Abstract
Aim:
Cancer stem cells (CSCs) or tumor-initiating cells have self-renewal and uncontrolled tumor growth capacity that promotes metastasis and recurrence. Challenges in anticancer have found a lateral dimension of treatment against a new pattern of tumor microcirculation, known as vasculogenic mimicry (VM), involved in cancer progression. Increasing evidence suggest that CSCs are involved in the formation of VM. In this review the correlation between CSCs and VM formation is been enlightened.
Materials and Methods:
The literature search was done in Medline, PubMed, Wiley, Science Direct, and Scopus. The keywords used for database search were cancer stem cells, vasculogenic mimicry, and anticancer therapy.
Results:
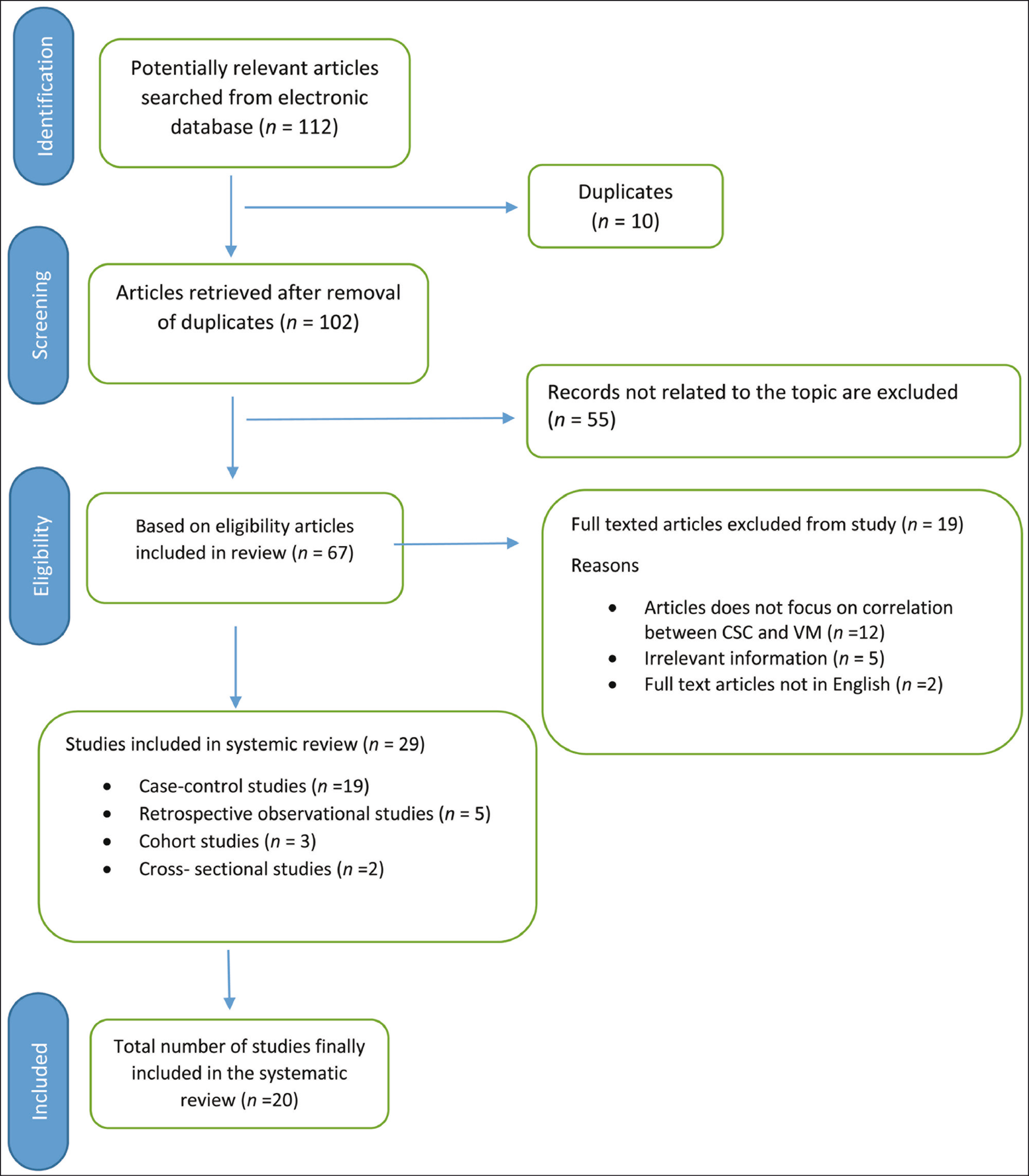
A total of 112 articles appeared from various sources, of which 102 were subjected for screening and 20 were related to the research objective.
Conclusion:
Based on the literature a positive correlation exists between CSC and VM, which plays a key role in tumor progression, and hence, can serve as a potential target in anticancer therapy.
Introduction
Cancer stem cells (CSCs) first identified by John Dick in acute myeloid leukemia in the late 1990s and proved important role of CSCs in cancer research. 1 CSCs, also known as tumor-initiating cells or tumor-propagating cells, are small subpopulation of tumor cells that demonstrate properties similar to normal stem cells. 2 CSCs are cancer cells that possess the ability to self-renew and give rise to all cell types found in a particular cancer sample. A genetic and epigenetic change in tumor alters the signaling pathway which results in phenotype similar to that of stem cells, progenitors, and differentiated cells.1,3 CSCs play a vital role in maintaining the population of tumor mainly responsible for cancer recurrence and drug resistance. 4 Proper oxygen and nutrient supply is necessary for the growth and metastasis of tumors.
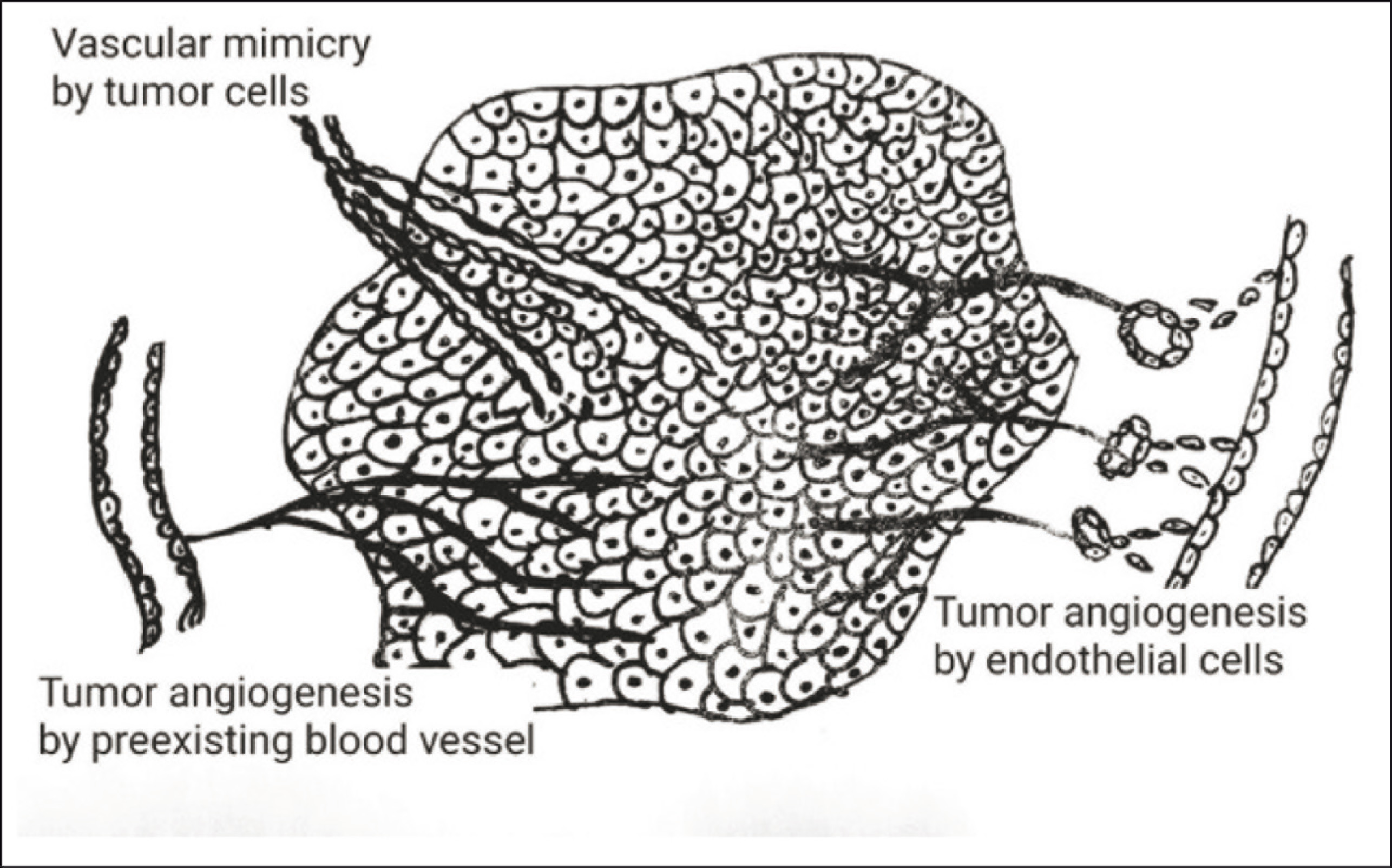
Tumor angiogenesis alone is not an exclusive method for nourishing the tumor. Recently, apart from sprouting angiogenesis two mechanism for blood supply and metastasis has been discovered in aggressive neoplasm: vasculogenic mimicry (VM) and vascular co-option (VCO). 5 In VCO, cancer cells closely adhere to the pre-existing blood vessels or capillaries to obtain nutrients and oxygen. This eventually compresses the vessel, which undergoes hypoxia followed by necrosis, and leads to neovascularization. Hypothetically, both VM and VCO would explain the failure of antiangiogenic therapies, while VCO would be essential in the metastatic growth.5,6,7 Vasculogenic Mimicry (VM), describes the de novo formation of perfusable, matrix-rich, vasculogenic-like networks by destructive tumor cells (Figure 1). VM chiefly functions through the perfusion pathway for quickly rising tumors by transporting fluid from spongy vessels and/or involving with endothelial-lined vasculature. 8 In this review, the role of CSC in microvessel formation like VM has been enlightened.
Tumor angiogenesis formed by direct branching from adjacent blood vessels to tumor cells (right side). The endothelial cell proliferation from adjacent blood cells joins to form new blood vessels and supply to tumor cells. Vasogenic mimicry is formation of blood vessels by tumor cells.
Materials and Methods
Search Criteria
Studies, which were included in the review, were selected on the basis of the following points:
Articles published from 1999 to 2020. The articles’ titles, study design, and their abstracts. Including only full-text articles in the review. The research question was based on correlation between CSC and VM formation.
The search for the systematic review was performed using four databases: Cochrane library (www.cochranelibrary.com), Embase (www.embase.com), PubMed (www.ncbi.nlm.nih.gov/pubmed), Scopus (www.scopus.com), and Web of Science (www.webofknowledge.com). The search was based on MeSH entry terms mixed with Boolean phrases “AND” or “OR” and keywords such as CSC, VM, and CSC in squamous cell carcinoma and anticancer therapy. Duplications were discarded. Clear representation of the included literature and summarization of the available studies were done using Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) checklist.
Inclusion and Exclusion Criteria
The inclusion criteria were that the studies must be descriptive and experimental, full-text and articles on CSC in VM formation. The exclusion criteria were that studies must be review articles, cell and/or animal studies, letters, clinical cases, conference summaries, comments and studies that provided irrelevant information, and articles which did not focus on the correlation of CSC in VM formation. Articles not relevant to the topic of interest or objectives of the study were excluded. Gray literature such as unindexed or unpublished studies were not included.
The initial selection of the articles to be included was accomplished by two independent authors, reading the titles and abstracts related to CSC and VM formation. Subsequently, the full text of each potentially relevant study was retrieved and analyzed by the two independent reviewers. The quality of the methodology of each study was evaluated by two reviewers independently who assessed the quality of the studies identified. Only studies which qualified the evaluation criteria were included after thorough review by two independent reviewers.
Data Analysis
Among 112 potentially relevant related articles finalized after elimination of those with weak relationship to objectives of the study, 20 fully related articles were included in the study and were completely studied and investigated. The number of articles included at each stage based on the eligibility criteria are shown in Figure 2. After completing the search, the selected documents were summarized and categorized based on the topic and its implications. Finally, the selected articles were categorized based on indexation and journal source (namely Fronti Onc, Stem Cell Int, Mod Path, Trans Onc Nat Rev Drug Discov, J Exp & Cli Cancer Research, Journal of Cell Science, Int J BCB, Nat Library Med).
Discussion
Origin of CSC
Earlier two major models were hypothesized for the evolution of CSC: the clonal evolution model (mutation hypothesis) and the cancer stem cell model (cellular hierarchy organized in a tumor).9,10 In tumor, different types of cells are seen, including cancerous cells, infiltrating normal cells, and induced pluripotent stem cells. One of the hallmarks in cancer in tumor heterogeneity is the initiating factor for cancer. This heterogeneity is chiefly reflected by genetic instability and epigenetic alterations which are responsible for the differentiation potential in a cell. In solid tumor, cancer cells exist in different states of differentiation and this can be expressed by the cell surface markers in various tumors.11,12 Cancer stem cell theory states that a small subset of cells within a tumor characterized by stem-like properties can lead to tumor initiation, progression, and metastases. The self-renewing and differentiating capacity of CSC are responsible for tumor heterogeneity. 11
Apart from genetic mutation, tumor plasticity is mainly affected by its microenvironment. Similar to normal stem cells, CSCs also require a CSC niche to maintain their self renewal and differentiation properties. In tumor, this is achieved by direct cell–cell interaction and signaling factors from the perivascular niche. The reprogramming mechanism in these vascular niches can be explained through the following example: epidermal growth factors secreted from endothelial cells in head and neck tumors are responsible for stemness in tumor cells. 13 The CSC niche contains cellular components such as cancer-associated fibroblasts, tumor-associated macrophages, tumor-associated neutrophils, MSCs, cell-mediated adhesion, and soluble factors that play critical roles in cell–cell communication. 14 Several hypotheses for CSC origin have been described, including cell fusion, horizontal gene transfers, genetic instability, and cell microenvironment influences 10
Origin and Structural Characteristics of VM
VM was initially described as an aggressive, uveal form of melanoma originating because of “dedifferentiation” of tumor cells into an “endothelial-like” phenotype. 15 In these tumors, VM was identified as periodic acid-Schiff (PAS) positive and was devoid of bona fide endothelium. VM channels appeared to contain erythrocytes and were hypothesized to connect with the existing vasculature.16,17 Recent research have focused on two main concepts for vasogenic mimicry. According to the first concept, some tumor cells take on certain characteristics of vascular endothelial cells and line the tumor’s blood vessels. A study also suggests that the tumor cells in the immediate environment of the nascent vessel are co-opted for the purpose. The co-opted cells are thought to retain most of their tumor-cell characteristics while acquiring a limited number of endothelial-cell features.18,19 A second possibility is that putative CSC are the cell-of-origin for some tumor cells. According to this idea, tumor stem cells are both refractory to most traditional therapies and are capable of regenerating the tumor following treatment.20,21
A structural difference exists between the fluid-conducting vessels such as angiogenesis, lymph vessel formation, and VM. In blood vessels formed by angiogenesis, the lumen is lined by a single layer of endothelial cells with surrounding continuous basement membrane. Similarly, lymph vessels consist of an inner layer of endothelial cells where the basement membrane is not continuous. In VM, the cancer cells sit on the glycoprotein-rich membrane (matrix) surrounding the central lumen. Studies suggest that VM vessels also have a glycoprotein-rich inner coating composed by collagens and laminin, among other proteins. The architecture of the matrix-associated vascular channels are characterized by interconnected loops and networks which are not typical of angiogenesis22,23
Tubular type and the classical matrices type are the two distinct types of VM reported in the literature. The tumor cells associated with VM structures revert to an undifferentiated, embryonic-like phenotype and acquire the characteristics of endothelial cells. These malignant cells that gained endothelial cell characteristics play an important role in invasion and metastasis.24,25
Relation Between CSC and VM
Similar to normal stem cells, CSCs exhibits multilineage differentiation potential and plasticity. Plasticity is the capacity of stem cells to either differentiate or transdifferentiate into many cell types. 26 Treatment resistance and existence of CSC are linked by the presence of cells with altered phenotype, and stem-like characteristic in solid tumors contributes to intratumoural heterogeneity. The plasticity of tumor cells induces heterotypic signals between these cells and the adjacent stromal cells in the form of stromal signals which induce the signaling cascade. This leads to the expression of transcription factors and various target genes responsible for the formation of various mesenchymal traits. 27
The role of CSCs in neovascularization, including angiogenesis and vasculogenesis, is still a controversy. Several studies were done to show the positive correlation of CSC and VM in cancer progression (Table 1). Yao et al. studied that glioblastoma cells express G-protein coupled receptor which upon activation promotes migration, growth, and production of vascular endothelial growth factor (VEGF). The results were conclusive of CSCs association in angiogenesis and their role in novel therapeutics. 28 Schatton et al. identified a group of malignant melanoma stem cells in tumor which are responsible for tumor initiation, increase in growth and volume, and formation of VM by the expression of nestin and VE cadherin genes from melanoma cells. 29 Therefore, melanoma stem cells with transdifferentiation property may play a critical role in VM. In previous studies done by Pezzolo et al., involvement of tumor cells in the VM formation was noted in human neuroblastoma. In the same study the microvessels displayed an open lumen with red blood cells indicating that these vessels were functional and tumor-cell-derived vascular endothelium-like cells were different from normal endothelial cells in phenotype and function. Also, Rigolin et al. found tumor-associated endothelial microvessels, human B-cell lymphomas, and multiple myeloma. 30 In 2008, Shen et al. proposed that the precancerous stem cells representing the early stage of developing CSCs serve as tumor vasculogenic stem/progenitor cells capable of differentiating into tumor vasculogenic endothelial cells.31,32
Liu et al. demonstrated a positive correlation between the tumor cell-lined vessels in lymph node metastasis in oral squamous cell carcinoma (OSCC). 33 Wu et al. in 2017 studied the expression of CSC and VM in OSCC and their significant correlation with clinicopathological features of OSCC 34 Several studies were done in head and neck tumors where VM and CSC exhibited a definitive correlation and its association with TNM staging (tumour, lymph node and metastasis staging system), 35 increased tumor infiltrating neutrophil, and tumor-associated macrophages which are responsible for poor prognosis.34,36 In 2018, Soussan Irani et al., found increase in VM formation, CSC, EMT, and microvessel count in OSCC. 37
Studies Done for Understanding the Correlation Between Cancer Stem Cells and Vasculogenic Mimicry
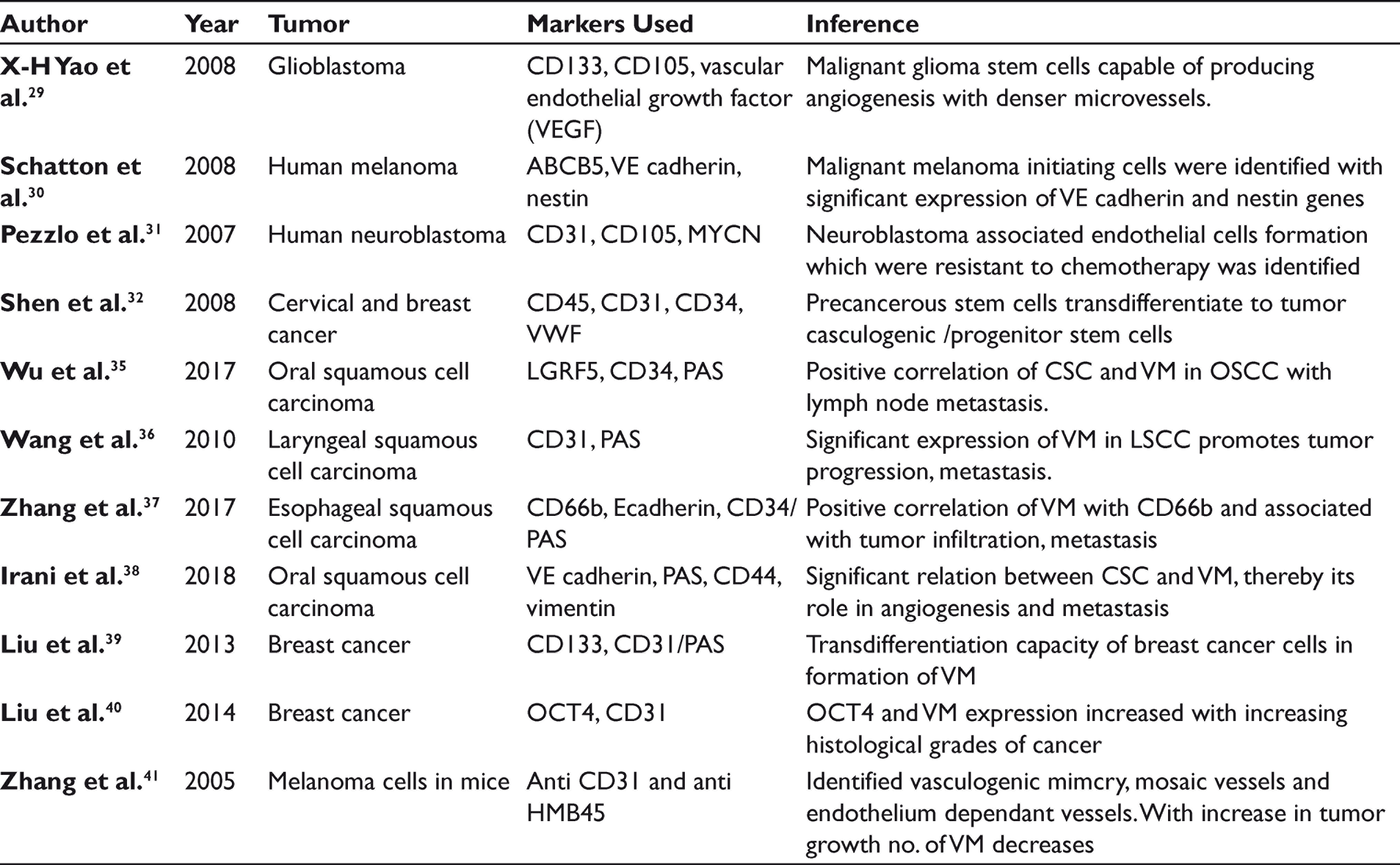
In previous studies, the demonstration of VM in solid tumors was done by double staining of CD31 and PAS stain. CD31-negativity and PAS-positivity in vascular-like pattern which are lined by tumour cells are identified as VM. Cytokeratin positive staining was also observed in the cell lining of VM.38,39 According to the observation of Zhang et al. in melanoma xenografts, the dominant blood supply pattern in early stage of tumor is VM, and as the tumor growth progresses, endothelial-dependent vessels increases. 40 Based on the studies, the authors proposed a three‑stage blood supply pattern consisting of VM, mosaic vessels, and endothelium‑dependent vessels. This inverse change tendency between VM and endothelium‑dependent vessels may be because of the persistent differentiation of CSCs to endothelial cells without tumor cell phenotypes.24,40,41 The above literature provides a promising correlation between, CSC and VM, its association in tumor progression and lymph node metastasis thereby enhances its effect in the survival of the patient. Thus, the cancer-cell-lined vessels can be a potential target in novel therapeutics to reduce tumor progression.
As a Therapeutic Target
CSCs help in cancer progression by providing alternate blood supply to the tumor mass, which proves the inefficiency of current treatment strategies in advanced cancer. Hence, targeting the alternative blood supply (VM) formed by tumor cells can be a novel target in cancer therapy. Itshaki et al. in 2013 studied nicotinamide-driven downregulation of vascular endothelial cadherin (VE cadherin) which further destroys VM formation and its channels. Nicotinamide also significantly inhibited melanoma cell proliferation and an inverse effect in tumor cell invasion. 42 In 2014, Rui-Jun Ju et al. conducted an in vitro studies targeting epirubicin plus celecoxib liposomes in breast cancer cells. The liposomes were capable of penetrating and destroying cancer cell spheroids by regulating apoptosis proteins. VM formation was prevented by downregulation of MMP-9, VE-Cad, FAK, EphA2, and HIF-1a. 43 In 2015, Yun Xia investigated fasudil-induced destruction of VM formation by reducing B16 melanoma cell xenograft in tumor without any toxicity in the mice model. Fasudil destroys VM formation by using Rho-GTPase agonist lysophosphatidic acid, which blocks the RhoA/ROCK signaling pathway. 44 In 2016, Maria Serova et al. studied the effect of sunitinib in renal clear cell carcinoma by both in in-vivo and in-vitro. Sunitinib-resistant tumor cells developed with aggressive phenotype were destroyed by the second line therapy of everolimus which suppressed the VM formation. 45 In 2009, Bozec et al. studied the combination of cetuximab, sunitinib, and radiation therapy which lead to improvement in the reduction of tumor cell proliferation and differentiation of head and neck cancers. 46 In 2017, Xiaoxu Li et al. studied niclosamide, an antihelminthic drug, that inhibits VM formation in oral cancer by inhibiting proliferation and promoting apoptosis of cancer cells. Niclosamide also causes downregulation of VM-related genes (VEGFA, MMP2, ROCK1, STAT-3, and Cdc42) and upregulation of miR-124 which decreases the expression of STAT-3. 47
Conclusion
Self-renewal capacity and plasticity of CSCs play a key role in malignant transformation of normal cells in the surrounding tumor environment and new vascular channels for tumor cell nourishment. Targeting these vascular channels and CSCs is vital to prevent chemo-resistance and tumor relapse. Anticancer therapy usually includes eradicating the tumor cells completely or pushing them toward a differentiated state. The formation of CSC niche and the tumor-associated vascular pattern make the anticancer therapy more challenging. Hence, understanding CSCs and their role in formation of VM may serve as a novel treatment therapy to battle against the drug resistance promoted by CSC.
Future Scope
CSCs not only induce vasculogenic mimcry but also have possible influence on angiogenesis, formation of cancer-associated fibroblast, and induction of hypoxia, which promotes resistance to cancer therapy. CSC-induced VM formation is chiefly involved in tumor cell nourishment, proliferation, and survival, which is directly related to the progression and prognosis of the tumor. Hence, in this review the authors have highlighted the possible correlation of CSCs and VM in the formation of OSCC and how OSCC can serve as a potential target for anticancer therapy.
Footnotes
Acknowledgements
We would like to thank Dr C. Bharath for his expert advice and encouragement in writing this review, and also thank the anonymous reviewers for their useful suggestions.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
