular volume of 67.7 fL/cell (80-96 fL/cell) and transferrin saturation of 4% (20% to 50%). Her FOBT turned out positive, while serology for celiac disease was negative. EGD showed an actively oozing umbilicated submucosal lesion in the gastric antrum, with a greatest dimension of 10 mm (Figure 1). The lesion was treated with bipolar electrocoagulation that effectively curbed the hemorrhage. Additionally, colonoscopy performed on the same day was unremarkable. EUS demonstrated a 10-mm deep mucosal or superficial submucosal lesion manifesting superficial erosion, with no evidence of invasion into the surrounding structures or deeper layers (Figure 2). It was removed en bloc by endoscopic mucosal resection after the administration of a submucosal injection. Pathologic examination of the resected specimen revealed superficial erosion with inflammatory submucosal infiltrate and fibroblastic proliferation (Figure 3). The stroma of the lesion consisted of bland spindled cells, small vessels, and eosinophil-rich mixed inflammatory infiltrates (Figure 4). High-power examination showed a perivascular onion-skinning pattern and an inflammatory infiltrate composed of lymphocytes and eosinophils (Figure 5). The patient responded well to the endoscopic resection and was discharged home in a stable condition. At the 2-month follow-up, she demonstrated unremarkable recovery with resolution of her IDA-related symptoms. Her hemogram revealed normalization of hemoglobin (12.7 g/dL) after oral iron therapy. She continues to do well to date.

Endoscopic appearance of a bleeding submucosal lesion (arrow) with umbilication, located in the gastric antrum.

Endoscopic ultrasound showing a hypoechoic lesion (as pointed out by the label—mucosal nodule) arising from the gastric submucosa and extending into the mucosa.

Pathologic examination of the resected gastric polyp showing a superficial erosion, inflammatory submucosal infiltrate (star), and fibroblastic proliferation (arrowhead) (hematoxylin and eosin staining; 40×).
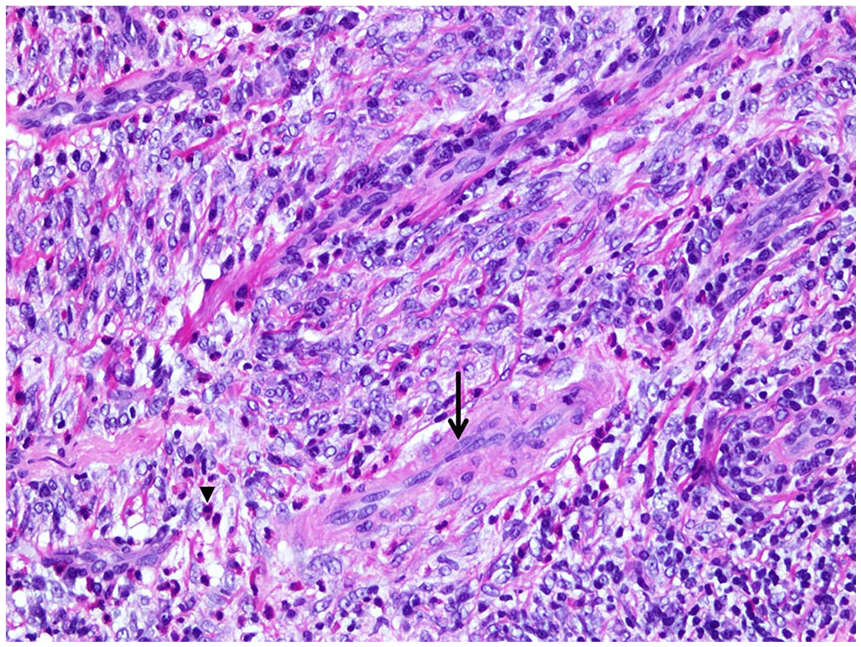
Tumor showing bland spindled cells (arrow), small vessels, and an eosinophil-rich mixed inflammatory infiltrate (arrowhead) (hematoxylin and eosin staining; 200×).

Tumor showing inflammatory submucosal infiltrate of eosinophils and lymphocytes, fibroblastic proliferation, and perivascular onion skinning (arrow) (hematoxylin and eosin staining; 400×).
Discussion
IFP was initially reported as “polypoid fibroma” in 1920 by Konjetzny. 5 Vanek 6 described 6 cases of similar lesions in 1949 and named them “gastric submucosal granuloma with eosinophilic infiltration.” Later, Helwig and Ranier 7 first coined the term inflammatory fibroid polyp in 1953. Since then, several case reports and small case series of IFPs have been described. These lesions are still relatively rare, accounting for less than 0.1% of all gastric polyps. 2 IFPs are mesenchymal tumors that originate in the mucosa and submucosa of the gastrointestinal tract. Although recent literature favors their benign behavior, local invasion into the subserosa and destruction of the muscularis propria has also been described. 8 The exact pathogenesis of this tumor remains unclear. However, a reactive phenomenon resembling granuloma formation due to unknown triggers has been considered plausible. 9 The proposed causal associations include Helicobacter pylori infection, parasitic infestation, hypersensitivity reactions, and physical and metabolic factors.9,10 Familial clustering and PDGFRA gene mutations have also been reported in patients with IFPs.11,12
In order to evaluate the recent trends pertaining to gastric IFPs, we conducted a systematic search of MEDLINE database. A total of 52 cases published between January 2000 and March 2020 were identified.13-51 The mean age of patients was 63 years (range = 19-93 years). Female gender predominance was noted, with 60% of the patients (n = 31) being females (female-to-male ratio: 1.5:1). Although a clear geographical distribution has not yet been established, most sporadic cases (23%, n = 12) included in this review were reported in Japan. That said, one case series from the Czech Republic described 14 cases. 14 In aforementioned studies, gastric IFPs predominantly afflicted the gastric antrum (61%) followed by pyloric/pre-pyloric region (15%), cardia (7%), and body of the stomach (4%). Gastric sites were not specified in 13% of cases.
Previously, occult upper gastrointestinal bleeding has been attributed to several etiologies, including angiodysplasia, celiac disease, esophagitis, peptic ulcer disease, gastric cancer, and gastritis. 9 However, gastric IFP as a cause of occult upper gastrointestinal bleeding, presenting as IDA and positive FOBT testing, is an exceedingly rare entity with only a handful of cases reported thus far.1,40 It is imperative to highlight that our patient was on anticoagulation at the time of presentation, which might have predisposed her to the bleeding from the gastric IFP. In this review, 17% of the lesions were asymptomatic and were diagnosed incidentally on routine checkups or while investigating for other medical conditions. In symptomatic patients (83%), the presenting features correlated with the size and location of the lesion. Gastric IFPs of relatively larger sizes involving the gastric antrum may present with an intermittent GOO referred to as “ball-valve syndrome.” 45 In this analysis of previously reported cases, gastric clinical symptoms were dominated by epigastric pain (35%, n = 18) followed by overt gastrointestinal bleeding (8%, n = 4), nausea and vomiting (8%, n = 4), and dyspepsia (4%, n = 2) (Table 1).
General Characteristics of Cases of Gastric Inflammatory Fibroid Polyp Reported Between 2000 and 2020.
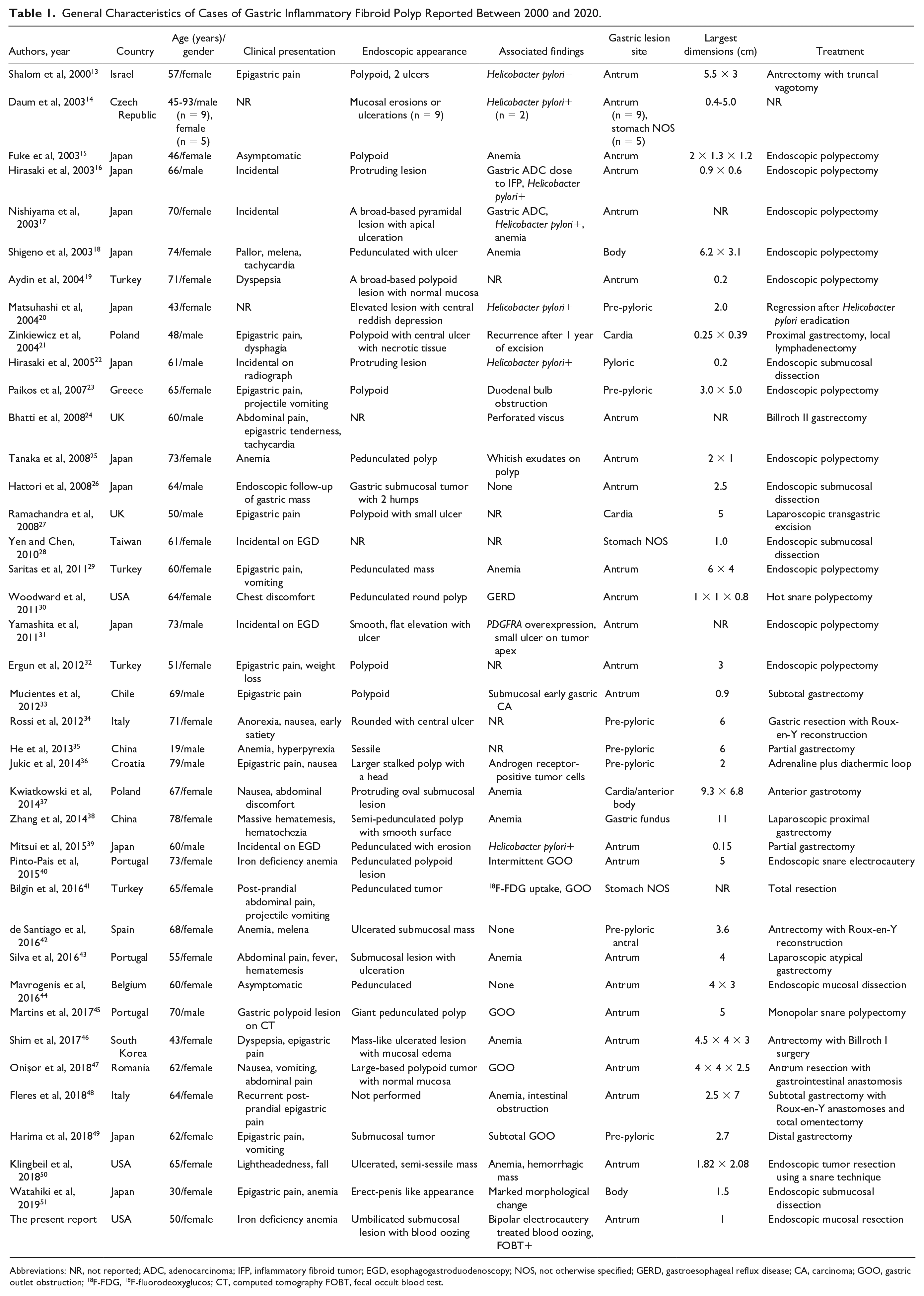
Abbreviations: NR, not reported; ADC, adenocarcinoma; IFP, inflammatory fibroid tumor; EGD, esophagogastroduodenoscopy; NOS, not otherwise specified; GERD, gastroesophageal reflux disease; CA, carcinoma; GOO, gastric outlet obstruction; 18 F-FDG, 18 F-fluorodeoxyglucos; CT, computed tomography FOBT, fecal occult blood test.
EGD often divulges firm, solitary, sessile, or pedunculated lesions. These tumors may also show mucosal ulcerations. 52 Even though the actual diagnostic role of EUS is not clear, it may help in better characterization. EUS shows gastric IFPs as hypoechogenic and homogeneous lesions with inconspicuous margins, originating from within the second and third mucosal layers. Internal echoes indicate the presence of blood vessels in these tumors. 53 Gastrointestinal stromal tumors (GISTs) can be differentiated from IFPs owing to their transmural growth and distinct margins.
In this review, gross morphologic features of gastric IFPs included polypoid or pedunculated (40%, n = 21), ulcerated mass (33%, n = 17), sessile (3%, n = 6), protruding (6%, n = 3), and oval-to-round lesions (4%, n = 2). The diameter of gastric tumors varied widely in these cases, ranging from 0.15 cm to 11 cm. Clinical associations or complications such as anemia (19%, n = 10), H. pylori positivity (13%, n = 7), GOO (13%, n = 7), gastric adenocarcinoma (6%, n = 3), and gastrointestinal ischemia (2%, n = 1) were also reported. Cross-sectional features of IFP on imaging were usually nonspecific, showing gastrointestinal intraluminal growth. Additionally, diffusion-weighted magnetic resonance imaging (DWI or DW-MRI) can show a peripheral enhancement likely due to a significant hypervascularity in the IFPs. It shows an external ring of accelerated diffusion (peripheral fleshy bud) followed by an internal ring of restricted diffusion (fibrous ring). The central part correlates with the edematous and myxoid area, which is a testament to the homogeneous diffusion observed.48,54
Based on imaging studies and endoscopic evaluation, differential diagnoses of gastric IFP include GIST, leiomyoma, inflammatory pseudotumor, solitary fibrous tumors, spindle cell carcinoid, hemangioendothelioma, schwannomas, hemangiopericytoma, T-cell lymphoma, and pancreatic rests.48,55 Diagnostic yields of endoscopic biopsy and EUS are low, with only 10% of gastric IFPs diagnosed in the preoperative setting. The diagnosis is thus often established on histopathology following complete resection.21,56 On hematoxylin and eosin staining, submucosal proliferation of spindle cells along with prominent blood vessels, circumferentially surrounded by fibroblasts, is a major histologic feature. 57 Historically, an inflammatory infiltrate of eosinophils and macrophages has also been described. This characteristic finding is described as the onion-skin appearance. Ectatic blood vessels with irregular shapes are also found concurrently with muscular walls of variable thickness. 57 Immunohistochemical analysis is helpful in differentiating gastric IFPs from GISTs since both the lesions are CD-34 positive, but only GISTs are positive for CD-117 (c-kit). Due to overlapping immunohistochemical features and exclusive occurrence in the gastrointestinal tract, Wille et al 57 considered IFPs as the nonneoplastic counterparts of GISTs.
With regard to treatment of gastrointestinal IFPs, endoscopic polypectomy can be employed in polypoidal lesions.58,59 Notably, this therapeutic modality may be associated with certain complications such as incomplete resection of the lesion or potential risk of gastrointestinal perforation. 59 These lesions usually do not show recurrence and local or distant metastasis. However, in sessile lesions, endoscopic resection may also incur the risk of local recurrence due to a submucosal growth pattern. Surgical intervention may be warranted for en bloc resection in patients with giant gastric IFPs. In this context, wedge resection is considered curative and yields negative margins. In this review pertaining to gastric disease, endoscopic treatment was used in 38% (n = 20), while surgery was performed in 33% (n = 17) of patients. Endoscopic therapeutic modalities such as polypectomy (19%), submucosal dissection (10%), snare cautery (8%), and adrenaline plus diathermic loop 2% were used. It is also notable that (2%) of patients showed regression of the gastric IFP lesions after H. pylori eradication therapy. Zinkiewicz et al 21 described a unique patient with an IFP located in the cardia, which was removed endoscopically. However, after 12 months, the lesion recurred, and the patient had to undergo curative proximal gastrectomy with local lymphadenectomy. 21 Therefore, gastric IFPs may show a propensity to recur. The existing clinical evidence is insufficient and cannot be extrapolated to denote EUS or histopathologic features that may portend a malignant trajectory. Thus, no post-resection surveillance guidelines are currently available.
Learning Points
This study prompts clinicians to include gastric IFP in the list of differentials of occult upper gastrointestinal bleeding.
The clinical features of IDA and/or positive FOBT in a female patient should raise the possibility of an IFP, warranting endoscopic examination.
Based on this review, gastric IFP remains one of the rare benign tumors of the stomach, which most commonly involves the gastric antrum. The precise pathogenesis of this entity is yet to be determined.
Overall, a majority of patients are asymptomatic. However, symptomatic individuals are now increasingly presenting with epigastric pain, early satiety, or symptoms of GOO.
Preoperative diagnosis remains a challenge. Histopathologic analysis with immunohistochemical staining is the mainstay of diagnosis.
Complete resection of gastric IFPs is generally recommended in patients with bleeding or those with symptomatic lesions. Given the lack of evidence for malignant potential or recurrence, postresection endoscopic surveillance is not performed in most cases.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
