Abstract
Aim:
The aim of the current study was to isolate stem cells from various dental sources such as dental pulp, periodontal ligament (PDL), and apical papilla, and to characterize stem cells by staining for the presence/absence of specific surface markers and also to differentiate stem cells into osteogenic, chondrogenic, and adipogenic cell lineages by exposing them to specific growth factors under the ideal conditions.
Materials and Methods:
A total of 117 samples were included in the study, consisting of 30 pulp, 50 gingival, 35 PDL, and 2 apical papilla samples. The pulp was extirpated and transported to the Central Research Laboratory. Gingival connective tissue was collected from the participants undergoing any crown lengthening procedure or any gingivectomy procedure from the Department of Periodontology. A similar procedure was also followed for apical papilla and PDL. Isolation was done followed by the identification of the cells by immunocytochemistry using different markers. Once the identity of cells was confirmed, these cells were treated with different culture media to attain 70% to 100% confluency. Then the medium was replaced with a conditioning medium containing specific growth factors for differentiation into osteogenic, chondrogenic, and adipogenic cell lineages.
Result:
In our study, the number of samples collected and processed was 117. The isolation rate of stem cells from the above-collected samples was 70%.
Statistical analysis—no statistical analysis was done as there was no variability expected.
Conclusion:
Our study showed that stem cells could be isolated, differentiated, and characterized from different dental sources.
Introduction
The ultimate goal of periodontal therapy is to achieve periodontal regeneration with all the components of the periodontium which include periodontal ligament (PDL), gingival connective tissue, cementum, and alveolar bone. Many techniques and procedures have been investigated and documented in the literature to try and promote periodontal regeneration. These include guided tissue regeneration, with the introduction of alloplastic materials, autografts, allografts, and xenografts. Unfortunately, all of them have several limitations and are not entirely satisfactory. As a result, current research trends have been developed toward cell-based therapies for periodontal tissue regeneration, which mainly involves the use of mesenchymal stem cells (MSCs).
Stem cells are generally defined as clonogenic cells capable of both self-renewal and multilineage differentiation. Postnatal stem cells have been isolated from various tissues such as bone marrow (BM-MSCs), adipose tissues, umbilical cords, Wharton’s jelly, synovial tissues, and other tissues,1–3 including different dental sources. 4
However, stem cells from different dental sources are highly heterogeneous and differ from each other as well as from BM-MSCs morphologically, histochemically, and in their proliferation potential. Hence, it becomes imperative to investigate which source would be ideal to obtain stem cells.
There are hardly any studies and none from India that have simultaneously studied stem cells from different dental sources for their characterization and proliferative potential. Hence, the main objective of our study was to isolate, characterize, and differentiate stem cells from various dental sources.
Materials and Methods
Sample Collection
A total of 117 samples were included in the study, consisting of 30 pulp samples, 50 gingival samples, 35 PDL samples, and 2 apical papilla samples. The age range considered for the study was between 6 and 24 years. Patients were selected from the Outpatient Department of the institute. The inclusion criteria of the study were teeth which were extracted for an orthodontic treatment atraumatically, deciduous teeth which were about to exfoliate and have no carious lesions, deciduous teeth wherein three-fourth of the root was resorbed, and third molars which were not carious and not periodontally compromised. Exclusion criteria for both groups were teeth which were extracted from patients below 6 years or above 24 years of age, deciduous teeth with carious lesions or any other pulpal pathology, deciduous/permanent teeth from medically compromised patients, and third molars with lesions, cysts, or which were periodontally compromised. Subjects who fulfilled the inclusion criteria were enrolled for the study after obtaining their informed written consent.
Collection of Tissue
Dental pulp was obtained from the teeth extracted for the orthodontic treatment and from deciduous teeth. An access cavity was prepared with the help of a sterile aerator and a round bur. The pulp was then carefully extirpated from the pulp chamber using a file/reamer. The extirpated pulp was then transported to the Central Research Laboratory in a sterile vial containing Dulbecco’s modified Eagle’s medium (DMEM) with antibiotics (streptomycin + penicillin + gentamicin) for further analysis. The extracted tooth after the pulp extirpation also was transported to the Central Research Laboratory for periodontal tissue analysis.
In the laboratory, the tooth was thoroughly washed in phosphate buffered saline (PBS) containing antibiotics (streptomycin + penicillin) and betadine; PDL was then carefully scraped off from the tooth in a sterile petri dish with all aseptic precautions. 5 Similarly, apical papilla was collected from the normal impacted third molars with immature roots. Gingival connective tissue was collected from the participants undergoing any crown lengthening procedure or any gingivectomy procedure from the Department of Periodontology. The gingival sample was de-epithelialized with a no. 15 scalpel blade, leaving only the connective tissue which was transferred to a sterile container after a thorough wash in PBS with antibiotics and betadine. A similar procedure was also followed for apical papilla and PDL.6,7
In addition to the above tissues, we also checked the isolation, characterization, and differentiation of stem cells from granulation tissue (healthy and diseased). The healthy granulation tissue was obtained from the patients undergoing a crown lengthening procedure and the diseased granulation tissue was obtained from the patients undergoing a flap surgery procedure. In both the collection methods, sterile curettes were used. The granulation tissue was collected from the deepest site with the curette and then transferred in the transport medium to the Central Research Laboratory for further investigation.
Isolation of Mesenchymal Stem Cells
Each tissue was divided into two portions: one was used for the explant method of growth and the other portion was used for the enzymatic digestion and then followed by the isolation of stem cells.
Explant Method
In this method, the tissue was minced into small pieces in a sterile petri dish, mixed with alpha minimum essential medium (α-MEM), transferred to a 12-well culture plate, and incubated at 37°C in a CO2 incubator for the isolation of stem cells (Figure 1a).
Enzymatic Method
In this method, a portion of the tissue was treated with 3 mg/mL collagenase type I and 4 mg/mL dispase for one hour at 37°C for stem cell isolation. The enzymes were then neutralized by adding DMEM enriched with 10% fetal bovine serum (FBS) and then filtered using a 70-µm cell strainer. The separated cells were mixed with α--MEM and transferred to a 12-well plate. Each sample was incubated for at least 15 days to observe for stem cell growth with media change twice a week.8,9 The tissues that showed the presence of fibroblast-like cells that were adherent to the plastic surface were taken up for further processing (Figure 1b).
Photographs Showing Stem Cells Obtained by (a) Explant Method and (b) Enzymatic Method

Identification of Mesenchymal Stem Cells
The identity of the cells was confirmed by collecting a portion of the cells and staining them for surface markers by immunocytochemistry. The markers that were used for testing included: CD90, CD73, CD105, CD146, and CD45. The manufacturer’s instructions were strictly adhered to during the entire procedure.
Immunocytochemistry Procedure
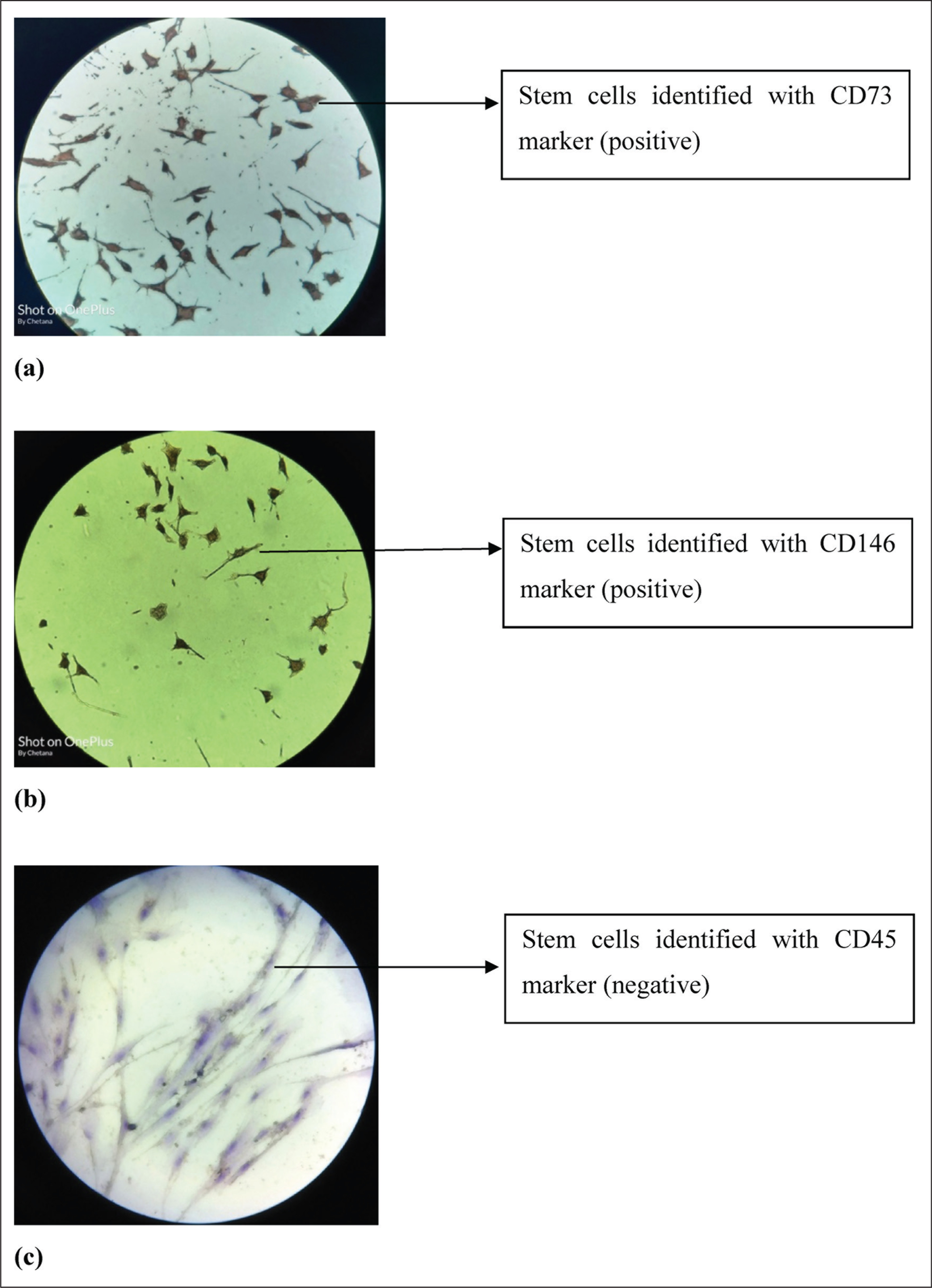
Cells of passage P3 were subcultured into two chamber slides. The cells were fixed in 4% paraformaldehyde for 30 to 45 min and then blocked with a blocking solution (immunocytochemical analysis kit) and incubated with primary antibodies (CD90, CD105, CD73, CD146, and CD45). A further procedure was carried out according to manufacturer’s instructions (Figures 2a, 2b and 2c).
Photographs Showing Identification of Stem Cells With Various Markers. (a) Stem Cells Identified With CD73 Marker (Positive), (b) Stem Cells Identified With CD146 Marker (Positive), and (c) Stem Cells Identified With CD45 Marker (Negative)
Cellular Differentiation
Once the identity of cells was confirmed, the MSCs were tested with different culture media for their efficiency to support growth. After the cells grew to 70% to 100% confluency, the medium was replaced with a conditioning medium containing specific growth factors for differentiation into osteogenic, chondrogenic, and adipogenic cell lineages. 10
Adipogenic, Osteogenic, and Chondrogenic Differentiation
The induction media for osteogenic, adipogenic, and chondrogenic assays were prepared. Briefly, osteogenic media 11 consisted of α-MEM with 15% FBS, 1% (v/v) penicillin/streptomycin solution, 10 mM β-glycerophosphate, and 250 µm L-ascorbic acid. Adipogenic media 12 consisted of α-MEM with 100 µL of 0.1 mM dexamethasone, 1 mL of 45 mM 3-isobutyl-1-methylxanthine, 10 µL of 10 mg/mL insulin, and 50 mM indomethacin. Chondrogenic media 13 consisted of α-MEM with glucose, 10-7 M dexamethasone, 1 µM L-ascorbic acid, 1% sodium pyruvate, and 10 ng/mL transforming growth factor-β1. The cells of passage P5 were plated in 6-well plates at a density of 1×105 cells/well and cultured in a basal medium until subconfluence. The medium was then changed to osteo-induction and adipo-induction medium. The osteo- and adipo-induction media were refreshed every three days. To determine the ability of cells to form lipid droplets (adipogenesis) or mineralized nodules (osteogenesis), the cells were induced for three weeks for adipogenesis or four weeks for osteogenesis.
Staining Procedure
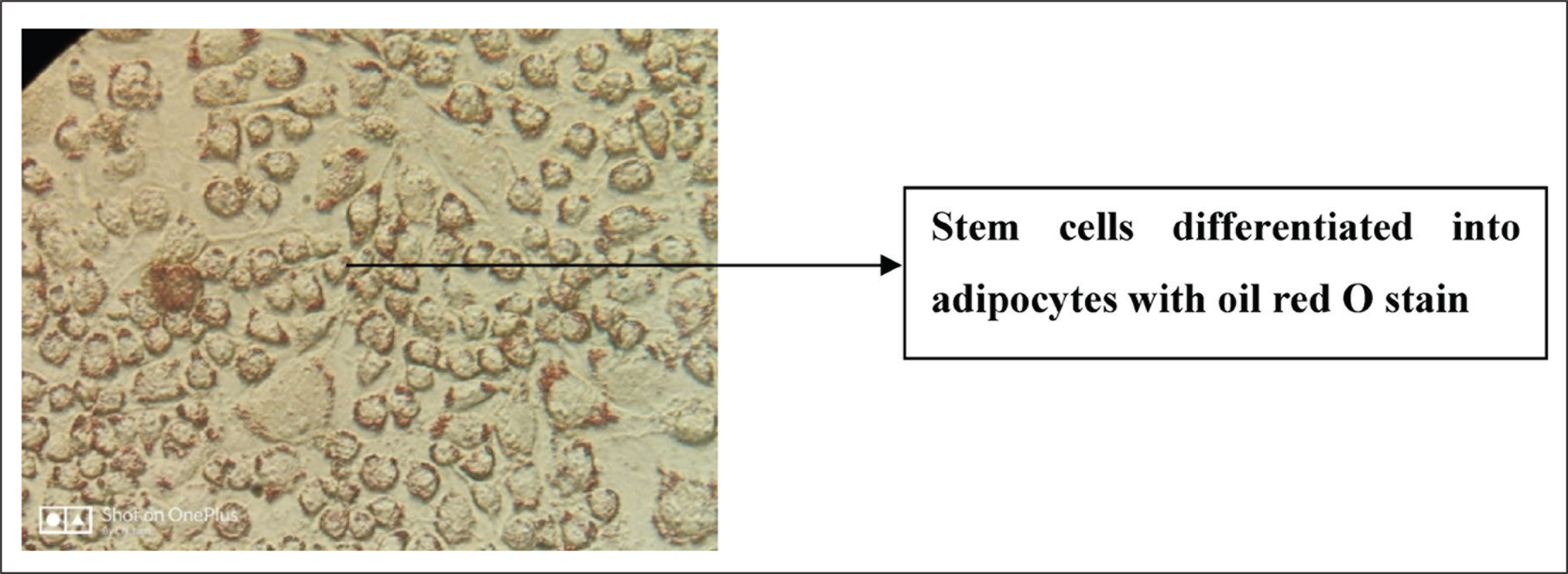
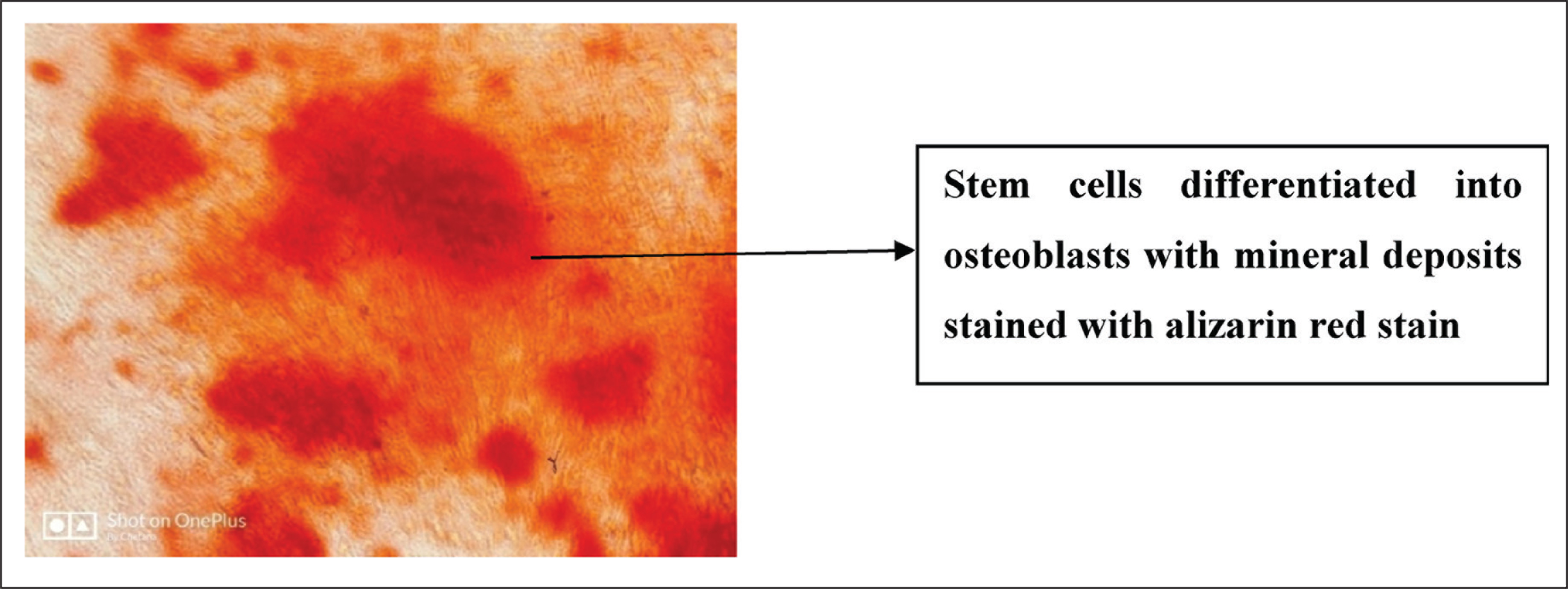
Briefly after adipo-induction, the cells were fixed with 4% paraformaldehyde for 30 min at room temperature and stained with 0.3% oil red O staining for 20 min, and the lipid droplets were then imaged under an inverted microscope. Alternatively, after four weeks of osteogenic induction, the cells were fixed with 4% paraformaldehyde and stained with 0.1% alizarin red S at pH 8 for 30 min and then the mineralized nodules were imaged.
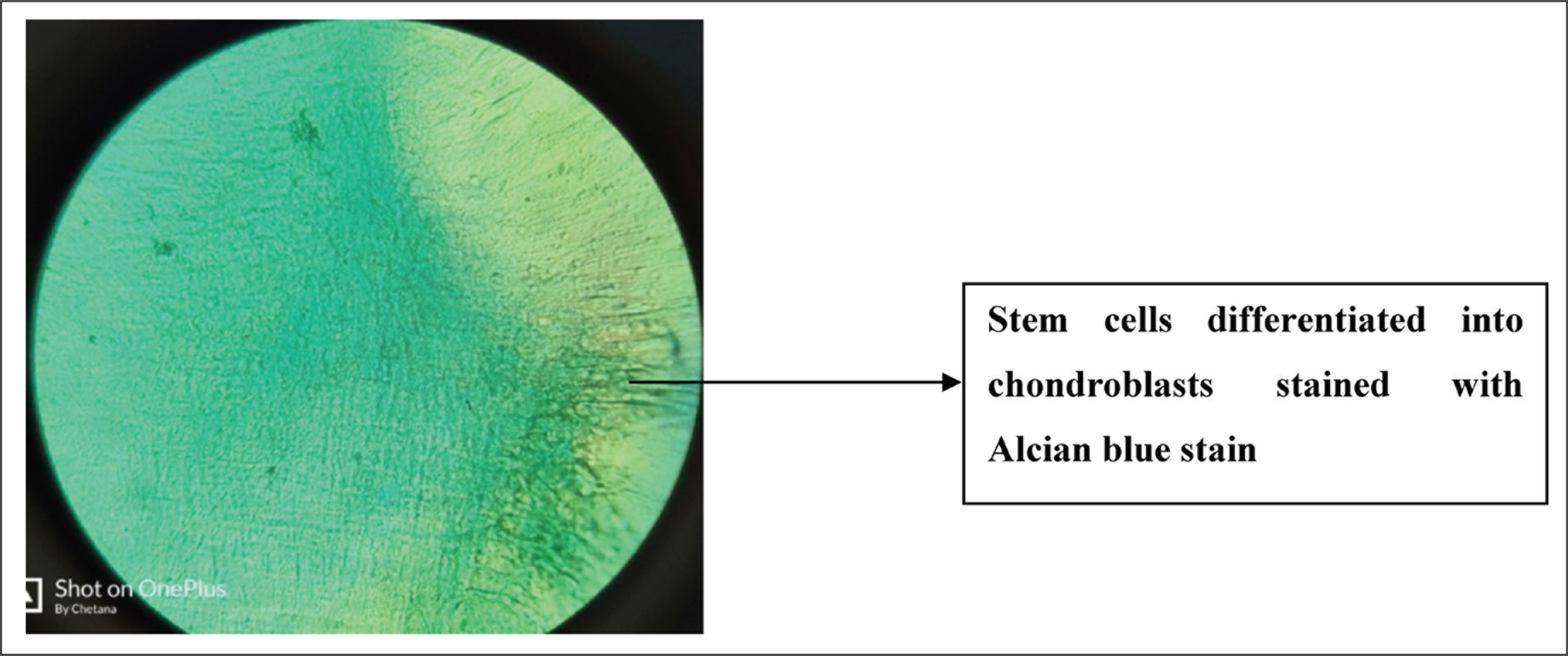
Alcian blue staining is used for chondrogenic differentiation. Cultures were rinsed twice with PBS and incubated with chondrogenic media for three weeks and fixed with methanol for 30 min at –20°C, then washed with distilled water, and covered with Alcian blue at pH 0.2 (0.5% Alcian blue 8 GS in 1N HCL). After overnight staining, cultures were washed extensively with distilled water and imaged (Figures 3, 4 and 5).
Photograph Showing Stem Cells Differentiated into Adipocytes With Oil Red O Stain
Photograph Showing Stem Cells Differentiated into Osteoblasts With Mineral Deposits Stained With Alizarin Red Stain
Photograph Showing Stem Cells Differentiated into Chondroblasts Stained With Alcian Blue Stain
Statistical Analysis
There is no statistical analysis possible in the study as there is no quantification of the stem cells generated, there are no quantitative comparisons of the groups, and there is no variability expected because it is only observational (isolated/not isolated).
Results
In the current study of isolation, characterization, and differentiation of stem cells, the total number of samples collected and processed was 117. The isolation rate of stem cells from the above-collected samples in our study was 70%.
In our study, 20 samples were processed with both the enzymatic method and explant method. We observed that the growth rate of stem cells was quicker when the sample was processed with the enzymatic method, i.e., usually it was three to four days. However, in the explant method the stem cells’ growth rate was slower, i.e., usually took eight to ten days for the cells to grow. We also observed that the proliferation rate of stem cells was better and faster with the explant method as shown by passaging. At the same time, the cells obtained from the explant method were of much pure breed and had a longer life span as compared to the cells obtained from the enzymatic method.
It was also observed that in lineage differentiation, some dental pulp samples showed stem cells with typical neuronal morphology. 14 Our study showed that there was no much difference in the differentiation capacity of stem cells isolated from different sources. In addition to the aforementioned sources for collection, we also collected 20 samples of granulation tissue from both healthy (10) and diseased subjects (10). We found that the differentiation potential of stem cells into osteogenic, adipogenic, and chondrogenic cells was higher for the healthy group subjects when compared to the diseased group.
Our study showed that there was no much difference in the differentiation capacity of stem cells isolated from different sources and these can be further used for various regeneration purposes in the field of dentistry especially periodontology.
Discussion
Tissue engineering is a promising discipline with many advantages as a regenerative therapy. Accessibility to the tissues for the derivation and culturing of stem cells with minimal complications for the donor is of importance. 15 Hence, in our study, we made an attempt to isolate, characterize, and differentiate stem cells from various dental sources. In the present study, we were able to isolate MSCs from the human gingival connective tissue, which showed characteristics similar to those typically described for BM-MSCs, which is in accordance with Mitrano et al., 15 who isolated stem cells only from the gingival tissue. This is also in accordance with the study conducted by Jin et al. 16
In our study, we could also isolate MSCs from the dental pulp of both primary and permanent teeth, which is in accordance with Naz et al. 14 We could also isolate MSCs from PDL, which is in accordance with Khoshhal et al. 17 We first isolated a cell population by its plastic-adherent property, as described by Friedenstein et al. 18 Our results indicate that gingival connective tissue-derived MSCs are CD90+, CD105+, and CD45−, which is in accordance with Mitrano et al., who had similar results. 15 We were able to induce osteogenic, chondrogenic, and adipogenic differentiation of stem cells under specific differentiation media, which is in agreement with Pittenger et al. 19 and Nöth et al., 20 who described the multilineage potential of adult BM-MSCs.
In our study, we processed the tissue for derivation by both the enzymatic method and explant method. We found that the growth rate of stem cells was quicker when the sample was processed with the enzymatic method, i.e., it was usually three to four days. But, in case of the explant method, the stem cells’ growth rate was slower, i.e., it usually took eight to ten days for the cells to grow. We also observed that the proliferation rate of stem cells was better and faster with the explant method as shown by passaging. At the same time, the cells obtained from the explant method were of much pure breed and had a longer life span as compared to those obtained from the enzymatic method.
Stem cells have received increasing attention in tissue engineering, but many difficulties lie in their use as invasive procedures such as bone surgery or liposuction may be required to obtain these cells from the bone marrow or fatty tissues. Stem cells from the umbilical cord can also be obtained but only at a certain stage. In contrast, MSCs from gingiva and other dental sources can be isolated, characterized, and differentiated by minimally invasive procedures at any time in life and can be a promising alternative source of stem cells. Hence, to conclude, we could isolate, characterize, and differentiate stem cells from various dental sources with minimally invasive procedures. Further studies are needed to use stem cells as 3D stem cell sheets and use the same for periodontal regeneration.
In the current study, we could have only two apical papilla samples as the method to collect apical papilla tissue is technique sensitive and the tissue is not readily and easily available.
Conclusion
In conclusion, we isolated, characterized, and differentiated stem cells from various dental sources. We also have optimized the protocol keeping in mind the financial constraints of individual investigators.
Footnotes
Acknowledgements
This research work was supported by Research Grants from the Rajiv Gandhi University of Health Sciences (RGUHS), Bangalore. We thank our colleagues from the Central Research Laboratory at the Maratha Mandal Institute of Dental Sciences and Research Centre, Belgaum, who provided insights and expertise that greatly assisted us in carrying this research. We thank Mr Vijay Kumbhar for his assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
The study was commenced after obtaining the ethical clearance from the Institutional Ethical Review Board of Maratha Mandal’s Nathajirao G. Halgekar Institute of Dental Sciences and Research Centre with the approval number 536 and dated 12/7/2016.
All the procedures have been performed as per the ethical guidelines laid down by Declaration of Helsinki (1964).
Funding
This work was supported by the Research Grants from the Rajiv Gandhi University of Health Sciences (RGUHS): 16D026.
