Abstract
Background:
Keratocystic odontogenic tumor (KCOT) is an odontogenic lesion which manifests distinct biological behavior. Predominant studies in KCOT attribute this behavior to high epithelial proliferative capacity. Besides, a few studies facet loosely arranged collagen can contribute to the behavior of KCOT. Matrix metalloproteinases (MMP) are enzymes that degrade extracellular matrix components under both physiologic and pathologic conditions. The loosely arranged collagen in connective tissue wall of KCOT could be related to the degree of MMP-9 expression.
Aim:
To evaluate the arrangement of collagen fibers along with immunoexpression of MMP-9 and to relate to its neoplastic biologic behavior in sporadic cases of KCOT.
Materials and Methods:
KCOT (n = 23) and dentigerous cyst (DC) (n = 15) samples were processed for the following techniques: Masson’s trichrome stain for light microscopy, PMA-PSR stain for confocal microscopy, and MMP-9 for immunohistochemistry.
Results:
In Masson’s trichrome analysis, correlation of collagen fiber arrangement in the deeper regions with color intensity for KCOT was found to be statistically significant (P = .033). In confocal microscopy, there was no difference between intensities in KCOT (P = .990) and DC (P = .233), respectively. The immunoexpression of MMP-9 in the connective tissue wall of DC (73.3%) was relatively higher than that of KCOT (60.8%). However, on comparison between KCOT and DC in the presence of inflammation, the immunoexpression of MMP-9 was higher in DC (100%) than KCOT (69.9%) and was statistically significant (P = .028).
Conclusion:
It was concluded that the loose connective tissue wall in KCOT is because of the inherent nature of the lesion that could facilitate its biologic behavior. If inflammation is present, this could further aggravate the tumorigenic behavior.
Keywords
Introduction
Odontogenesis is a highly coordinated and complex process which depends upon epithelial mesenchymal interactions that result in the initiation and formation of the tooth. 1 Following odontogenesis, the odontogenic epithelium degenerates and forms epithelial remnants that may get triggered by several mechanisms to form pathological entities. Lesions of odontogenic origin comprise of a heterogeneous group ranging from hamartomatous proliferations, cysts to benign, and malignant tumors. 2 Numerous studies have been done to classify these lesions, which define the clinico-pathological criteria necessary to diagnose them. With the development of newer technologies in diagnosis such as special stains, immunohistochemistry, molecular biology and genetics, along with clinical and epidemiological follow-up, some of the lesions had been re-classified according to their biological behavior.1,3
Odontogenic keratocyst (OKC) is one such highly controversial lesion with respect to its histopathologic features and clinical behavior. The OKC in 2005 was designated by the World Health Organization as a keratocystic odontogenic tumor (KCOT) and is defined as “a benign uni- or multicystic, intraosseous tumor of odontogenic origin, with a characteristic lining of parakeratinized stratified squamous epithelium and potential for aggressive, infiltrative behavior” and it has a high rate of recurrence, especially compared to other odontogenic cysts even in its sporadic form. 4
Interplay between the epithelium and connective tissue is assumed to play a significant role in the pathogenesis of odontogenic cysts. Despite the fact that proliferation of epithelial cells is an indispensable part of cyst formation, connective tissue may be regarded as a functional part of cyst and not just a structural support. 5 Experimental studies have shown that the transplanted epithelium formed new cysts only if supported by its own stroma. 6 Thus, the mesenchymal influence of the fibrous capsules may play an important role in the maintenance of epithelial expression of KCOT. Collagen which constitutes 34% of the total extracellular matrix (ECM) proteins forms an integral part of connective tissue stroma maintaining structural integrity and determining tissue function. In pathological conditions, collagen can show variations in the way the individual fibrils are organized into fibers and their diameter. 7 Therefore, the analyses of the organization of collagen fibers in KCOT may pave way in understanding aggressive behavior. Connective tissue stains such as Van Gieson, Masson’s trichrome (MT), and Gomori stain have been used for light microscopy, picrosirius stain in polarizing microscopy, and phosphomolybdic acid-picrosirius red (PMA-PSR) stain for confocal microscopy. MT is a connective tissue stain which basically helps in differentiating collagen, muscle, and fibrin. The collagen fibers are colored blue distinctly, while the cytoplasm, muscle, and fibrin appear red. Confocal scanning microscopy is used by researchers in biological science to obtain an image of the three-dimensional architecture of a specimen, such as a cell or tissue. PMA-PSR staining methodology in recent times had been used to study the arrangement and distribution of collagen in odontogenic entities. 8
Matrix metalloproteinases (MMP) comprise a family of calcium- and zinc-dependent enzymes that are able to degrade ECM components under both physiologic and pathologic conditions.9–11 Studies suggest that enzymatic degradation of the bone matrix and basement membrane (BM) by MMP-9 is involved in the expansion of odontogenic cysts.12–14 The loosely arranged collagen in the connective tissue wall of KCOT can be highly related to the degree of MMP-9 expression. Hence, this study evaluates the neoplastic potential in the connective tissue wall of KCOT by studying the arrangement of collagen fibers and the expression of MMP-9 in order to pertain to the aggressive biological behavior.
Materials and Methods
The study material consisted of 38 formalin fixed paraffin embedded tissue samples of KCOT (23 cases) and dentigerous cyst (15 cases). The cases were retrieved from the archives of the Department of Oral Pathology, Saveetha University, from 2000 to 2013. The specimens selected were histopathologically confirmed cases of KCOT (n = 23) and dentigerous cyst (n = 15) stained with hematoxylin and eosin. The exclusion criteria were syndromic cases of KCOT and orthokeratinized variant of KCOT.
Masson’s Trichrome Staining Analysis in Light Microscopy
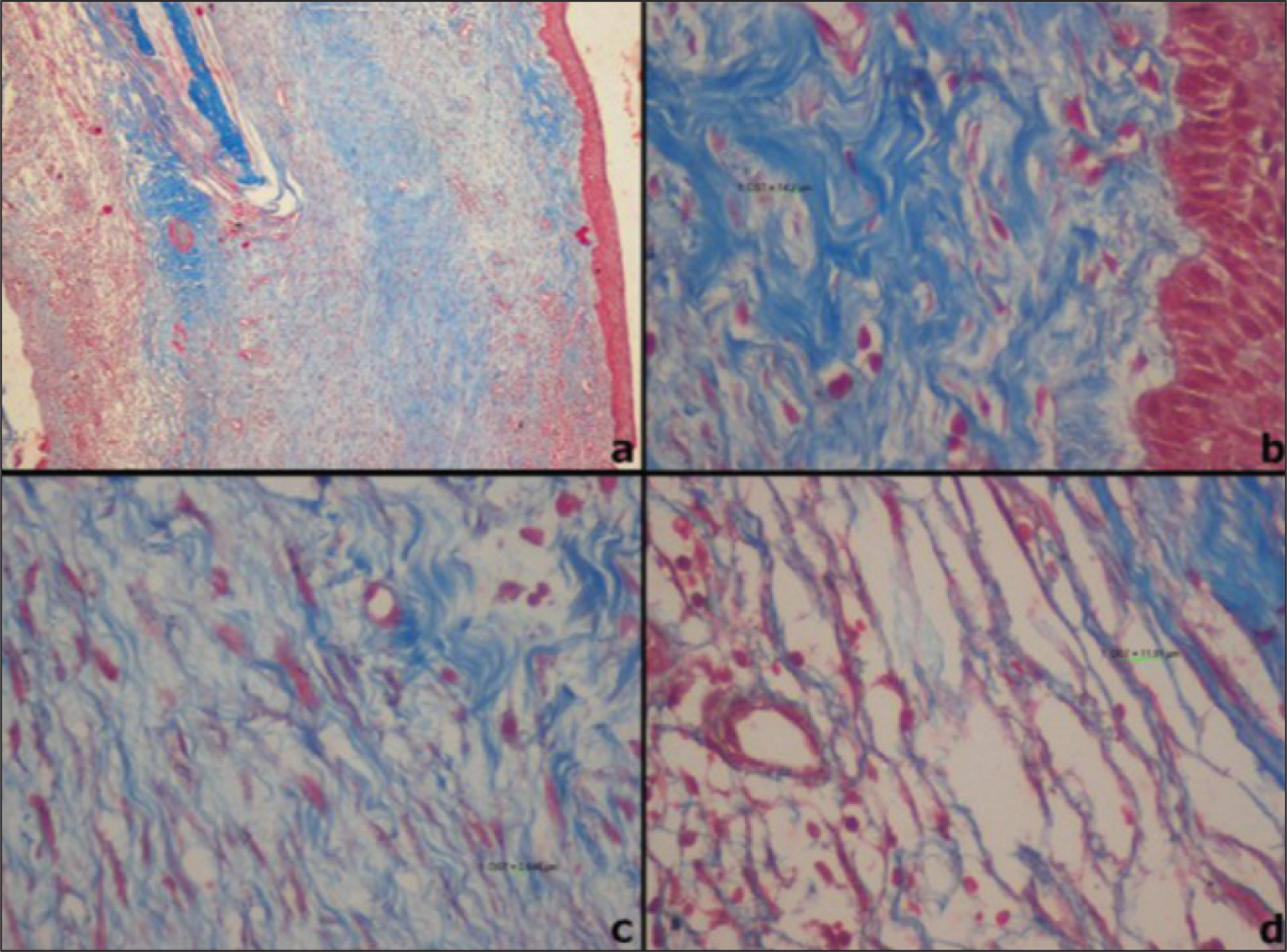
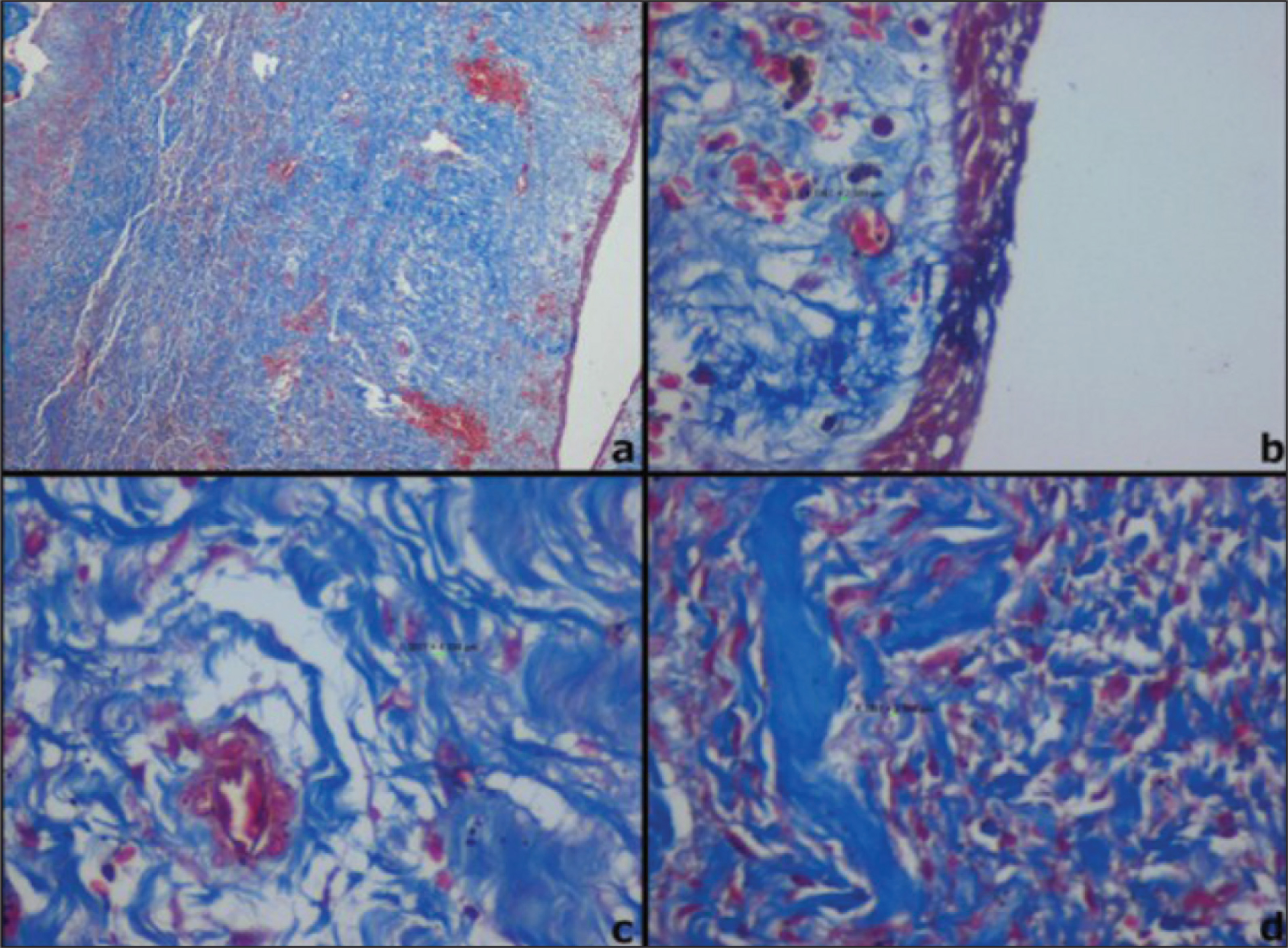
Sections of 3-micron thickness made using Leica semiautomated microtome were subjected to MT stain procured from Sigma Aldrich, USA (Catalogue no HT 15) and exhibited collagen fibers blue. Sections were viewed under light microscopy on dividing the connective tissue wall into three regions such as subepithelial, middle third, and deep. Each field was viewed under 40× magnifications along with recording of corresponding pictures for analysis using Image J software. The following parameters that were observed are average distance between collagen fibers, arrangement of collagen fibers, color intensity staining, and inflammation. Images of the three representative fields obtained were subjected to Image J software to measure the average distance between the fibers for both groups (Figures 1 and 2). The average mean values obtained ranged from 0.003 to 0.031 µm. The arrangement of fibers for the three fields were graded as loose (≥0.015µm) and dense (<0.015µm) based on the average mean values obtained by the distance between collagen fibers. The color intensity of MT staining in the viewed fields was graded as bluish tinge light blue, blue, and dark blue. The amount of inflammation present was classified as absent, minimal, mild, moderate, and severe. The semiquantitative analysis of MT-stained specimens was compared between two groups based on the arrangement of collagen fibers versus the color intensity and inflammation by chi square test using SPSS software version 15.0 with the significance level set at 5% (P < .05).
Phosphomolybdic Acid-Picrosirius Red Staining
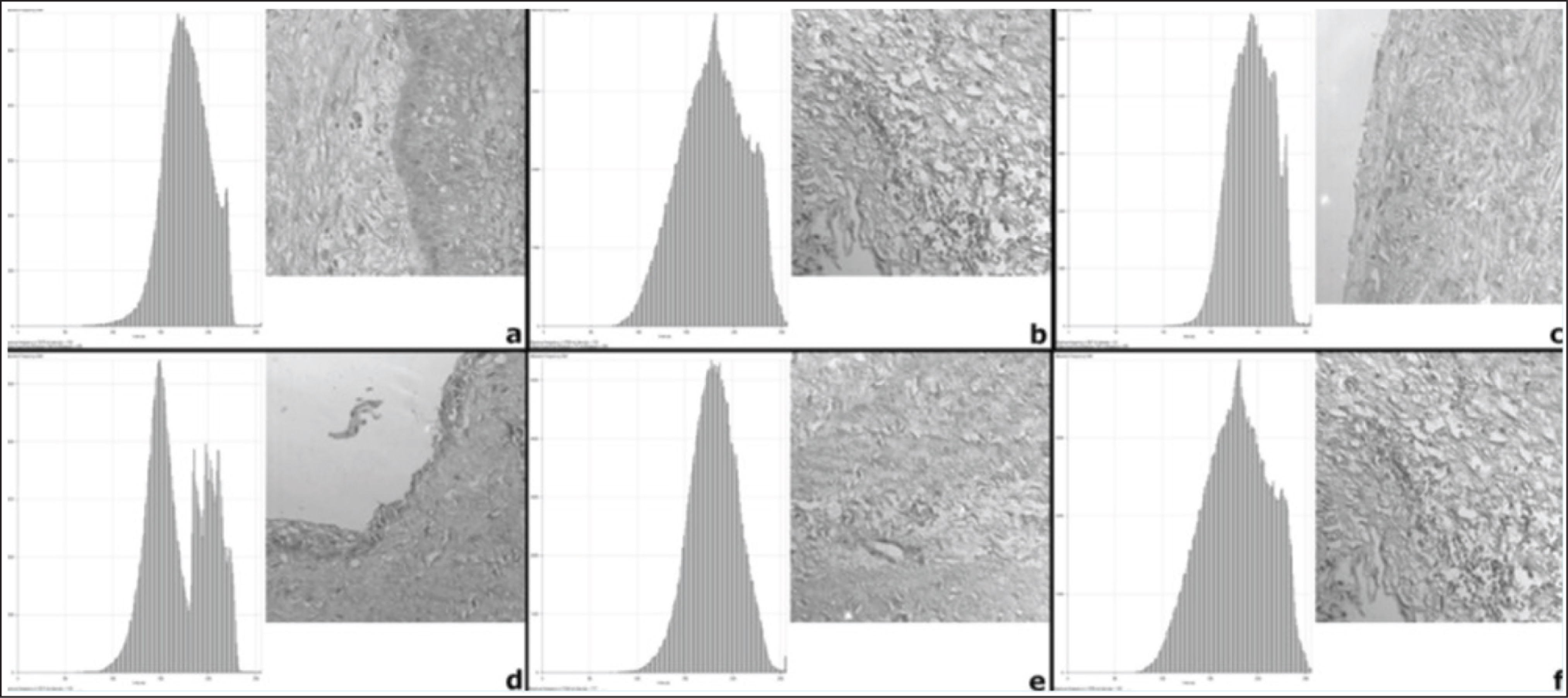
Sections of 3-micron thickness made using Leica semiautomated microtome stained with PMA-PSR demonstrated collagen fibers in red color in a pale yellow background. These sections were viewed under confocal microscopy (Zeiss LSM 510 META confocal laser scanning microscope [Zeiss, Germany] with HeNe laser [543 nanometers]) in the same three fields observed under light microscopy. The confocal images were obtained for the subepithelial, middle third, and deep areas of the connective tissue wall at 20× magnification. All images were recorded by +LSM Image Browser software, version 3.5 (Zeiss, Germany), and configured to show collagen fibers in white and gray tones, whereas interstitial space and epithelial tissue were displayed as black areas. The images were then produced as converted histograms based on black, grey, and white pixels (Figure 3). Each image quantified the intensity of the connective tissue wall based on the pixels obtained. The statistical analysis was performed comparing the arrangement of collagen fibers seen in light microscopy and the intensity of collagen fibers seen in confocal microscopy using Levene’s test using SPSS software version 15.0 with significance level set at 5% (P < .05).
Immunohistochemistry
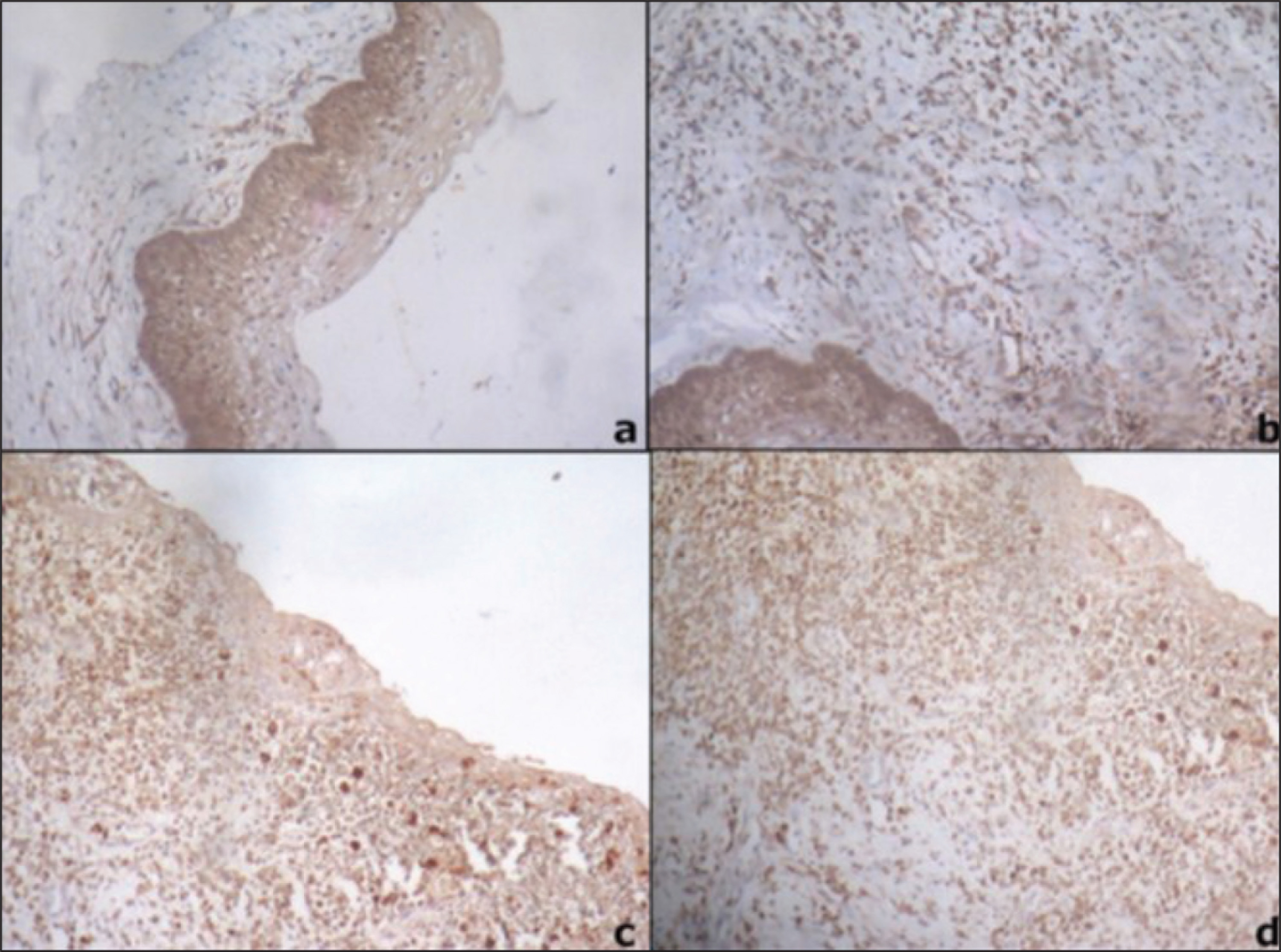
Immunohistochemical analysis was performed on 3-micron paraffin embedded tissue sections on gelatin-coated glass slides. After heat drying, sections were deparaffinized in xylene and subsequently rehydrated in gradients of ethanol. Antigen retrieval to unmask antigenic sites was done using 10 mM citric acid solution (pH 6) followed by a washing step with tris buffered saline (TBS, pH 7.6). Further incubations with pre-diluted ready-to-use primary mouse monoclonal antibody MMP-9 (Biogenex EP 1255Y, USA) was performed at 37°C for 30 min. Sections were washed again and incubated with biotinylated secondary antibody mouse IgG antibody (Novolink min polymer detection system (Leica, Germany) for 30 min followed by the streptavidin-biotin-peroxidase for 30 min at room temperature. Colored reactions were developed by incubating with 3’3’-diaminobenzidine (0.05 diaminobenzidine in 0.05 M Tris buffer, pH 7.6, and 0.01 H2O2) and subsequently counterstained with Harris hematoxylin and mounted with DPX (Distyrene Plasticizer Xylene). Breast carcinoma was used as a positive control. The negative control was prepared by omitting the primary antibody and incubated instead with a negative control reagent. This tissue demonstrates the absence of specific staining. The presence of brown colored reaction localized to the nucleus or cytoplasm was considered as a positive reaction. Immunoexpression of MMP-9 observed has been recorded in terms of intensity and proportionality score in the epithelium and connective tissue of KCOT and dentigerous cyst (Figure 4). The scoring of MMP-9 was completed by two interobservers at different times and each examiner interpreted the findings using the scoring method shown in Table 1.
Scoring Method of Immunoexpression of MMP-9
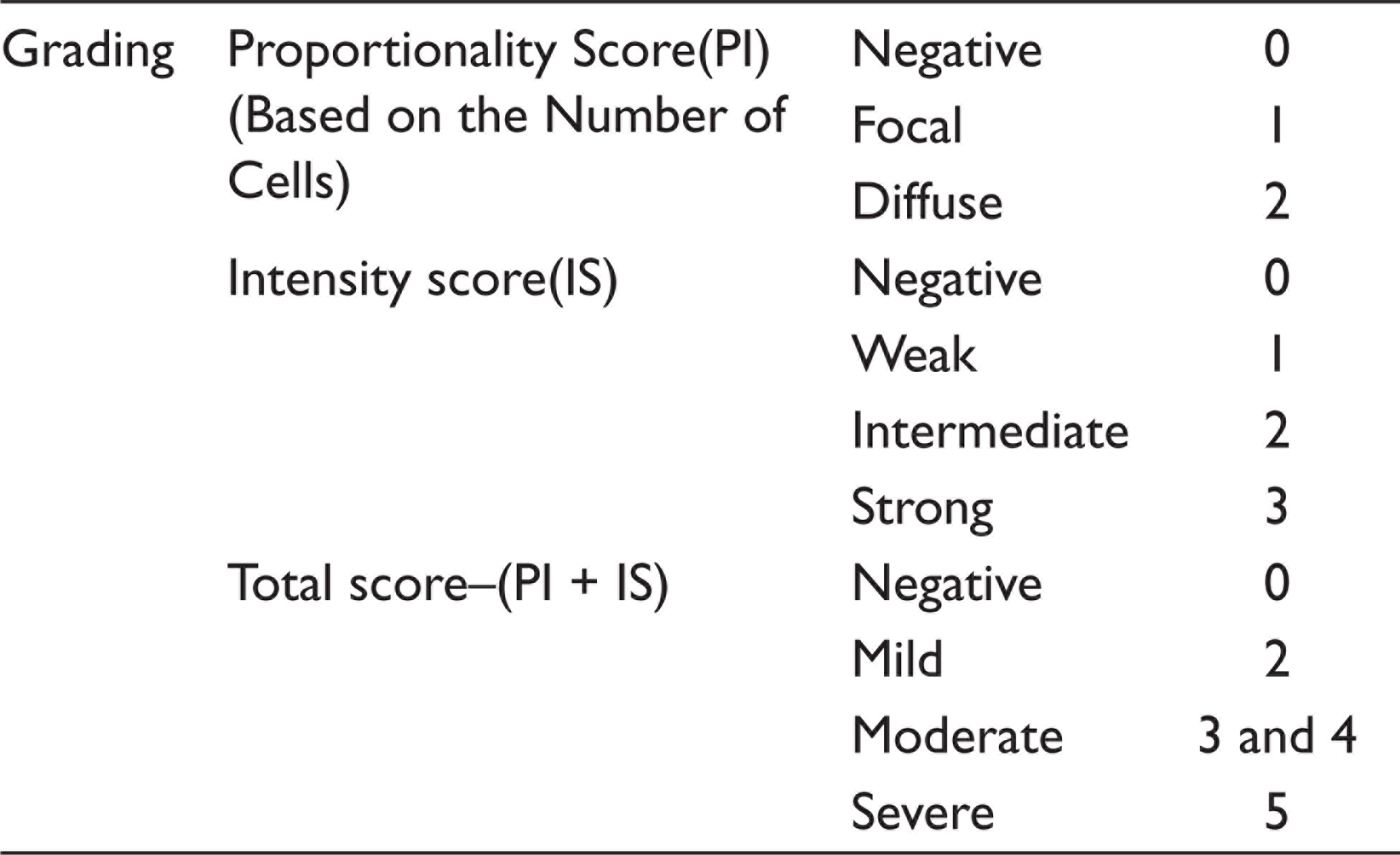
Statistical Analysis
All the differences between the two groups were statistically tested by chi-square test using SPSS software version 15.0 with significance level set at 5% (P < .05). Interobserver agreement between the two observers was evaluated applying kappa statistics.
Results
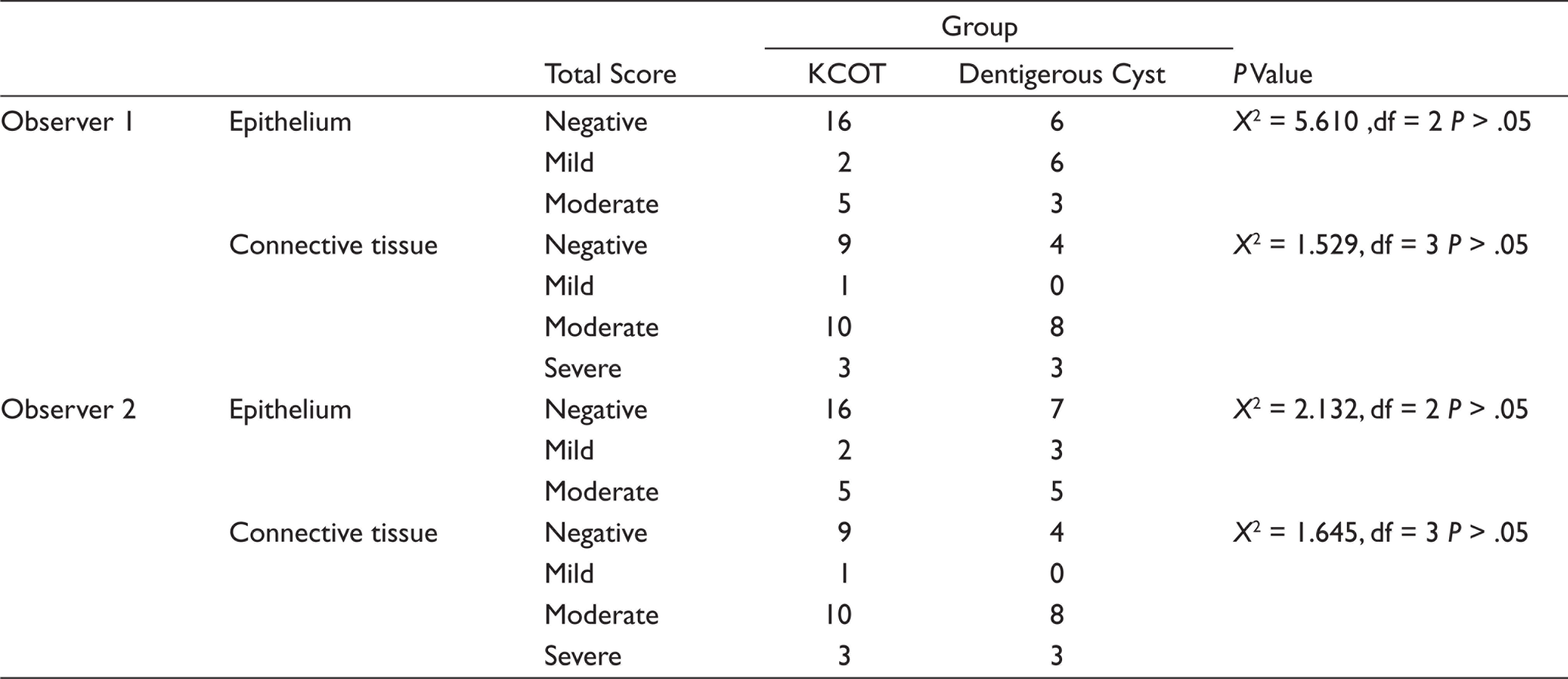
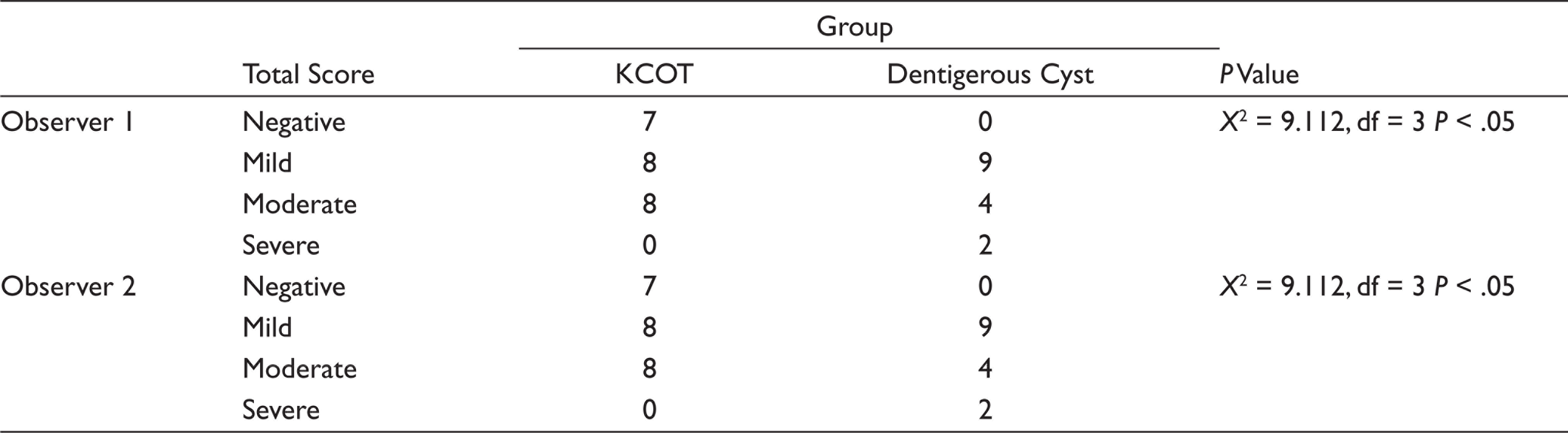
In MT analysis, on scanning the entire connective tissue wall, KCOT (86.9%) showed relatively more loose arrangement in comparison with DC (53%). Deeper third of connective tissue wall in KCOT is loose and correlates with color intensity ranging between bluish tinge to blue which is statistically significant (Table 2). Analysis between the arrangement of collagen fibers in the connective tissue wall and inflammation stained with MT are shown in Table 3. A quantitative analysis in confocal microscopy showed that there was no difference between intensities in the three regions in KCOT (P = .990) and DC (P = .233; Table 4). The immunoexpression of MMP-9 in KCOT and dentigerous cyst by observer one and two is shown in Table 5. Intermediate and diffuse positive of MMP-9 immunoexpression was seen in the epithelial and connective tissue wall of KCOT. Weak and diffuse positive immunoexpression was noted in the epithelial lining of DC and intermediate and diffuse positive expression was observed in the connective tissue wall of DC. MMP-9 expression by inflammatory cells in connective tissue wall was statistically significant between the groups (Table 6). Interobserver agreement by two observers was statistically significant for the immunoexpression of MMP-9 in KCOT and DC at kappa values 1.000 and 0.762, respectively.
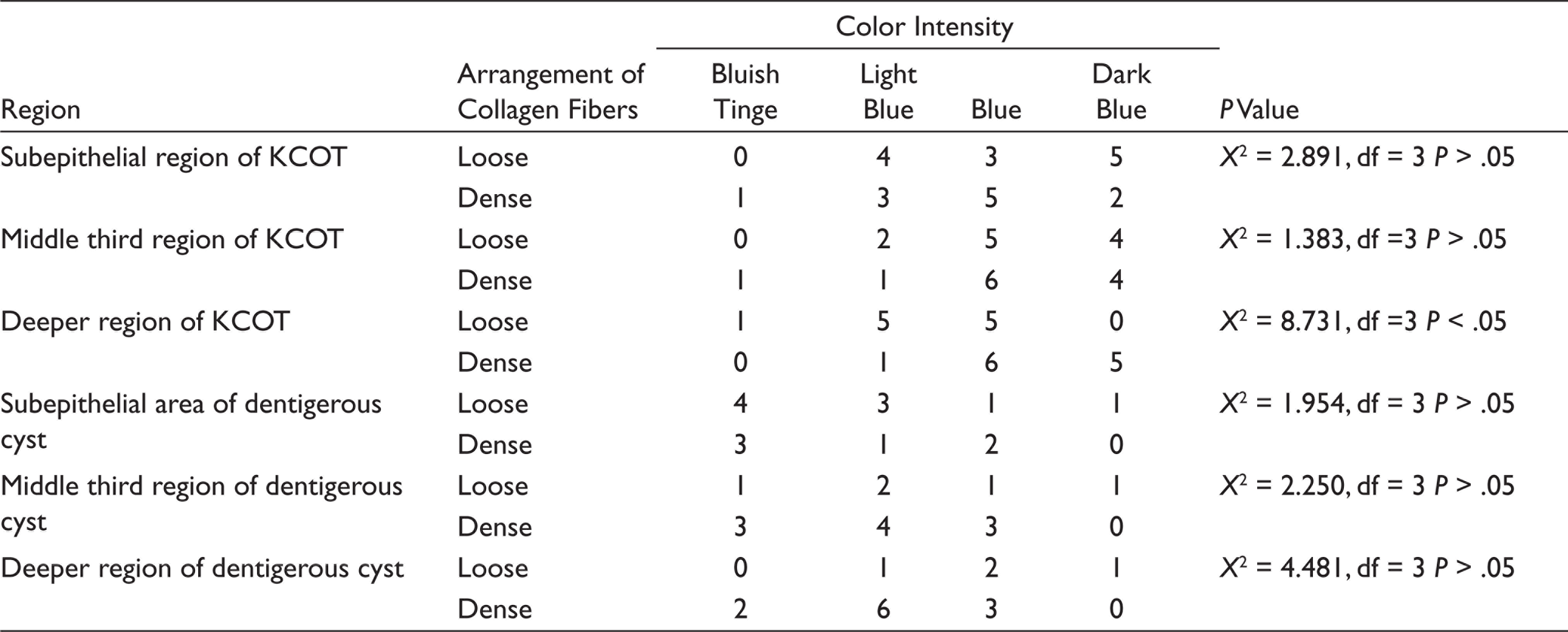
Arrangement of Collagen Fibers in the Connective Tissue Wall and Color Intensity Using MT
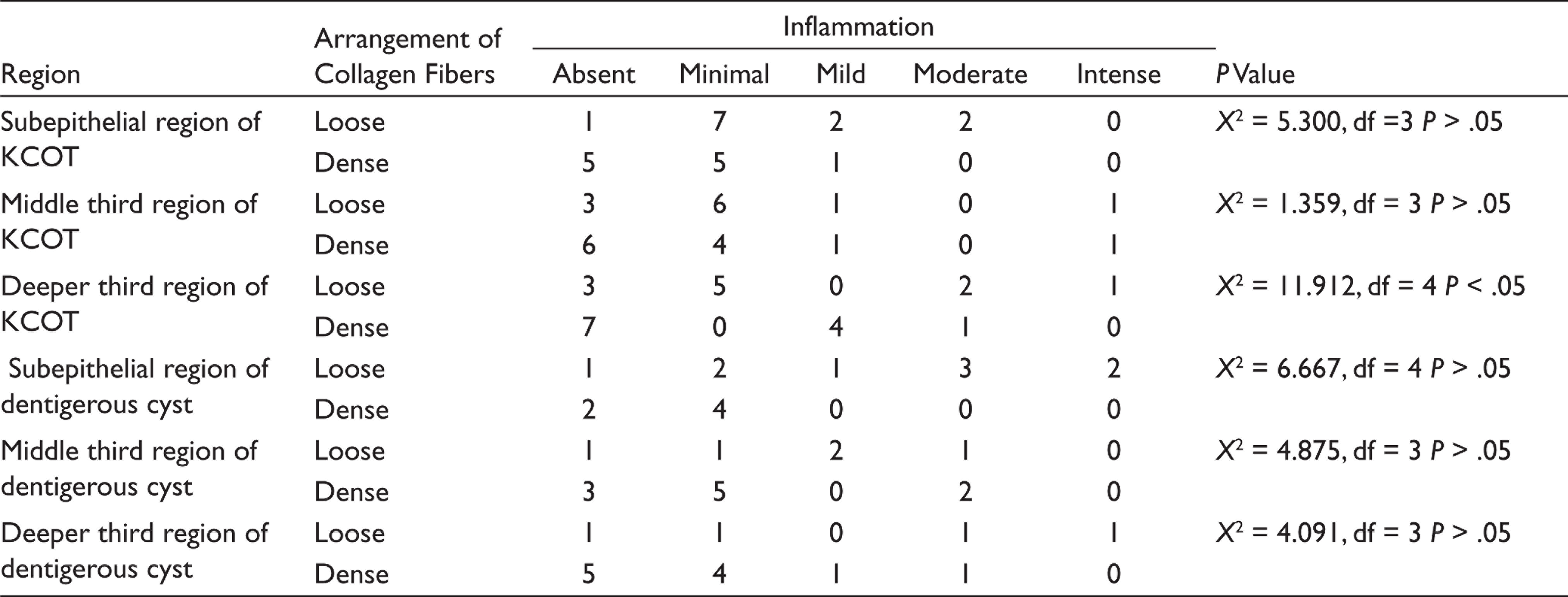
Arrangement of Collagen Fibers in the Connective Tissue Wall and Inflammation Using MT
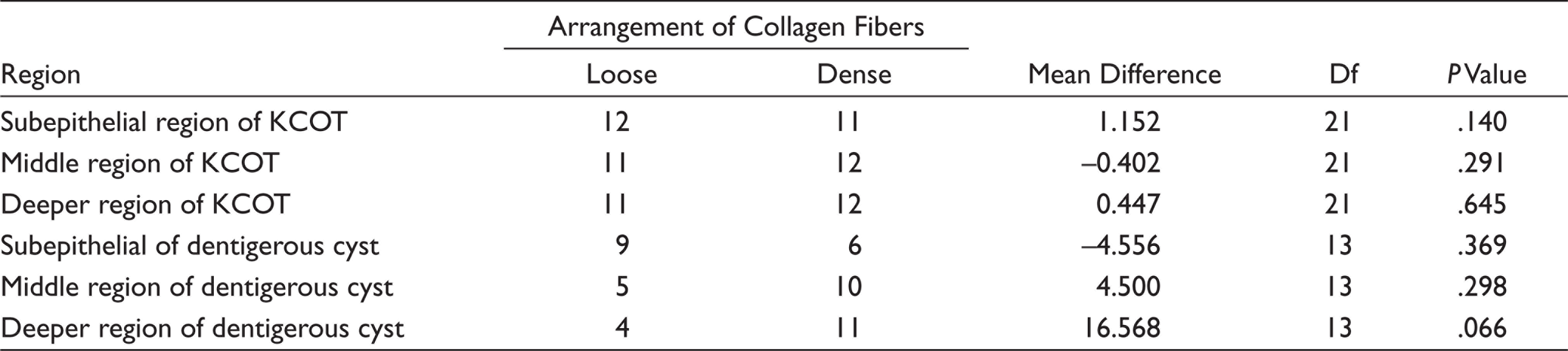
Intensity of the Connective Tissue Wall in Confocal Microscopy and Arrangement of Collagen Fibers
Immunoexpression of MMP-9 in KCOT and Dentigerous Cyst by Observers 1 and 2
Inflammation in the Connective Tissue Wall of KCOT and Dentigerous Cyst by Observers 1 and 2
Discussion
KCOT is regarded as a cystic lesion with a potential for aggressive, infiltrative behavior, and high rate of recurrence compared to other odontogenic cysts even in its sporadic form.4,14 Speculations have been made on aforesaid aggressive biological behavior. Most studies claim an epithelial component to play an integral part in this behavior by the effect of increased proliferative potential, eg, Ki-67, AgNOR, IPO-38 and impaired expression of tumor suppressor genes and their protein products PTCH, P 53, and MDM2.15–18
The role of ECM in the pathogenesis and behavior of epithelial odontogenic lesions suggests that these lesions arise as a result of defective epithelial mesenchymal interaction. Hirshberg et al. imparted stroma was loosely arranged in KCOT similar to that of other odontogenic neoplasms and therefore connective tissue stroma could not be regarded just as a structural support of the cyst wall. 5 Previous studies show that connective tissue wall plays an important role in the pathogenesis of KCOT.5,8,19,20
MT is a differential connective tissue stain that particularly colors collagen fibers blue which aids in light microscopic examination of the arrangement of collagen fibers. 21 Evaluation of the characteristics of connective tissue wall of KCOT and dentigerous cyst was carried out using MT stain by dividing into subepithelial, middle third, and deeper areas, and comparison with color intensity between the two lesions. Collagen fiber color intensity was expected for loose areas to appear bluish tinge, light blue, or blue in color and dense areas to appear blue or dark blue in color. The subepithelial region showed that the arrangement of fibers was loose in 12 cases (52.2%) and the remaining 11 cases (47.8%) were dense in KCOT. On correlation with the color intensity, loose collagen fiber arrangement ranged between light blue to dark blue. The dark blue color associated with loose arrangement in this region could be hypothesized as a result of subepithelial hyalinization. According to Hannah et al. subepithelial hyalinization could contribute greater tendency for recurrence proving the biological behavior of KCOT. 22 The color intensity viewed in the dense arrangement ranged between bluish tinge to dark blue. This appearance of bluish tinge and light blue in dense areas could be because of disorganization of the connective tissue wall caused by fibroblasts and inflammatory cells. The arrangement of fibers in the subepithelial region was correlated with color intensity in KCOT and was found to be statistically insignificant (P = .409).
The deeper region showed loose arrangement of fibers in 11 cases (47.8%) and dense arrangement in 12 cases (52.2%) in KCOT. On comparing the deeper third arrangement of fibers with color intensity in KCOT, it was found to be statistically significant (P = .033). SP Moure et al. (2011) suggested that the loose packing of collagen fibers is associated with changes in the neoplastic epithelia, promoting ECM degradation and infiltrative growth. 8 Regardless, the arrangement of fibers was not statistically significant between the two groups (P = .33) which was due to absence of loose nature of the entire connective tissue wall. However, when the entire connective tissue wall was observed, twenty cases (86.9%) showed the loose arrangement of collagen fibers. Like Hirshberg et al. 5 and Singh HP et al., 23 our study also rationalizes that the loose arrangement of collagen fibers could contribute to the aggressive behavior of this KCOT.
Dentigerous cyst showed the collagen fibers in subepithelial, middle third, and deeper third as loose arrangement in nine cases (60%), five cases (33.3%), four cases (26.7%), respectively, and dense arrangement in six cases (40%), ten cases (66.7%), eleven cases (73.3%), respectively. On correlation of color intensity with subepithelial third, middle third, and deeper third arrangement of fibers it was found to be statistically insignificant at P = .582, P = .522, and P = .214, respectively. Nonetheless, the dentigerous cysts showed dense collagen arrangement (53%) throughout the connective tissue wall. This arrangement possibly could be because of its developmental nature proving its distinct biological behavior. 24
The association of arrangement of collagen fibers with inflammation was carried out in both the groups which brought about to assess the loose arrangement was due to inherent nature of the connective tissue or because of inflammation (although only minimal inflammation is seen in KCOT according to the literature).25,26 In KCOT, the association of the arrangement of fibers with inflammation in the subepithelial and middle third regions was found to be statistically insignificant (P = .151, .715) while in the deeper region, it was found to be statistically significant (P = .18). Even so, there was a statistical significance (P = .027) between the arrangement of collagen in entire connective tissue wall and inflammation in KCOT. According to Browne et al. 19 inflammation plays a role in the tumorigenesis of KCOT. Our study findings were congruent with Hirshberg et al. that inflammation contributes to the loosely packed arrangement and might be composed of procollagens, intermediates, or pathologic collagens than tightly packed fibers. 20
In DC, the collagen arrangement was compared with inflammation in entire connective tissue and was found to be statistically significant (P = .020) though its association pertaining to the subepithelial, deeper, and middle third regions was found to be statistically insignificant (P = 0.155, 0.181, 0.394). According to Rakprasitkul et al., 27 the DC capsules have dense arrangement of collagen. According to Browne et al., 19 inflammatory stimuli is responsible for changes in the arrangement of collagen fibers when dentigerous cyst may show similar changes as collagen component of KCOT. Our study results were congruent proving the inherent dense nature of dentigerous cyst and are affected only when inflammation is present.
We have assessed the morphological characteristics of collagen fibers in light microscopy proceeded along with three-dimensional confocal laser microscopy in KCOT and DC. PMA-PSR is a modality used to carry out quantitative analysis collagen in odontogenic entities in confocal microscopy because of its characteristic higher image resolution. 8 The intensity values of the connective tissue wall obtained in confocal was recorded as a histogram for the same three areas observed in MT staining. On comparing the intensity of connective tissue wall in the subepithelial, middle, and deeper regions with the arrangement of collagen fibers, it was statistically insignificant for both KCOT (P = .140, .291, 0.369) and DC (P = .369, .298, .066), respectively. Both KCOT and DC could show altered results in the subepithelial, middle, and deeper third areas because of higher intensities taken up by both nuclei and cytoplasm of basal epithelial cells and inflammatory cells. A quantitative analysis showed that there was no difference between intensities in the three regions in KCOT (P = .990) and DC (P = .233). Our study was consistent with the study of Moure et al. which inferred that although the amount of collagen was similar, the types of collagen could have been different.
MMP form an enzyme family capable of degrading almost all ECM and BM components. They play an important role in normal tissue remodeling and growth, as well as in many destructive pathological conditions such as inflammation, tumor growth, and metastasis. Studies suggest that enzymatic degradation of the BM collagen type IV and bone matrix by MMP-9 is involved in the expansion of odontogenic cysts. 9 The loosely arranged collagen in the connective tissue wall of KCOT could be related to the degree of MMP-9 expression. These factors may be interrelated and can be used to ascertain the neoplastic potential in the connective tissue wall of KCOT. The immunoreactivity to MMP-9 seen in both the epithelium and the connective wall of these lesions can be explained by the fact that the protein is produced by epithelial cells, fibroblasts, macrophages, plasma cells, lymphocytes, and neutrophils. 28
The immunoexpression of MMP-9 in the epithelium between groups (KCOT and DC) was statistically insignificant (P = .061). The immunoexpression of MMP-9 in the connective tissue wall between groups (KCOT and DC) was statistically insignificant (P = .676). The expression in the epithelium of DC (60%) was higher than that of KCOT (30.4%). The expression in the connective tissue wall of DC (73.3%) was slightly higher than that of KCOT (60.8%). Also, on comparison between DC (100%) and KCOT (69.9%), the inflammatory cells in DC demonstrated higher expression of MMP-9 which was statistically significant between the groups (P = .028). Thereupon it is suggested that the high expression of MMP-9 in dentigerous cyst was because of the increased amount of inflammatory cells and not an inherent nature of the lesion.
However in KCOT, the immunoexpression of MMP-9 was seen to be expressed more in fibroblasts, collagen fibers rather than only inflammatory cells which prove the inherent nature of its aggressive behavior. Our study was consistent with Santos et al. 9 higher expression of MMP-9 in connective tissue wall of KCOT was similar to other odontogenic neoplasms 29 which could contribute to the biologic behavior and promote the degradation of ECM. Thus, the loose arrangement and MMP-9 expression in fibroblasts, collagen fibers, and inflammatory cells in KCOT could contribute to its biological behavior. Taken together, these findings and the results of the present study suggest that the higher expression of MMP-9 in the connective tissue of KCOT could contribute additional aggressive behavior.
Conclusion
Our study is a maiden attempt to evaluate the collagen arrangement using MT in light microscopy and it examined the same focus in confocal microscopy using PMA-PSR along with the correlation of MMP-9 expression. This study shows that the loose arrangement in the connective tissue wall associated with increased MMP-9 expression may have a promoting role in the aggressive behavior of this lesion. If inflammation is present, it could further contribute to the tumorigenic behavior in KCOT.
Footnotes
Future Scope
Further genetic studies with long-term follow up are required to understand the true behavior of KCOT. The study limitations were smaller sample size and expensive kits for performing the study.
Acknowledgements
I gratefully acknowledge Mr B. Jayaraj, my father for his invaluable support, Dr Malini, and Sri Ramachandra University, for helping me with confocal microscopy. I would also like to thank Dr GS Kumar, K.S.R Dental College, for helping me with image analysis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
