Abstract
Aim:
Neopterin is a marker associated with cell-mediated immunity produced by interferon-Ɣ-stimulated macrophages. The levels of neopterin in body fluids are elevated in infections, autoimmune diseases, malignancies, and allograft rejections. Recently, it was shown to be associated with the initiation and progression of periodontal disease. This investigation was designed to explore the use of salivary neopterin in predicting the outcome of nonsurgical periodontal therapy (NSPT) in aggressive and chronic periodontitis patients.
Materials and Methods:
This case-control study with stratified sampling comprised 45 subjects divided into three groups: a control group of 15 periodontally healthy participants, a test Group A of 15 participants with chronic periodontitis, and a test Group B of 15 participants with aggressive periodontitis. Periodontal parameters, including plaque index (PI), gingival index (GI), probing depth (PD), and clinical attachment level (CAL), were recorded at baseline and three months after scaling and root planing (SRP). An enzyme-linked immunosorbent assay (ELISA) analysis was performed to measure neopterin levels in saliva in study groups before and after SRP. Kruskal–Wallis analysis of variance and Mann–Whitney U tests were done for intra- and intergroup comparisons. The correlation between salivary neopterin levels with clinical parameters was done by Karl Pearson’s correlation coefficient.
Results:
Baseline salivary neopterin levels were least in the control group (5 62 nmol/L), higher in test Group A (9.30 nmol/L), and highest in test Group B (9.76 nmol/L). Three months after SRP, there was a significant reduction in all the clinical parameters, and the reduction in mean salivary neopterin levels were 2.79 and 2.57 in test Group A and test Group B, respectively; the difference was not statistically significant.
Conclusion:
Salivary neopterin levels have the potential to be utilized as a diagnostic tool for detecting the presence, activity of the disease, and the course of the lesions. It cannot, however, be utilized to distinguish between chronic and aggressive periodontitis.
Introduction
Periodontitis is an inflammatory disease characterized by the destruction of the tooth-supporting structures (periodontal ligament and the alveolar bone) resulting from the interaction between the host defense cells with dental biofilm-associated microorganisms. 1 , 2 As reported in earlier studies, host factors, such as heredity, and environmental factors, such as smoking, are important determinants for periodontal disease. Evidence states that major tissue destruction in established periodontitis, like chronic periodontitis, is because of the recruitment of host cells via activation of monocytes/macrophages, lymphocytes, fibroblasts, and other cell types. 3 An intermediate mechanism between bacterial stimulation and tissue destruction is the production of cytokines, which stimulates inflammatory events that activate effector mechanisms. 4 However, in aggressive periodontitis, direct and indirect bacteriological effects influencing the body’s immune system play a role in destroying periodontal structures. 5
Routine clinical diagnostic methods provide reliable information about the conventional periodontal parameters, i.e., bleeding on probing, probing depth, clinical attachment level (CAL), etc., and radiological findings describe only past disease activity. However, they don’t seem to be reliable diagnostic indicators of active periodontal disease because of their site-specific distribution. A consistent approach would be assessing the substances in the body fluids that correlate with the level of disease activity and tissue destruction. 5
Neopterin, a metabolite of guanosine triphosphate, belongs to the class of chemical compounds known as pteridines; it is an early and valuable indicator of cellular immunity, which increases in parallel with the clinical course of infection with viruses, intracellular bacteria, and parasites. It is biologically stable and can be quantified easily in human fluids, serum, and urine. Determination of neopterin levels reflects the stage of activation of the cellular immune system, which is important in the pathogenesis and progression of various diseases like viral infections, autoimmune or inflammatory diseases, and rejection episodes following allograft transplantation, and several malignant diseases. 6 Recent evidence also indicates an association between the severity of inflammation in SARS CoV-2 infection and neopterin levels characterized by a profound exacerbation of immune activation. 7 , 8
Although serum and gingival crevicular fluid (GCF) is an ideal medium to assess the biological indicators, saliva is appealing to use in oral-related diseases because of its easy and noninvasive nature in terms of its collection and its abundant nature. It is believed that the measurement of neopterin in saliva might be useful for diagnosing and predicting the prognosis of periodontal disease because it has been well-documented that T cells in addition to other inflammatory infiltrates, mediate the immunopathological events in periodontal disease. 9 In light of the abovementioned facts, the present investigation aimed to explore the role of salivary neopterin levels in predicting the outcome of nonsurgical periodontal therapy (NSPT) in aggressive and chronic forms of periodontitis.
Materials and Methods
Study Design and Ethical Clearance
This was a prospective case-controlled clinical and biochemical study carried out at Vishnu Dental College from January 2017 to March 2019 after obtaining institutional ethical clearance (ref: VDC/RP/2016/09) from the institutional research board. The participants were recruited from the outpatient pool attending the Department of Periodontics and Implantology. A single investigator/observer carried out all the evaluations.
Selection Criteria
The sample size was computed using G*Power 3.1 software (IBM, Newyork, USA), with the level of significance set at 5.1%, the power of the study set at 80%, and an estimated effect size of 0.66, requiring 12 samples per group in all three groups. Given a 20% dropout rate, the sample size was rounded to 15 per group. The samples were selected using stratified sampling.
Participants in the age range of 16 to 65 years with at least 18 teeth in the mouth who have not received periodontal treatment in the past six months, including oral prophylaxis, were included. Participants with a history of loss of teeth because of periodontal disease, uncontrolled systemic/infectious/inflammatory diseases, smokers, and pregnant women who have taken antibiotics/anti-inflammatory agents and immunosuppressants during the previous six months were excluded. Patients diagnosed with the systemic disease during the study period after enrolment were excluded.
After meeting the inclusion criteria, participants underwent complete medical and dental examinations. Primary outcome variables comprised probing pocket depth (PPD), CALs, salivary neopterin levels, and secondary outcome variables, i.e., plaque index (PI) and gingival index (GI). All the clinical parameters, i.e., PI, 10 GI, 11 PPD, and CAL were recorded with the University of North Carolina 15 (UNC 15) probe using an acrylic stent for standardization.
Patient Selection
A total of 45 patients (control group—15 periodontally healthy; test Group A—15 chronic periodontitis; test Group B—15 aggressive periodontitis) meeting all the inclusion criteria were considered and allocated to the three groups. Written informed consent was obtained from all the patients in both vernacular and English languages.
Based on the American Academy of Periodontology (AAP) 1999 classification of periodontal diseases, participants were categorized as: the control group (periodontally healthy) with uniformed probing depths (PDs) of 3 mm or below and with no evidence of loss of attachment but might be showing some signs of gingival inflammation with no bone loss confirmed by radiographs; test Group A (chronic periodontitis)—participants with CAL of ≥3 mm in at least four sites in different teeth, confirmed by the radiographic evidence of bone loss; test Group B (aggressive periodontitis)—participants with rapid attachment and bone loss in otherwise healthy individuals, generalized attachment loss affecting at least three teeth other than first molars, and incisors with CAL >4 mm in the affected sites. 12
Saliva Sample Collection
All participants were instructed to rinse their mouth with water thoroughly. About 2 mL of unstimulated saliva was collected in the Eppendorf tube. All the collected samples were stored at -20ºC and subjected to centrifugation at 3000 rpm for 10 min at room temperature. After centrifugation, the supernatants obtained were stored at -20°C until the analysis of salivary neopterin.
Evaluation of Salivary Neopterin
Analysis of salivary neopterin was carried out through enzyme-linked immunosorbent assay (ELISA) using human neopterin (Np) ELISA Kit Catalogue No E13651281, of Sincere™ (Sincero Biotech Co. Lid. Berg, China). 100 µL of saliva samples were pipetted into the appropriate walls, sealed, and incubated for 90 min at 37°C. Washing twice with 350 Tris-buffered saline (TBS) solution, the walls were then sealed with the sealing membrane, gently shaken, and incubated for 60 min at 37°C. 100 µL of biotinylated human neopterin antibody working solution was added to each well and incubated at 37°C for 60 min. Each well was washed five times with 350 µL TBS solution. 100 µL of prepared avidin-biotin complex (ABC) working solution was added into each well except the control well, and the plate was incubated at 37°C for 30 min. Each well was washed five times with 35041 TBS for around 1 min. 100 mL of prepared 33, 5, 6-tetramethylbenzidine (TMB) working solution was added into each well, and the plate was incubated at 37°C for 30 min away from light. When shades of blue could be seen in the wells, 100 µL of prepared TMB stop solution was added to each well to stop the reaction. Taking blank well as zero, the measurement of optical density (OD) under 450 nm wavelength was carried out within 10 min.
Treatment Procedure
In both test groups, after the collection of saliva sample, all the participants underwent scaling and root planing (SRP) with reinforcement of oral hygiene instructions. Patients were informed to revisit after three months. At three months, saliva was collected to evaluate neopterin levels, and all the clinical parameters were reassessed.
Statistical Analysis
Data collected were subjected to statistical analysis using SPSS software (v.21, IBM, NY). Kruskal–Wallis ANOVA test was used for intragroup comparisons. Intergroup pair-wise comparisons were made using Mann–Whitney U test. The correlation between salivary neopterin levels with other clinical parameters in all groups was done by Karl Pearson’s correlation coefficient. P < .05 was considered to be statistically significant. A level of significance of 5% was assumed (P < .05). The statistical power of the study obtained was 86.5%.
Results
At baseline, all the periodontal parameters and salivary neopterin levels were assessed in control and test groups. In both the test groups, the periodontal parameters and salivary neopterin levels were reassessed three months after SRP.
A total of 21 males and 24 females participated in the study (six males, nine females in test Group A; four males, 11 females in test Group B; 11 males, four females in the control group). The age of participants ranged from 21 to 58 years, with a mean age of 39.5 years.
At baseline, there was a statistically significant difference in mean PI scores between the control and test groups. There was a statistically significant reduction of 1.02 in mean PI scores three months after SRP in test Group A (P = .001) and 0.54 in test Group B (P = .001) whereas intergroup variation between test groups (0.64 and 0.67) is statistically not significant (P= .03; Table 1). Similarly, at baseline, there was a significant difference in mean GI scores between control and test groups. Intragroup comparison of GI scores showed a considerable difference between baseline and three months in both the test groups. (P < .001)
Comparison of Periodontal Parameters and Salivary Neopterin Levels Between Study Groups at Baseline
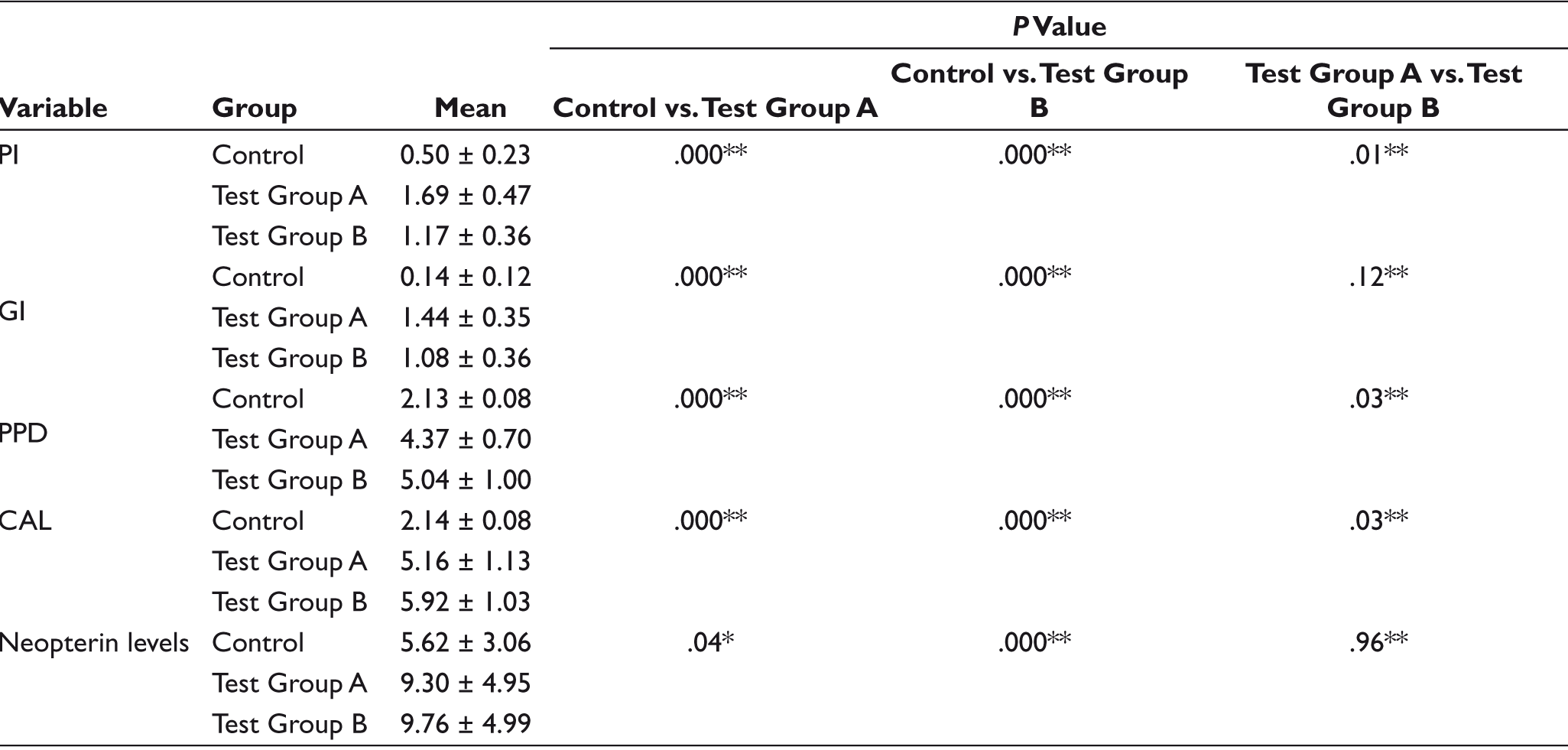
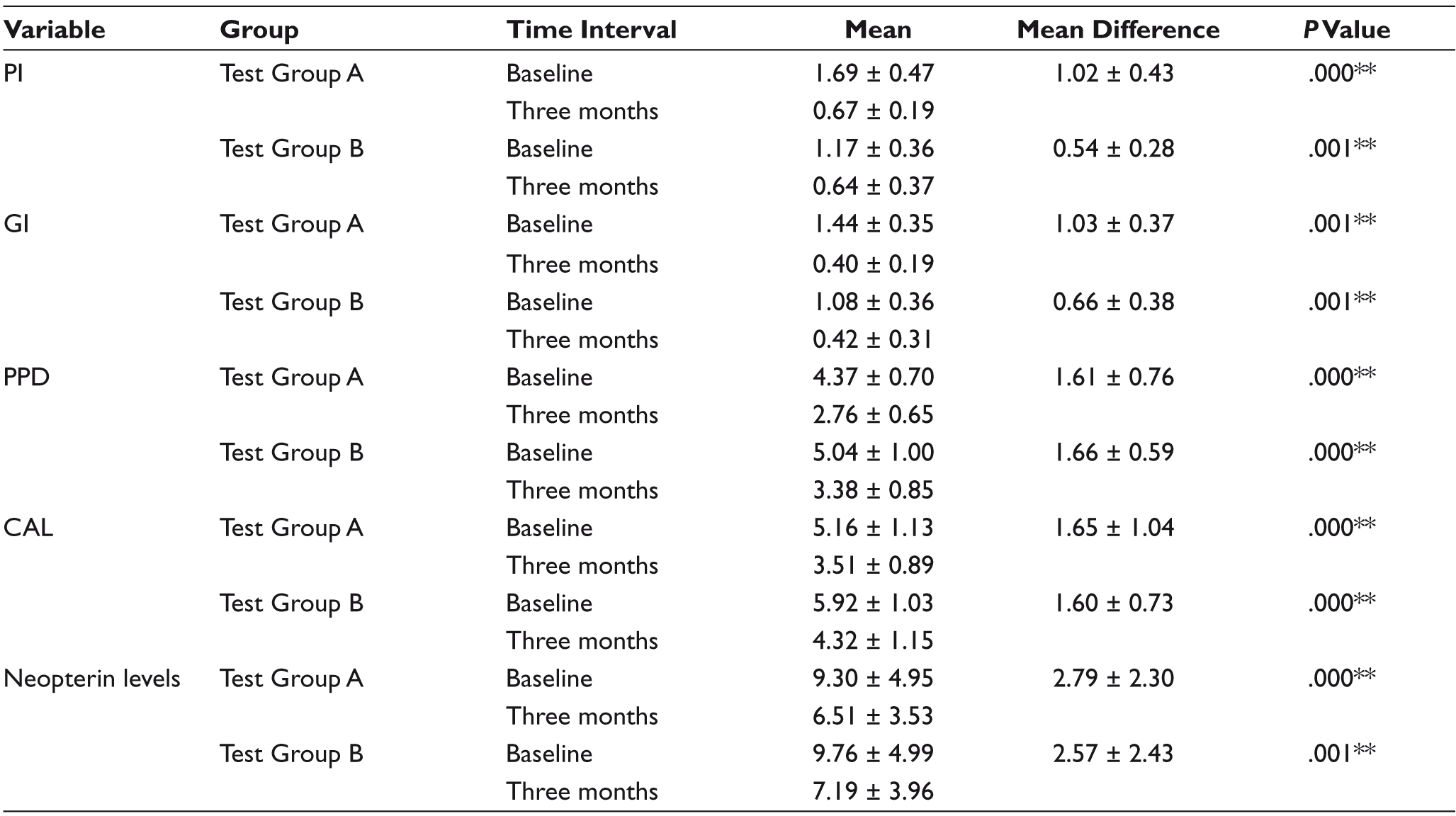
At baseline, there was a statistically significant difference in mean PD scores between the control and test groups. There was a statistically significant reduction of 1.61 in mean PD scores three months after SRP in test Group A and 1.66 in test Group B (P < .001), and intergroup variation between test groups is statistically significant (P = .03; Table 1). Similarly, at baseline, the mean CAL was observed to be statistically significant between the control group and both the test groups, but there was no statistical significance when compared to both the test groups (P- = .06; Table 1). In test Group A, a reduction of 1.65 in mean CAL scores three months after SRP and 1.60 in test Group B (Table 2).
The mean levels of salivary neopterin were found to be the least in the control group at baseline. However, the mean salivary neopterin levels in test Group A and test Group B were found to be curtly similar. While comparing the intergroup variations of neopterin levels in the control group with test Group A and test Group B, it was statistically significant (P =.04 and .000, respectively; Table 1). There was a statistically significant reduction of 2.79 in mean neopterin levels three months after SRP in test Group A (P = .000) and 2.57 in test Group B (P = .0001), whereas intergroup variation between test groups (6.51 and 7.19) is statistically not significant (P = .62; Table 2).
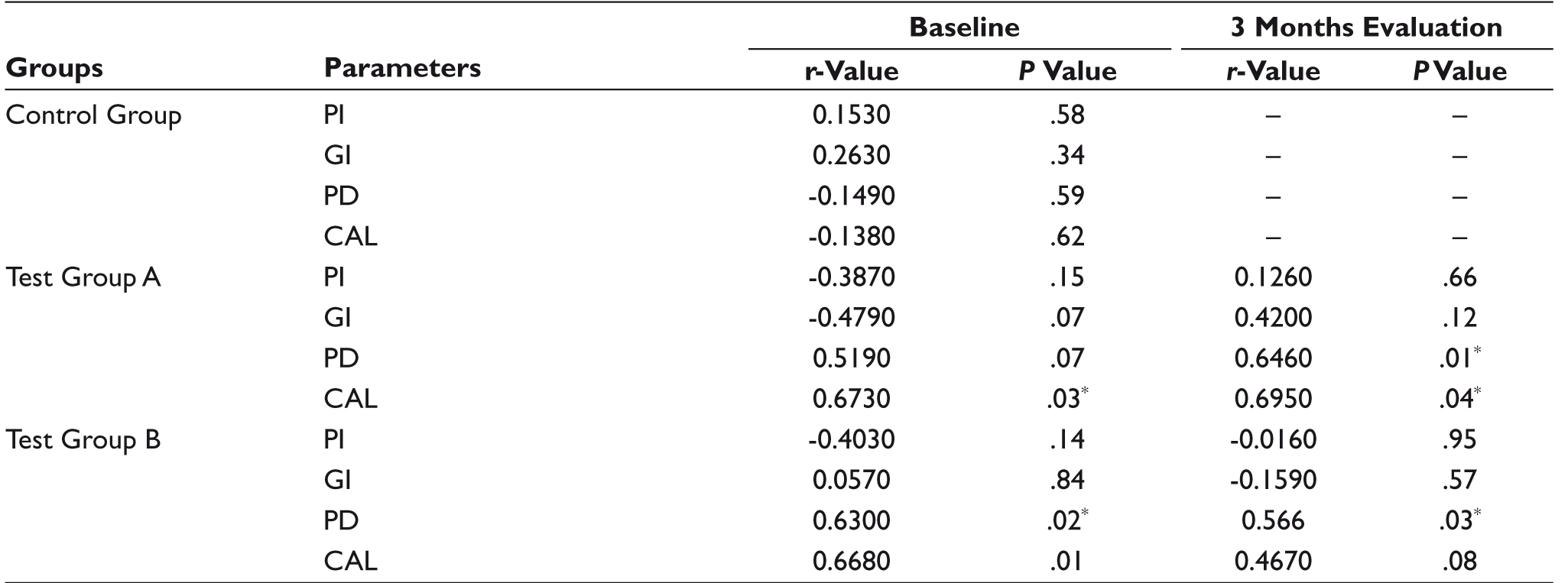
Salivary neopterin levels were found to have no positive correlation with periodontal parameters in the control group. In the test Group A, neopterin levels have a moderately strong positive correlation with PD preoperatively (r = .52) and a strong positive correlation with postoperative PD, pre and postoperative CAL (r > 0.6), which are statistically significant (P < .05). Similarly, in test Group B, salivary neopterin levels were found to have a strong positive correlation with PD and CAL preoperatively and moderately strong correlation with PD and CAL postoperatively, which are statistically significant (P < .05; Table 3).
Comparison of Periodontal Parameters and Salivary Neopterin Levels in Test Groups After NSPT
Correlation Among Salivary Neopterin Levels and Various Periodontal Parameters in Study Groups
Discussion
Periodontitis is an inflammatory disease involving the soft tissue and bone surrounding the teeth in the jaws, eventually leading to exfoliation of teeth following the diminution of periodontal supporting tissues. This process is attributed to the release of toxic products from the pathogenic bacterial plaque in addition to the inflammation of gingival tissues elicited by the host response. Complex inflammatory and immune reactions are involved in the progression of periodontitis.13–15
Recent research has focused on objective measures such as biomarkers to assess the current disease status and to predict risk and outcome of the treatment provided. Various molecules, including inflammatory mediators, host-derived enzymes, leukocyte breakdown products, and antimicrobial peptides, have been identified in oral fluids like saliva and GCF. Along with various inflammatory mediators that can be assessed in periodontitis, neopterin is one of the biomarkers that can be measured in inflammatory conditions. 16
Among the various modes of identification of neopterin in oral fluids, saliva offers many benefits over other biologic fluids in its easy and noninvasive way of collection, which is under the control of the clinician and its availability in adequate amounts. Hence, saliva was preferred as the diagnostic tool in our study for neopterin analysis. Earlier studies evaluated salivary neopterin levels in chronic periodontitis patients and compared them with periodontally healthy individuals, and the authors concluded that salivary neopterin levels are higher in chronic periodontitis patients compared to healthy individuals. 17 , 18
Similarly, a study was conducted to assess neopterin levels in GCF of aggressive periodontitis patients and periodontally healthy individuals, and the authors concluded that there were elevated levels of neopterin in periodontitis compared to health. 2
A study was done to assess neopterin levels in different body fluids such as GCF, saliva, and urine that reported involvement of neopterin in aggressive periodontitis, and this might be considered of value in understanding periodontal disease mechanisms. 19 However, there were no studies in the literature comparing neopterin levels in chronic and aggressive periodontitis and their usefulness in predicting the treatment outcome. Therefore, the present study aimed to assess the neopterin concentration in saliva samples from individuals with healthy periodontium, with chronic and aggressive forms of periodontitis, and their correlation with the treatment outcome.
The results of the present study demonstrated that the mean salivary neopterin levels were 5.62 ± 3.06 nmol/L in the control group and 9.30 ± 4.95 nmol/L in test Group A and 9.76 ± 4.99 nmol/L in test Group B at baseline which were in accordance with the results of studies, stating that salivary neopterin concentrations are significantly elevated in patients with periodontitis. An earlier study by Sahi et al. in 2019 reported a similar elevation in GCF neopterin levels in periodontitis patients compared to healthy and chronic gingivitis patients. 20 Because T cells, in addition to other inflammatory infiltrates, promote the immunopathologic processes in periodontal disease, evaluating salivary neopterin levels might be beneficial for diagnosing and predicting periodontal disease.
Although there was a significant difference in salivary neopterin levels between control group and test groups, there was no significant difference when compared between test groups. However, there are a few limitations in the present study; severe forms of periodontitis were not included, which could have affected the neopterin levels.
In the present study, the results have shown that PI, GI scores along with pocket depth and CAL values were decreased in test groups after NSPT, i.e., SRP, compared with baseline values. These results suggest nonsurgical periodontal treatment could effectively improve periodontal tissue conditions and reduce in inflammation. The reduction in salivary neopterin levels three months after SRP in test groups was considerably significant. Moreover, the levels of salivary neopterin in individuals after receiving treatment were compared with those in periodontally healthy individuals. From these results, it may be speculated that changes in the microbial composition and the ongoing inflammatory process in the pocket environment might have a close relationship with the neopterin levels in saliva. Reductions in key cells responsible for neopterin production in the periodontium, such as macrophages and inflammatory leukocytes, following treatment, may be an additional reason for diminished levels of salivary neopterin concentrations after nonsurgical periodontal treatment. The significant reduction in clinical parameters and their values close to the normal limits indicate the stability of the disease and a nonprogressive disease process. A previous study by Prasanna et al. in 2017 reported a reduction in salivary neopterin levels three months after NSPT in both pre and postmenopausal women, suggesting that neopterin can be used as a diagnostic marker for periodontal inflammation and destruction. 21
Inflamed periodontium may act as a focus for the spread of systemic inflammation resulting in systemic conditions which may be difficult to treat. An earlier study by TurgutÇankaya et al. in 2018 evaluated the GCF levels of neopterin in chronic periodontitis and acute myocardial infarction before and after periodontal therapy and reported a possible improvement in the efficiency of myocardial infarction treatment after periodontal treatment because of reduction in the infectious load of the body associated with inflamed periodontium. 22
Limitations
Mild to moderate forms of periodontitis were included, and only NSPT was included. Further studies, including severe forms of periodontitis requiring surgical and regenerative periodontal therapy, may add value for using neopterin as a diagnostic and prognostic biomarker.
Conclusion
From the present study, it can be assessed that salivary neopterin levels can be used as a potential tool for diagnosing the presence or absence of disease, the activity and stability of the disease, and the progression of the lesions. However, it cannot be used to differentiate the different types of periodontitis, i.e., chronic or aggressive forms.
Footnotes
Acknowledgements
The authors would like to thank Dr. C. D. Dwarakanath for his support and guidance in the study.
Authors’ Contribution
Conceptualization: Chaitanya A., Sruthima N.V.S. Gottumukkala
Formal Analysis: Chaitanya A., Sruthima N.V.S. Gottumukkala
Investigation: Sruthima N.V.S. Gottumukkala, Ramesh
Gautami S. Penmetsa, Suneetha Koneru
Writing—Original Draft: Chaitanya A., Sruthima N.V.S. Gottumukkala
Writing—Review and Editing: Sruthima N.V.S. Gottumukkala, Ramesh K.S.V.
K.S.V., Gautami S. Penmetsa, Swetha Pasupuleti Methodology: Sruthima N.V.S. Gottumukkala, Swetha Pasupuleti, Suneetha Koneru
Project Administration: Chaitanya A., Ramesh K.S.V.,
Ethical approval institutional statement
Ethical clearance (ref: VDC/RP/2016/09) was obtained from the Institutional research board.
Data Availability Statement
The publication contains all of the data supporting the current study. There is no extra information available.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for research, authorship, and/or publication of this article.
Patient Declaration of Consent
Participants were included in the study after obtaining written informed consent.
