Abstract
Aim:
A systematic review of the methods of 3D printing and the materials used so far for the manufacture of abutments was performed to evaluate whether their clinical use is indicated through the mechanical, chemical, and biological analyses carried out.
Materials and Methods:
An electronic search conducted by three independent reviewers was carried out in the PubMed, Web of Science, Cochrane Library, Science Direct, and Lilac databases. The inclusion criterion was researching articles in English that contained as subject the manufacturing of abutments through 3D printing/additive manufacturing. Any meta-analyses, reviews, book chapters, abstracts, letters, conferences papers, and studies without abutments printed were excluded.
Results:
We found 780 references, which after applying the exclusion criteria resulted in the final inclusion of seven articles for review. The studies had a high heterogeneity, showing different materials and methodologies to manufacture abutments, which makes a comparison between them difficult, and for this reason it was not possible to carry out a meta-analysis with the data found.
Conclusions:
Even with the limitations found in the present research, it is possible to conclude that printed abutments have adequate mechanical, chemical, and biological properties that can indicate their clinical use. 3D printing presents high accuracy and speed and can produce customized abutments according to each case.
Introduction
Technological advances have promoted automation of the production of dental materials, allowing optimization of the time of production, reduction of cost, possibility of structures with different geometries and complexities, and new treatment approaches by clinicians.1–5 Among the means of production Computer-Aided Design (CAD) and Computer-Aided Manufacture (CAM) and three-dimensional (3D) printing are considered versatile due to their varied options of use in the dental field, such as manufacturing of prosthetic restorations, surgical guides, implants, protocol structures, and abutments, thus optimizing the dental treatment for patients and professionals.6–9 Additive manufacturing, also called 3D printing, is based on addition, where the final object is obtained by a layer-on-layer deposition of source material.10, 11
Factors such as specific gingival phenotype, sex, age, the interocclusal and mesiodistal space, bite force, the biocompatibility of the selected material, and the need for hygiene need to be considered to select implant abutment in addition to issues such as thickness, height, existing micro gap between the implant and prosthesis.12–16 Of these, studies are conducted with different abutment designs and materials for reasons of durability, so that they are more durable and more biocompatible with peri-implant tissues. Thus, it becomes necessary to develop custom abutments for the result of each treatment to be optimized and individualized. For the customization of abutments, the oral cavity is scanned with an intraoral scanner to obtain the topography, size, height, and diameter of the gingival emergence profile so a specific design of the abutment can be made with CAD software. With the specific design created, the abutment can be manufactured by 3D printing.17, 18
The correct selection of the material used for printing directly interferes with the success of the treatment, as it needs a correct adaptation in the implant-abutment interface to maintain the health of peri-implant supporting tissue. Different materials have been used for the manufacturing of abutments, such as plastic, titanium, gold, zirconia, and alumina. 19
According to Rokaya et al. 20 the most used material for manufacturing abutments is titanium and its alloys due to their good mechanical and biological properties and corrosion resistance. Pure titanium can be classified into four grades. Each grade has different mechanical properties and is classified according to the presence of oxygen, nitrogen, hydrogen, iron, and carbon in its composition. However, commercially pure titanium has disadvantages such as low wear resistance, difficulty in polishing, and low corrosion resistance. Thus, it was necessary to develop new alloys that improve these disadvantages of pure titanium. Among its alloys are titanium-aluminum-vanadium (Ti-Al-V), titanium-aluminum-niobium (Ti-Al-Nb), titanium-aluminum-iron (Ti-Al-Fe), and nickel-titanium (Ni-Ti). 20
Different modifications can be performed to increase fatigue strength, ductility, elastic modulus, dimensional stability, corrosion resistance, biocompatibility, and osseointegration of the new alloys. These modifications occur in three distinct ways: 1) heat treatment and plasma modification; 2) nanocomposite coating; and 3) carbon-related coatings. 20
The adhesion and maintenance of bacteria may depend on the material used in abutment manufacturing. Microorganisms can invade the interface between implant and abutment and cause bone tissue lesions which are characterized by resorption of the crestal bone and peri-implantitis.21–23
Peri-implantitis has different etiologies, the most common being accumulation of biofilm. However, factors such as periodontal disease history, design, three-dimensional malposition of the implant, the angle of the abutment emergence profile, and the type of connection between the implant and the abutment are of high importance in maintaining peri-implant health. There are three types of connections: platform switched, butt-joint, and no-interface. In butt-joint connections, a bone loss of approximately 1.5 to 2.0 mm may occur. This loss occurs due to the accumulation of pathogenic bacteria present at the interface between the abutment and the implant, which generates an immune response by the host and causes bone loss. The platform switched connection has a lower bone loss compared to other types.20, 24 This is because of biological principles, through the increase in the distance between the implant-abutment interface and the bone surface and the increase in the horizontal dimension of soft tissues, which in combination can protect the bone crest from resorption. 25
Different studies have compared the size of the microgap present at the implant-abutment interface of milled, cast, and printed abutments. Gonzalo et al. compared the space at the implant-abutment interface in milled titanium and printed Co-Cr abutments. 26 The milled ones presented lower measurement of the existing space. However, both presented results for the microgap within the clinically acceptable limit, which consists of values up to 10 μm.27, 28 Corroborating these results, Molinero-Mourelle et al. compared the milled, printed, and “cast” methods and found that only the milled abutments were within the clinically acceptable limit. 29
Thus, the abutment must present adequate mechanical properties so that there is no increase in but maximum reduction of the existing microgap between the implant and the abutment, preventing accumulation of substances, mainly microorganisms that can affect the periodontal health of the patient. The chemical and biological properties of materials are highly important in the prognosis of treatments with dental implants because they influence the adhesion, reduction, or inhibition of pathogenic microorganisms on the surfaces of materials,30–32 and are subject to the corrosion process, mainly due to the presence of acids in the oral cavity with a certain frequency resulting from a citrus diet or even health problems such as esophageal reflux that can contribute to the corrosion and damage of the prosthetic component.33–35
The objective of this work is to identify through a systematic review of the literature the methods of 3D printing and the materials used so far for manufacturing abutments, and to evaluate whether their clinical use is indicated through the mechanical, chemical, and biological analyses carried out in the studies included in the present research.
Material and Methods
Protocol and Registration
This systematic review was structured by Preferred Reporting Items for Systematic Review and Meta-Analyses Protocols (PRISMA) and following their approval was registered in Open Science Framework (osf.io/gzdas).
Eligibility Criteria
The specific criteria of the present review were applied with the PICOS framework: population, abutments; intervention, manufactured by 3D printing/additive manufacturing; comparison, mechanical, biological, and chemical properties; outcomes, possibility of their clinical use; and study design, in vivo or in vitro research with mechanical, chemical, or biological analysis. The main objective of this research was to answer the following question: Do abutments manufactured through 3D printing have mechanical, chemical, and biological properties that allow their clinical use?
The inclusion criteria were articles that contained a mechanical, chemical, or biological analysis of abutments made using 3D printing techniques and articles published in the English language. Articles that studied abutments other than additive manufacturing as well as book chapters, abstracts, letters, and conferences were excluded.
Information and Search Strategy
An electronic search was carried out in the PubMed, Web of Science, Cochrane Library, Science Direct, and Lilac databases. The terms were based on PICOS as follows:
Population #1 – (Abutments [all fields]) AND; Intervention or exposure #2 – ((Additive manufacturing [MeSH]) OR (3D printing [MeSH]) OR (Rapid Prototyping [all fields])) AND;
Comparison #3 – ((Mechanical properties [all fields]) OR (Chemical properties [all fields]) OR (Biological properties [all fields])) AND; Outcome #4 – (3D printed abutment clinical use [all fields]). Filters: Language #5 – (English [lang] Search builder #1 AND 2# AND #3 AND #4 AND #5
These terms were used because they are the keywords present in most articles. A reference management program (Mendeley Desktop) was used to check and delete duplicate references to facilitate reading and final selection of sleeted articles.
Study Selection
The selection of the studies was carried out in two phases: in the first phase, three reviewers independently (M.R.C., T.T.O., and A.L.B.) read the titles and abstracts to identify possible studies to be included. In phase two, after excluding references that did not meet the selection criteria, the articles with the potential to be included in the review were read in full by all authors. To finalize the inclusion, a discussion of all articles from the last phase of the selection was made with a research coordinator (A.C.R.) to finalize the inclusion list.
Data Collection Process
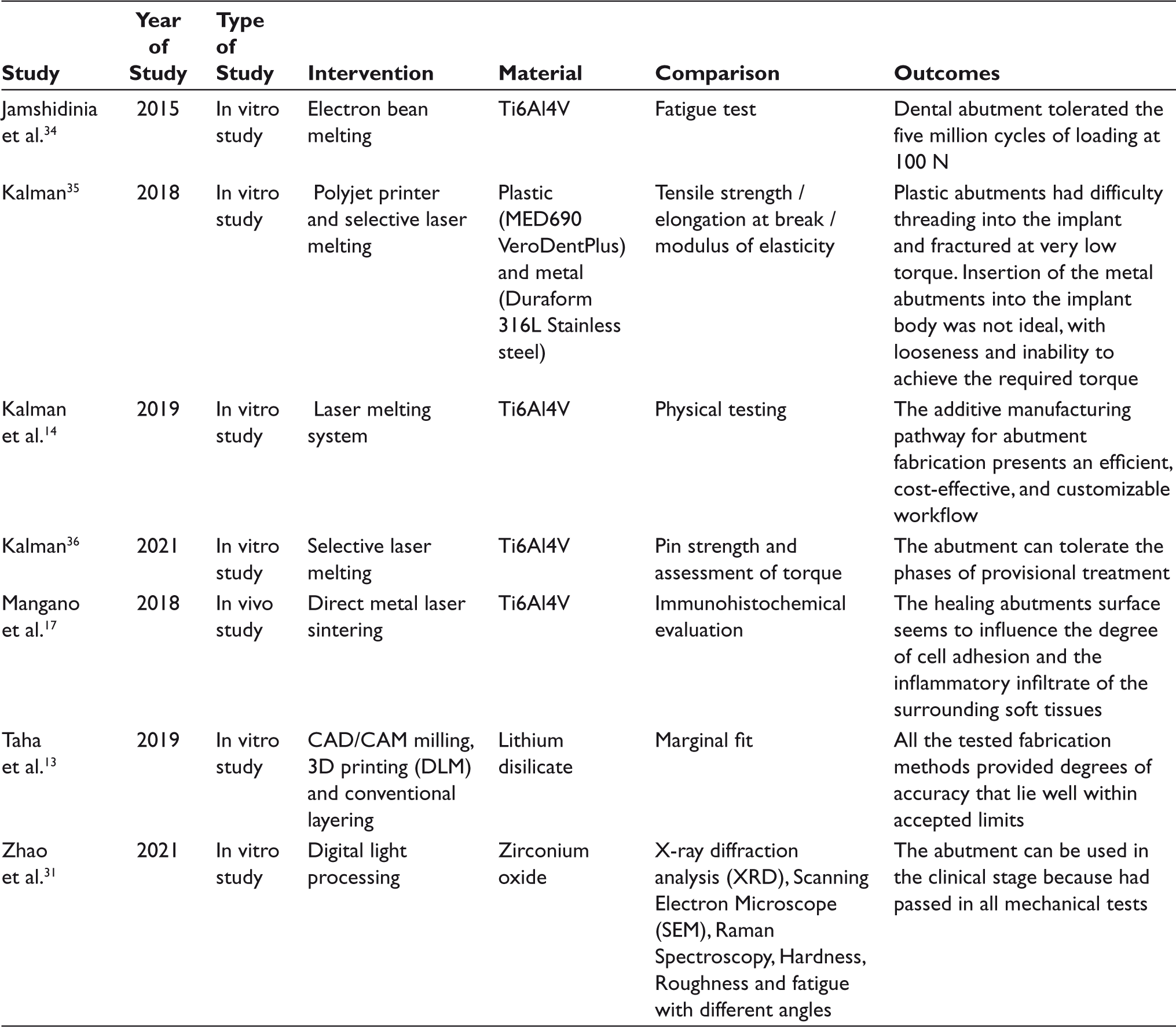
The data extracted from the included articles are shown in Table 1, which include the author(s), the year of publication, the mode of publication, the material used, the analysis performed, and the results found for each article.
The Manufacturing Method, Material, Analysis, and Results of the Selected Articles
Assessment of Risk of Bias
According to Tufanuru et al., 36 the articles were evaluated qualitatively to analyze the risk of bias to provide a more reliable result of the studies. The classification of the risk of bias has three values: low risk, when the article is clear, easy to understand and no bias can change the result; “unclear,” when the study presents a moderate risk when it presents an object of study that is not clear and it is necessary to justify it to evaluate it; and high risk, where the results cannot be obtained faithfully.
Results
Study Selection and Characteristics
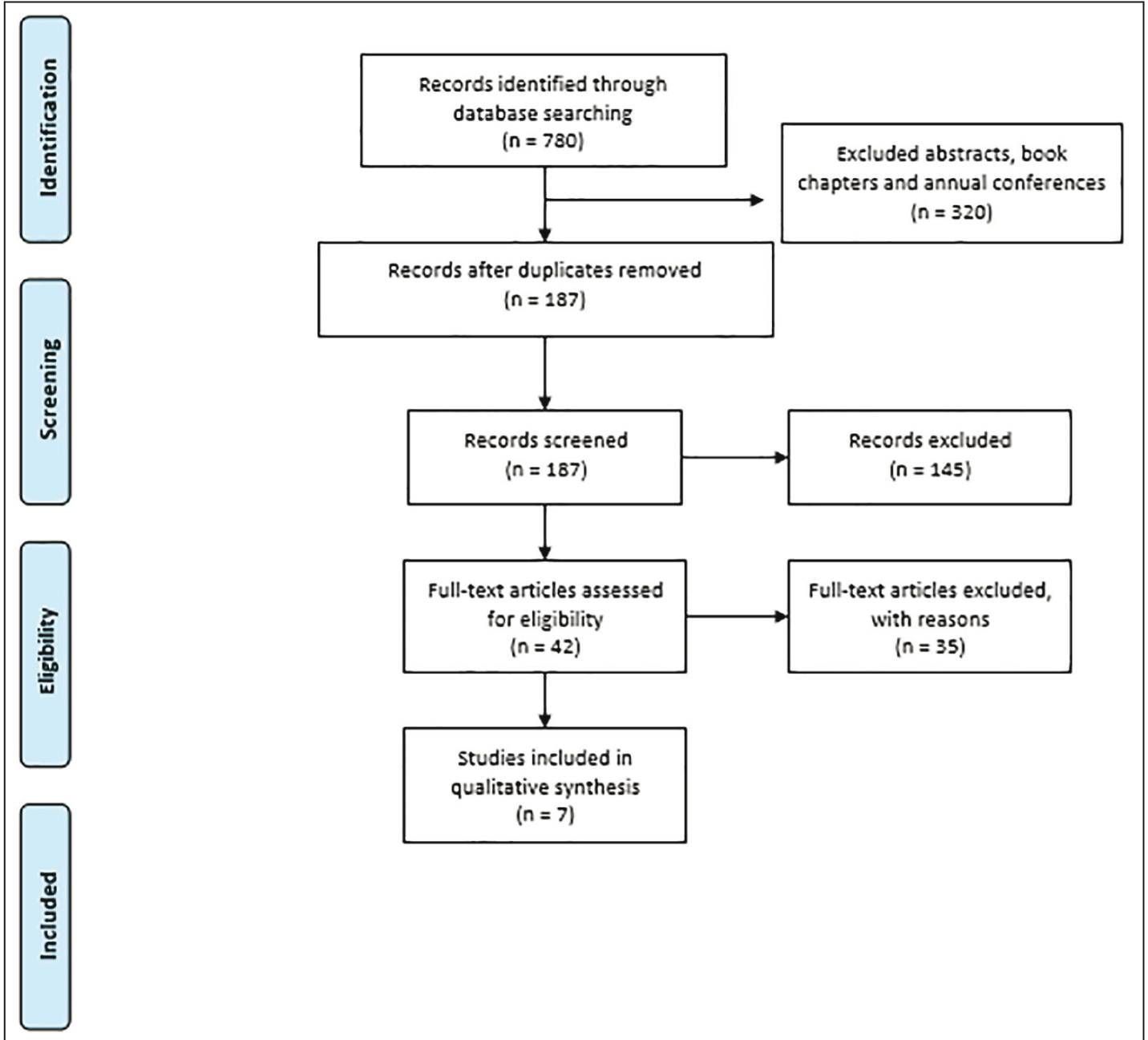
The search for the articles is detailed in the diagrammatic model of PRISMA (Figure 1). We found 780 references from the period between 2015 and 2021. After excluding 320 references that were abstracts, book chapters, and conference papers, 460 remained. Duplicates were then checked and excluded, which left 187 articles. After reading the abstracts, 145 were excluded for not meeting the inclusion criteria, while the remaining 42 articles were read in full. After this reading, 35 articles were further excluded, leaving seven articles for the final inclusion in this review.
Organization Chart of the Selection of Articles in the Databases
Risk of Bias Graph
The Risk of Bias
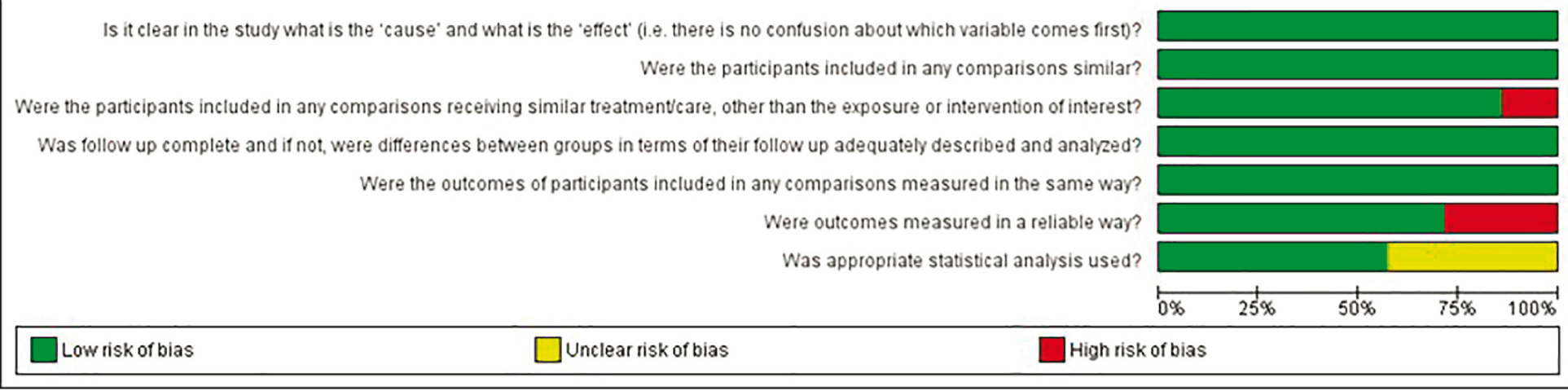
The risk of bias was evaluated according to the JBI checklist quasi-experimental studies methodology to conduct a critical and rigorous assessment of the quality of research included in this review to obtain a more reliable conclusion. In all, seven questions were analyzed based on high risk, unclear and low risk of bias (Figure 2). The questions included:
Is it clear in the study what is the “cause” and what is the “effect”? Were the participants included in any similar comparisons? Were the participants included in any comparisons receiving similar treatment/care, other than the exposure or intervention of interest? Was the follow-up complete, and if not, were differences between groups in terms of their follow-up adequately described and analyzed? Were the outcomes of participants included in any comparisons measured in the same way? Were the outcomes measured reliably? Was an appropriate statistical analysis used?
Of the selected studies, Kalman, 37 Kalman et al., 18 and Mangano et al. 21 presented one question each with a high risk of bias: question 3 for Mangano et al., 21 as the study is an In-vivo research, that is, each participant in the study may have presented different reactions to the same treatment. Kalman et al. 18 and Kalman, 37 presented high risk for question 6 as the measurements were taken manually by only one operator, who may have taken them improperly, using different force application during the insertion of abutments or even when measuring the torques obtained. Three studies18, 37,38 had an “unclear risk” for question 7 as they did not show what statistical analysis was used to reach their results.
Results of Individual Studies
The methods used in the selected studies for printing abutments are Electron Bean Melting (EBM), 39 Polyjet Printer, 37 Selective Laser Melting (SLM),18, 35,39 Direct Metal Laser Sintering (DMLS), 21 and Digital Light Processing (DLP).17, 35
The analyses found in the studies included fatigue test,35, 39 hardness, 35 roughness, 35 functionality to make provisionals, 37 magnification and radiological, 18 assessment of torque, 38 immunohistochemical, 21 image analysis to measure gaps, 17 flexural strengh 35 and characterization with ultra-high Temperature Synchronous Thermal Analyzer (TG/DSC), 35 X-Ray Diffraction (XRD), 35 Scanning Electron Microscopy (SEM) 35 and Raman Spectrum Analyzer. 35
The materials most used for printing the abutments were titanium alloy (Ti6A4V) in four studies,18, 21,38,39 and with one exemplar, plastic (MED690 VeroDentPlus), 37 metal (Duraform 316L stainless steel), 37 lithium disilicate, 17 and zirconium dioxide (ZrO2). 35
The studies showed promising results for the use of printed abutments in clinical settings. Two studies showed that abutments survived the provisional stages.18, 38 The hardness, fatigue tests, and roughness of ZrO2 abutments presented good results that enable its clinical use. 35 The wax pattern printed from lithium disilicate showed accuracy within acceptable limits for the existing microgap space between implant and abutment. 17 The fatigue tests of the octahedral structure made of Ti6A4V alloy demonstrated that they can tolerate five million cycles of loading at 100N. 34 The printed abutments healings showed that surfaces with DMLS had better molecular adherences between the abutments and peri-implant tissue when compared to the machined ones. They also presented fewer inflammatory cells. 21
The results showed that 3D printing using different materials to make abutments is very promising due to the lowest cost to production, the possibility to personalize each abutment, and decrease in the time of production and human resources.
Discussion
Success in oral rehabilitation with implant-supported prosthesis depends among other factors of the correct choice of the abutments, including the microgap formed between implant and abutment, mechanical and chemical properties to support the phases of treatment as well as to support cycles of chewing and the material used.22, 37,39,40
Different materials are used for the fabrication of abutments including polyetherketoneketone (PEKK) and polyetheretherketone (PEEK). 41 PEKK is a high-performance thermoplastic material used in medical setups mainly for its high mechanical strength. 41 PEEK is a semi-crystalline linear polycyclic aromatic thermoplastic polymer and has excellent mechanical properties, thermal stability, and chemical resistance. 42 Its behavior in tissues is important due to its low affinity for the biofilm, which reduces the chance of infections by microorganisms and the inflammatory response by the host. Thus, it can be a viable material for the manufacturing of abutments. When compared with each other in terms of biological behavior, PEKK produces a lower tissue inflammatory response and bacterial adhesion. 41 However, PEEK has a modulus of elasticity close to the human bone (3–4 GPa), which favors its use in implant dentistry. 43 Thus, making abutments with different materials and techniques can be beneficial for the prognosis of treatment with dental implants.
Taha and collaborators 17 demonstrated that wax patterns of printed abutments presented clinically acceptable results of the space existing between these structures with an average of 104.4 ± 13.8 μm. There are no fixed values in the literature regarding two-piece abutments; however, as established by McLean and von Fraunhofer, 44 after examining more than 1,000 crowns, a gap of up to 120 μm between two pieces allow an adequate biological sealing.44, 45 The accuracy of the space present between the implant interfaces and abutment has high importance for the success of treatments as bacteria can enter through these spaces and produce inflammatory reactions, which can lead to bone resorption and gingival recession.23, 46,47
Kalman reported in three studies18, 37,38 the manufacture of abutments with different materials, in which at first using plastic and metal did not reach the mechanical properties and necessary adaptation for manufacture provisional. 37 However, after optimizing the design, using the titanium alloy Ti6Al4V in two other studies,18, 38 was successful in the adaptation and tightening of the abutment, which presented the maximum torque of 49.9 N/cm to the implant for manufacturing provisionals. Torque is very important concerning the gap between the parts. Torques with values below 35 N/cm 3 present weaker contact between the parts, which increases the number of bacteria entering the system, that is, when the torque value increases, the gap and the bacterial seal between the parts decreases, which is more effective. 48
During chewing, there is a frictional contact between the implant-supported prosthesis and the antagonist tooth. This contact can generate oblique compression forces and lead to fatigue of the implant-abutment-crown system.37, 39,49,50 In the molar region, for example, the bite force during masticatory cycles can exceed 500 N, 51 which may demonstrate the disadvantage of the abutment used by Jamshidinia, 29 which only tolerated total load cycles with less than 100 N of bite force. However, this number is equivalent to fatigue with five million cycles, which demonstrates the relationship found by the author, where increased load decreased the abutment’s fatigue time.
The 30° angle also influences the result of fatigue, since oblique forces present 3.5 times greater stress on the prosthetic components than vertical forces. 52 Unfused particles and areas with greater roughness can influence the reduction of fatigue time, as they are more prone to cracks that will become fractures. 53 However, roughness can be a positive factor when the abutment is in contact with the bone, especially if a tapered implant is used, as it can facilitate bone formation around the surface. 54
Mangano et al. 21 report positive results in printed healing abutments, which present less inflammatory infiltrates in the tissues and better adhesion of integrins and hemidesmosomes to the abutment. The cellular response to an abutment is very important since it can be colonized by a biofilm, so a healthy epithelium can protect the adjacent bone and create a barrier when in contact with the abutment. 32 The result of the study by Mangano et al. 21 demonstrates the best cellular response to the fully printed abutment, which favors the use of technology, even though it has a rougher surface, in contrast to the previous studies that argue that smoother surfaces have less bacterial colonization.55, 56
Zhao et al. 35 demonstrated favorable results of printed zirconia abutments after parameter settings. Vickers hardness and roughness values are within the clinically acceptable limit sreported in the literature. Under different pre-angulations too it presented satisfactory results for fatigue. The unfavorable point is that the values of the sides are lower for Vickers hardness and higher for roughness, which may be due to a greater contraction of the material when printed on the Z-axis, compared to the X and Y axes. DSC and SEM were important to define printing parameters, seeking the best quality of the printed material, and thus, favoring the expansion of the possible indication of clinical use of zirconia abutment.
This systematic review presented different studies using different methodologies. However, their purpose was similar—to demonstrate the manufacturing of abutments using 3D printing. The main limitation of this review is the number of studies, which is low due to the technology being relatively new to the dental field and there are still no long-term studies on the subject. Another limitation is that the studies are very different, which makes a comparison between them difficult, and for this reason it was not possible to carry out a meta-analysis with the data found. In addition, for each technique used for printing, different materials and parameters are required for the accurate manufacturing of the abutment and limited details can influence the results.
As for the risk of bias, two studies were at high risk due to the analysis of the results, which were done manually by an operator. This can influence the results. Another fact to be considered is the cost, even if the abutment is low, depending on the printer to be used, it has relatively high values, which limits the use for most dentists in private offices. In any case, studies have shown positive results in which it is possible to see a direction in the manufacturing of printed abutments with good mechanical, chemical, and biological properties, and which can be used clinically. However, more studies are needed to prove their effectiveness in manufacturing these methods to simplify their production and benefit the greatest number of people using this technology.
Conclusions
Even with the limitations of the present research, it is possible to conclude that
Printed abutments have adequate mechanical, chemical, and biological properties that can indicate their clinical usability. The techniques and materials used in the manufacturing of abutments through 3D printing include Electron Bean Melting for the fabrication of Ti6Al4V abutments; Polyjet printer for the fabrication of plastic MED690 VeroDentPlus abutments; Selective Laser Melting for the fabrication of metal Duraform 316L Stainless Steel and Ti6Al4V abutments; Direct Metal Laser Sintering for the fabrication of Ti6Al4V abutments; Digital Light Processing for zirconium oxide abutments; and CAD/CAM milling and 3D printing for the fabrication of lithium disilicate abutments. 3D printing has high accuracy and speed and it can produce customized abutments according to each case. More studies are needed to prove the effectiveness and durability of printed abutments as well as to define the parameters for different materials that can be repeated in other studies and seek large-scale production, so the use of this technology can benefit both the dentist and the patient.
Footnotes
Acknowledgements
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Statement of Informed Consent and Ethical approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
