Abstract
Aim:
The cancer stem cells (CSCs) are known to be responsible for drug resistance and cancer relapse in the treatment of cancer. Identification and isolation of CSCs and study of their properties will play a crucial role in developing an effective drug against these targets. The aim of the study was to isolate CSCs from primary cancer by the tumorspheres enrichment method, to confirm by indirect immunofluorescence and gene expression of stem cell markers by using real-time polymerase chain reaction (RT-PCR) technique.
Materials and Methods:
In this in vitro study, we enriched oral CSCs through tumorsphere formation assay from seven primary cultures of OSCC patients with defined serum media. The expression and localization of the cell surface markers of CD133 and CD44 were tested by indirect immunofluorescence. Gene expression of stem cell markers such as CD44, CD133, Oct4, Sox2, and Nanog were quantified by RT-PCR technique. One-way analysis of variance was applied to analyze gene expression.
Results:
Tumorsphere formation has been used to isolate the CSCs from the OSCC tissue culture. Both CD133 and CD44 antibody confirmed the presence of CSCs through indirect immunofluorescence. In comparison to parental cell lines, the expression levels of CD133, CD44, Oct4, Sox2, and Nanog stem cell were significantly higher in CSC-enriched subpopulations.
Conclusions:
The cost-effective spheroid enrichment and the indirect immunofluorescence methods are useful for the isolation of CSCs from the primary tumor.
Introduction
Oral cancer is one of the most common cancer with annually 354,864 new cases and 177,384 cancer deaths reported worldwide. 1 Oral cancer accounts for around 30% of all cancers in India and it ranks among the top three types of cancer in the country with 119,992 new cases and 72,616 cancer deaths annually. This is the major cause of mortality in men relative to women. 2 The common risk factors for oral cancer are habits of smoking or chewing tobacco with pan masala and alcohol. The current treatment methods are surgical treatment along with adjuvant radiotherapy (RT) and sometimes chemotherapy (ChT), which effectively reduce the total tumor size but does not kill all the cells. 3 Despite a variety of advances in treatment methods, the 5-year survival rate after treatment of oral cancer (all the stages included) is around 50%. The treatment failure is due to loco-regional recurrence. 4 These failures may be due to tumors comprising minor populations of cells called cancer stem cells (CSCs) responsible for tumor initiation, propagation, and regeneration. These CSCs display ChT- and RT-resistant properties. 5
Identification and isolation of CSCs is an important step to study the properties of CSCs for developing the drugs to target these CSCs. Many in vitro methods are used for identification and isolation of CSCs including fluorescence-activated cell sorting (FACS), magnetic-activated cell sorting (MACS), and flowcytometer. These methods involve high-cost maintenance, machine calibration, and compensation gating protocols. 6 The method of tumorspheres or the spheroid enrichment is an effective, economical, and consistent approach to CSC analysis. The method involves the cultivation of cells in media containing growth factors and anchorage-independent conditions prevent the differentiation. 7 Using this method, several researchers have isolated the CSCs from primary head–neck squamous cell carcinoma (HNSCC), oral cancer cell lines, and gastric cancer cell lines.8–9 Locke et al. demonstrated in their study that holoclone morphology colonies have more highly clonogenic properties. Harper et al. revealed that holoclone morphology colonies contain undifferentiated stem cells, whereas paraclones or meroclones contain differentiation of very less number of CSCs. 10
A number of cell surface protein markers and expression profiles of oral cavity squamous cell cancer (OCSCC) tumor samples and cell lines have been studied as potential CSC markers. It will not be sufficient to classify CSC by a single unique marker in an overlapping hierarchy of cell population subsets. Thus, the CSC characterization studies were concentrated on the use of combination of such markers. 11 CD44 is cell surface protein and a receptor for hyaluronic acid, which is involved in cell–cell, cell–matrix interactions, and also in cell migration. It also helps the growth factors to accumulate on the cell surface, along with cell adhesion. 12 CD133 is cell surface protein and has a possible role in membrane organization. It has been verified to be a CSC marker in many cancers positive correlations within oral cancer patients. 13 The self-renewal and maintenance of the stem cell population in the undifferentiated state were known to be regulated by OCT, NANOG, and Sox2. 14 Many studies demonstrated that CD133 +ve and CD44 +ve (CSC surface marker) CSCs in HNSCC and OSCC have shown higher clonogenicity, self-renewal, proliferation, tumorigenicity, tumorsphere formation, and EMT phenotype. Indirect IF uses two antibodies, the unconjugated primary antibody directed by fluorophore and conjugated secondary antibody for detection. 8 Primary antibodies are costlier than the secondary antibodies, and thus additional cost can be reduced by using the same conjugated secondary antibody for detection of varied primary antibodies. 15
The studies related to head and neck stem cell identification and niche in Indian population are underreported and yet to be explored. Thus, this study implies to study isolation of CSCs from primary cancer cells by the tumorspheres enrichment method and also their confirmation by indirect immunofluorescence using CD133 and CD44 stem cell markers and by gene expression of stem cell markers OCT4, NANOG, and Sox2 using real-time polymerase chain reaction (RT-PCR) technique.
Materials and Methods
Setting and Design
This is an in vitro study carried out at MMNGH Institute of Dental Sciences, Belagavi, over a period of 6 month (Oct 2019 to March 2020). The study was approved by our institutional ethical committee (No-1452) and the patient’s consent was also taken. Patients (six male and one female) diagnosed with squamous cell carcinoma with habit history undergoing excision were included in our study. Patients with history of diabetes, hypertension, and other malignancies and those who were taking antibiotics were excluded for our study. The tissue samples were excised from the tumor of buccal mucosa of patients undergoing excision. One tumor sample was not processed due to contamination.
Primary Culture
The tissues samples were collected in Dulbecco’s Modified Eagle Medium (DMEM) containing 5% antibiotic medium during the standard surgical procedure. Primary tumor samples were rinsed with a 10% povidone-iodine solution for 1 min to prevent microbial contamination. The tumor was washed with PBS and culture media twice. Then tissues were cut into 2-mm-sized small pieces and transferred into a 15 ml centrifuge tube containing dissociation solution (DMEM-F12 with 300 U/mL collagen IV, 100 U/mL hyaluronidase, and 125 U/mL DNAse). The centrifuge tube was incubated at 37° C for 30 min and vortexing was carried out at regular intervals to dissociate cells from the tumor. Primary cancer cell cultures were formed by removing fibroblast-like cells by passage. The old culture medium was replaced with a fresh one twice a week. 16
Tumorsphere Culture
CSCs have unique properties to survive and grow in the form of spheroid bodies in the serum-free media with specific growth factors in low attachment culture plates. 7 In each well, approximately 5 × 103 cells were seeded in a 12-well ultra-low attachment plate. Then 200 µl of serum-free DMEM/F12 medium supplemented with fibroblast and epithelial growth factors. It was maintained in a CO2 incubator and the media was changed every third day. The tumorspheres were collected using serological pipettes along with media, as the spheres were floating, dense, compact three-dimensional groups of cells. The collected tumorspheres were transferred into centrifuge tube and centrifuged at 1,300 rpm for 3 min followed by washing with PBS. Trypsinization was carried out to get unicellular cell suspension, breaking the spheres without damaging cells. 17
Indirect Immunofluorescence
Cells from the single-cell colony were seeded in each well, containing cover slips of 24-well flat-bottom microplate and incubated overnight. Cells were then fixed in 4% paraformaldehyde for 30 min and washed with PBS. Then 100 µl of blocking solution was added containing PBS with 1% BSA, 0.1% Triton X-100 for 30 min followed by washing twice with PBS. Primary antibodies for CD133 (1:200) and CD44 (1:250) were added and incubated for 1 hour. After washing with PBS, fluoresce in isothiocyanate (FITC) (1:100) was added and incubated for 1 hour. The cells were then washed three times in PBS and examined under the fluorescent microscope (Olympus BX41) at 20× magnification. 18
Real-Time Polymerase Chain Reaction
Total RNA was extracted from samples using Qiazol reagent (Qiagen, Hilden, Germany). The cDNA conversion was carried out by using the PrimeScript RT reagent kit (Takara, Shiga Prefecture, Japan) following manufacturer instructions. The sequences of specific primers PCR thermal cycling conditions were shown in Table 1. The reaction mixture was prepared in a total volume of 20 µl, PCR thermal cycling conditions were performed in Realplex master cycler (Eppendorf, Germany), and melting curve analysis was performed to confirm the specificity of primers. Cycle thresholds (ct value) for all the samples were obtained. The ratios of specific mRNA expressions were normalized by the value of the housekeeping gene β-actin. 19
Primers Used for the Real-Time Polymerase Chain Reaction Analysis

Statistical Analysis
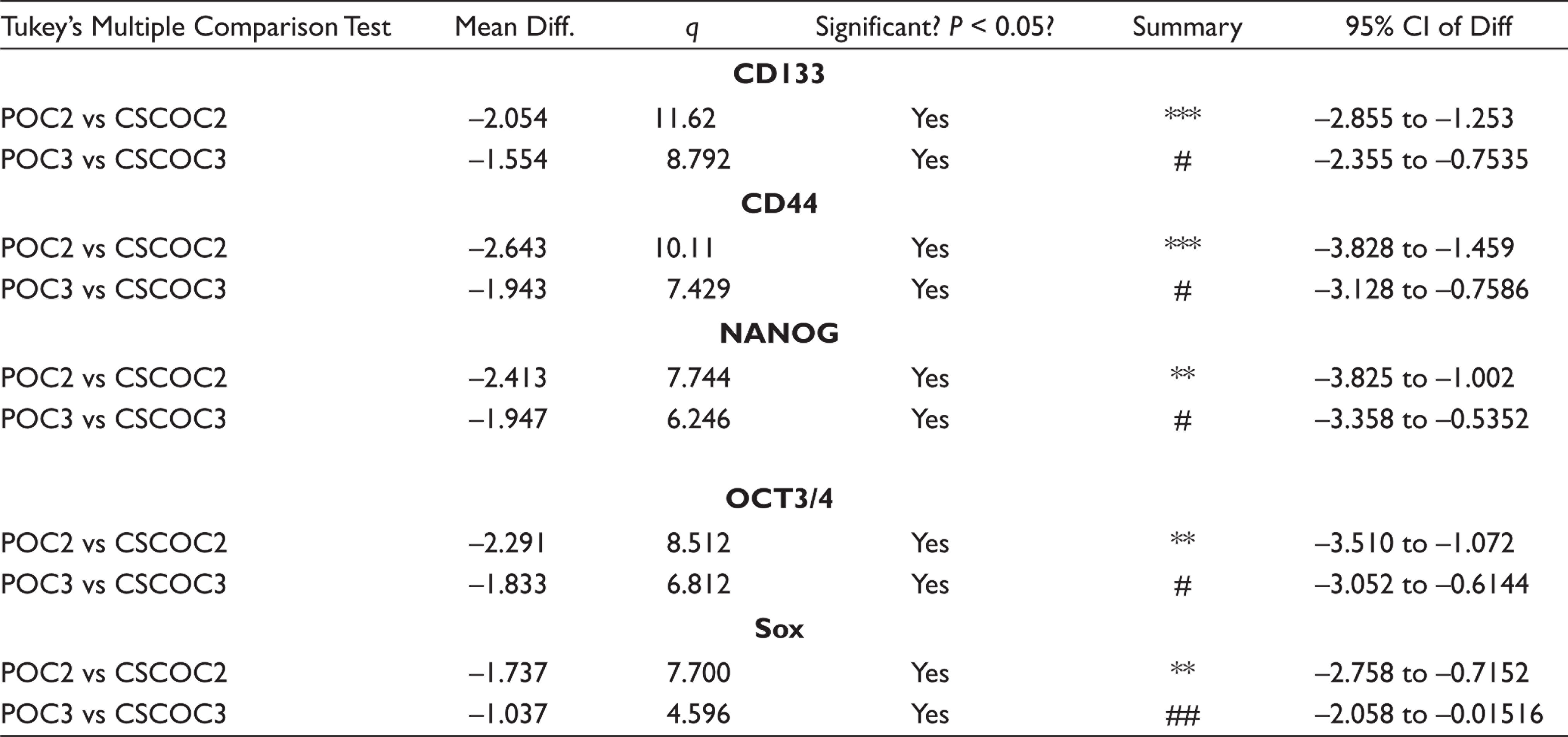
The experiments were carried out three times, and results were expressed as mean and standard deviation. Statistical analysis was performed using GraphPad PRISM software version 5.1 (GraphPad Software Inc., USA) with one-way ANOVA followed by Tukey’s multiple comparison tests. The difference was regarded as significant when *P < 0.05 (significant), **P < 0.01 (moderately significant), and ***P < 0.001 (highly significant).
Results
Sphere Forming Assay
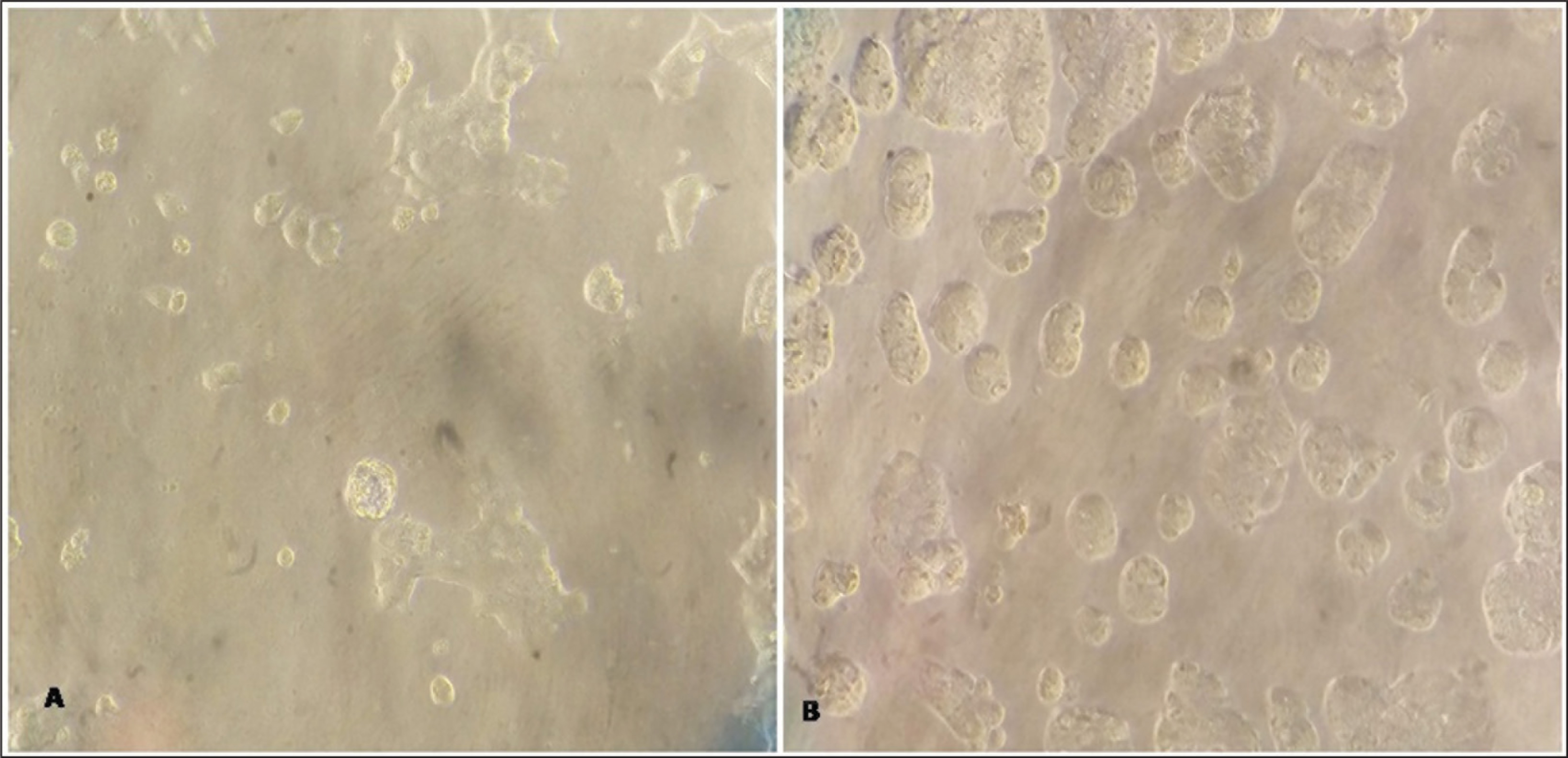
Sphere formation has been used to isolate the CSCs from the primary oral carcinomas cell line. After completion of 2 weeks of incubation, we observed that the sphere formation occurs in all samples as shown in Figure 1.
Indirect Immunofluorescence
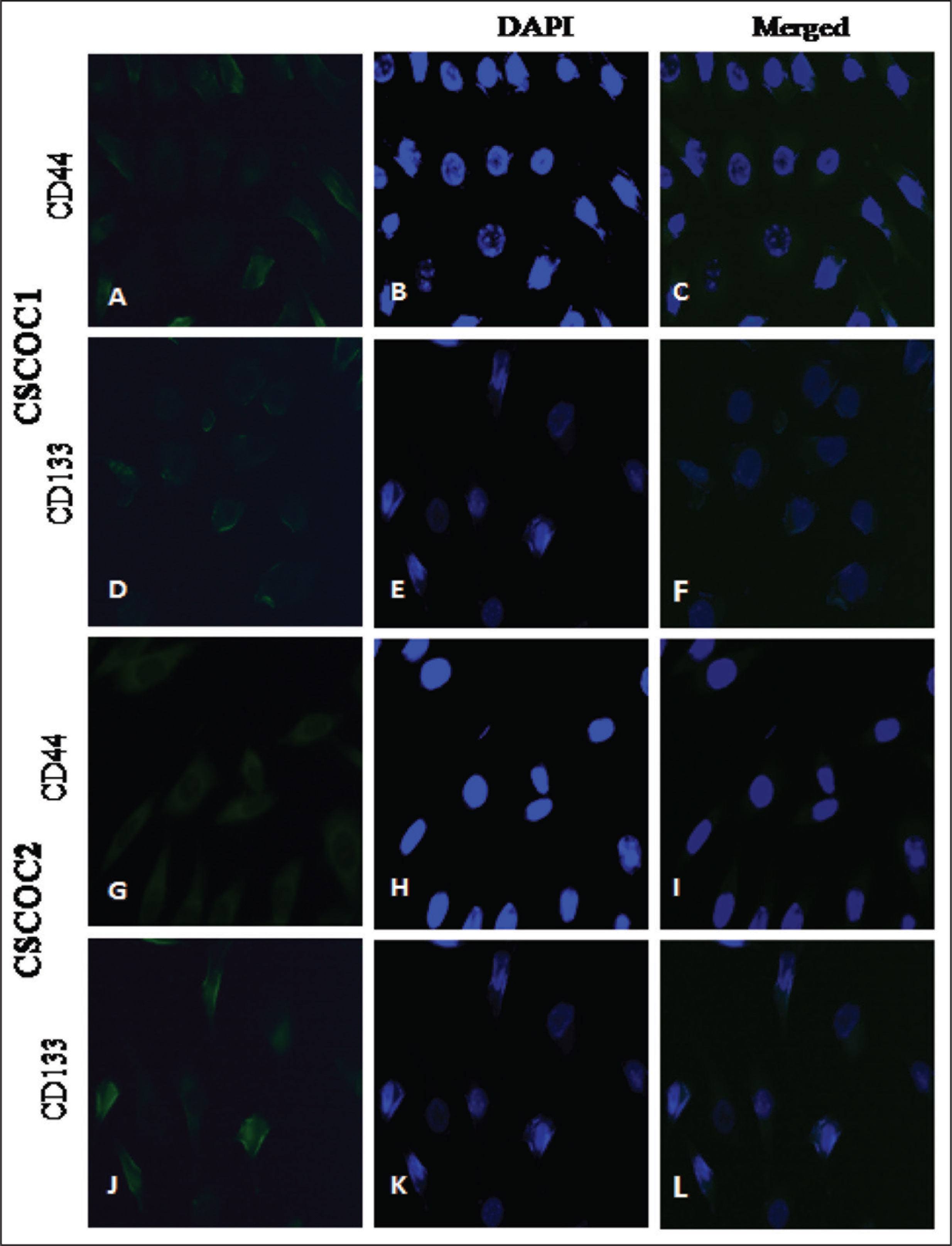
The FITC-conjugated CD133 antibody (green) and APC-conjugated CD44 antibody (red) were used for the confirmation of the presence of CSC by immunofluorescence. The counter-stain DAPI was used to stain the nuclei. The fluorescent distribution pattern of all samples of immune staining confirmed the existence of CD44+ and CD133+ cells as shown in Figure 2.
Microscopic pictures presenting the morphology of tumorsphere: (A) Cells gradually started to detach and started sphere formations in DMEM/F12 serum-free medium with bFGF and EGF; (B) after completion of 14 days incubation, spherical-sized colonies of various sizes and shapes of tumorspheres (20× magnification)
Indirect Immunofluorescence (IF): Immunofluorescent Staining of CD133 Antibody and CD44 Antibody Markers in CSCs Populations (20× Magnification)
Tukey’s Multiple Comparison Tests Table
Real-Time Polymerase Chain Reaction
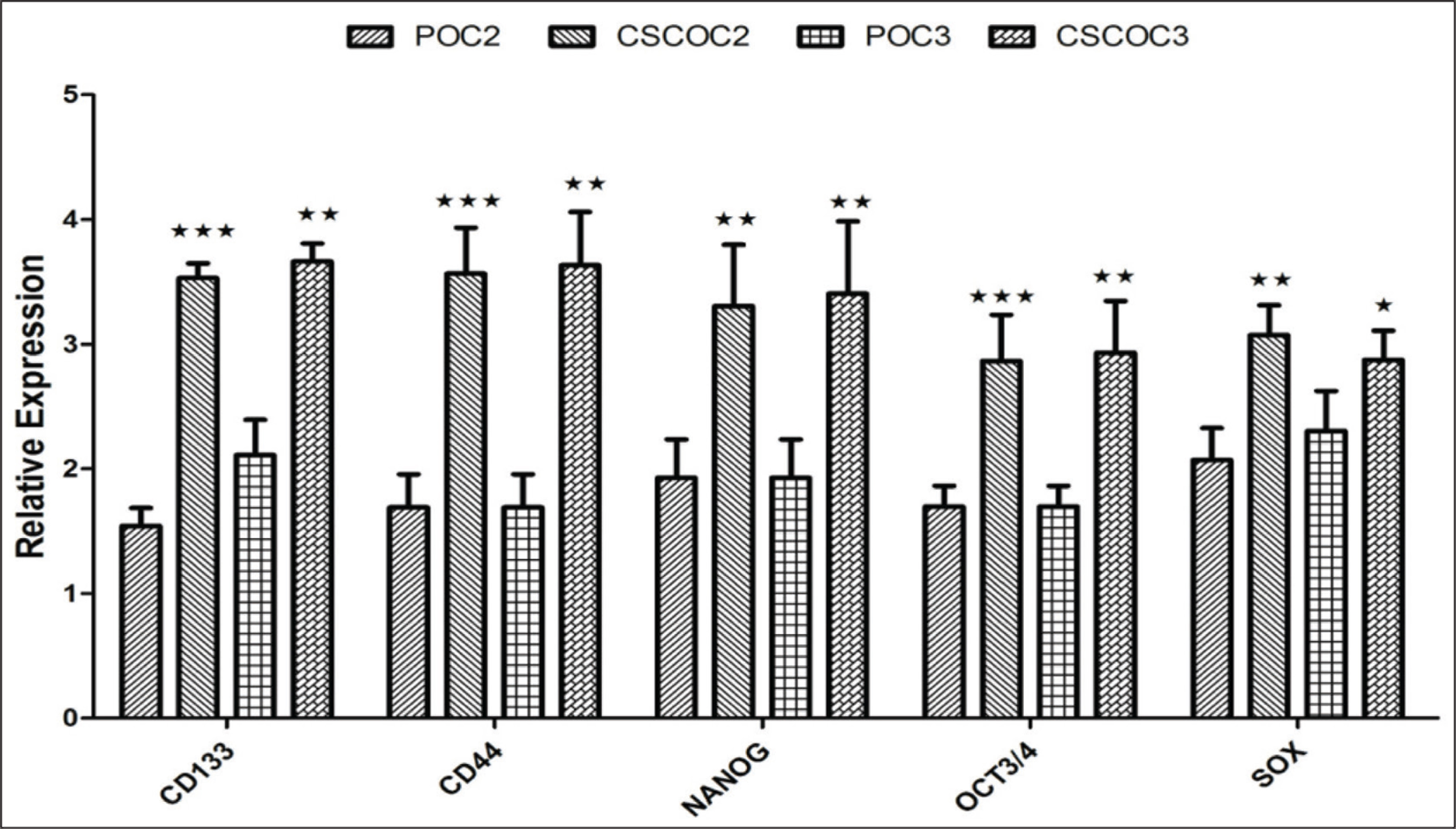
The quantitative mRNA expression of stem cell markers such as CD133, CD44, OCT4, Sox2, and NONG were significantly up-regulated in CSC-enriched subpopulation compared to parental cell lines as shown in Figure 3 and Table 2.
Discussion
The poor clinical outcomes of oral cancer patients are due to the presence of CSCs which are responsible for local recurrence, distant metastasis, and therapeutic resistance. 9 To characterize the CSCs, we need to isolate and culture them. FACS, MACS, flowcytometer machines are used for isolation of these CSCs and these machines are costly with high maintenance. 6 Spheroid or tumorsphere enrichment is a resourceful, cost-effective method to isolate the CSCs and this assay is used regularly to study the stemness, self-renewal, and clonogenicity of CSC. 20
The main features involves the development of tumorspheres in suspension conditions which are resistance to apoptosis and also initiation of stemness-associated pathways. 21 The method includes cells that were cultured in ultra-low attachment plates in serum-free conditions supplemented with specific mitogens, such as EGF and basic FGF. 7 These factors were involved in activating signaling cascade (MAPK, AKT, and JAK-STAT) and transcription factors (Notch, Shh, Oct3/4, and Wnt). These signal cascade and transcription factors are involved in the maintenance of pluripotency and self-renewal of the stem cell population. 22 The tumorsphere formation efficiency of subpopulation CSC of primary oral carcinoma was above 35% after few passages confirm the self-renewal property which is a trademark of CSC. However, this method has several drawbacks due to spontaneous uncontrollable fusion between the spheres and adherent sphere development in the course of culture process resulting in differentiation and instability in sphere shapes and sizes of CSCs. 23 To overcome this, the seeding density of the cells were limited between 1000 and 2000 cells/ml. The most commonly used markers for isolation of CSCs from the primary oral carcinoma are CD133, CD44, and ALDH. A single surface marker approach failed to produce an optimized method of identification and isolation of CSC in HNSCC. Cells have shown two or multiple positive markers with more resistance to treatment and also higher in vivo cultures. 9
In the present study, indirect immunofluorescence techniques with double-positive antibodies (CD133 and CD44) conjugated FITC as secondary antibodies were used for confirmation of CSC.OCT4, NANOG, and Sox2 are regarded as key molecules for self-renewal and helps stem cells population in undifferentiated condition. 14 Vaiphei et al., in 2014, showed that overexpression of Oct4 and Nanog genes, found in CSC-enriched subpopulation derived from HNSCC sphere formation colonies, positively correlated with treatment failure and stage while negatively correlating with differentiation status. 19 OCT4 is one of the embryonic stem cell (ESC) markers which controls the POU domain. It has been involved in the ESC embryogenesis and pluripotency. The study revealed that transformation, tumor originality, invasion, and metastasis of OCSCC is carried by OCT4. 24
The Sox2 is transcription factor which regulates multiple signal transduction pathways. It involves the cancer progression process that included cell proliferation, migration, invasion, stemness, tumorigenesis, apoptosis resistant, and resistance to ChT. 11 Studies have shown that OCT4 and Sox2 expression has been strongly correlated with the presence of CSC subpopulation, in moderately differentiated buccal mucosal SCC (BMSCC) and oral tongue SCC (OTSCC). 11 NANOG is a well-studied ESC marker as well as transcription factor. The higher expression of NANOG occurs in different types of cancers. It is involved in tumor transformation, tumorigenicity, and metastasis within OCSCC. The study showed up-regulation of NANOG in OCSCC tumor samples. 11
In OCSCC and oropharyngeal SCC cell lines, NANOG is overexpressed in the CSC population compared to the parental population. OSCC patients have shown higher expression of Oct4, Nanog, and CD133 with the poorest survival prognosis as confirmed by Singh et al. 2018. 25 CD133, CD44, Oct4, Sox2, and Nanog were expressively higher in CSC-enriched subpopulations in comparison with the parental cell lines.
The concept of CSCs in the role of cancer is developing as evident from increasingly sophisticated research hoards. It is shown that CSCs can retain characteristics analogous to those of normal cells. Hence, monitoring them might provide novel targets and interventions in the treatment of cancer by inhibiting tumor growth and its behavior, thus aiding better treatment, prognosis, and overall survival rate of the cancer patients. 26 There are only limited stem cell markers studied in HNSCC, where the marker positive cells required for tumor formation were higher than other malignancies. Hence, further refinement of CSCs in HNSCC is required. 27 A good understanding of molecular biology of head and neck cancer along with understanding of the CSCs, and their behavior, will definitely contribute future therapeutic improvements like development of nanoformulation to target CSCs. 28 The limitation of this study is the small sample size.
Conclusions
This study showed that cost-effective spheroid enrichment and the indirect immunofluorescence methods may represent an alternative to high-cost machines that were useful for isolation of CSC that can isolate from the primary tumor. Exploration of CSCs provides the ability to produce new cancer targets that can conquer drug resistance and successfully counter tumor cell metastasis. Tumorsphere assay might also guide as a consistent platform for both revolution of anti-CSC vehicles and the advancement of CSC-based drug targeting immunotherapy.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
