Abstract
Aim:
To investigate whether bacteria that play a major role in periodontal disease pathology and in the formation of dental plaque also affect the formation of dental calculus, which is a predisposing factor for the initiation and progression of periodontal diseases.
Materials and Methods:
This was an in vitro study, and cultures of bacteria were obtained from the American Type Culture Collection and Department of Biology, Faculty of Science, Atatürk University. Young cultures of bacteria of Streptococcus mutans (S. mutans), Streptococcus sanguinis (S. sanguinis), Streptococcus gordonii (S. gordonii), Aggregatibacter actinomycetemcomitans (A. actinomycetemcomitans), Porphyromonas gingivalis (P. gingivalis), Fusobacterium nucleatum (F. nucleatum), and Corynebacterium matruchotii (C. matruchotii) were prepared in media containing their specific enriching factors. B2 solid, B4 solid, and B2 liquid media were used to determine active calcification, whereas the mineral salt basal (MSB) medium was used to observe passive calcification. Calcification in the media was measured under light microscopy and in MSB using a spectrophotometer and was recorded as the percent transmittance.
Results:
S. mutans, S. sanguinis, and S. gordonii showed calcification in the B2 medium. S. mutans, S. sanguinis, S. gordonii, and C. matruchotii demonstrated calcification in MSB. A. actinomycetemcomitans, P. gingivalis, and F. nucleatum did not show any calcification.
Conclusions:
It was concluded that streptococci present in dental plaque take part in the formation of dental calculus, whereas periodontopathogens have no role in the formation of dental calculus.
Introduction
Dental plaque is the primary etiologic factor in the development and progression of periodontal diseases. Dental plaque is made up of a biofilm layer containing different types of microorganisms, and colonization follows a regimented pattern with adhesion of the initial colonizers to the enamel salivary pellicle followed by secondary colonization through interbacterial adhesion.1–4 Streptococcus sanguinis (S. sanguinis) is the primary colonizer in dental plaque formation, while Streptococcus mutans (S. mutans) and Streptococcus gordonii (S. gordonii) co-aggregate to the primary colonizers through specific molecular interactions.5,6 The formation of periodontitis, a chronic inflammatory periodontal disease, depends on the presence of specific bacteria rather than the total bacterial load. 7 Aggregatibacter actinomycetemcomitans (A. actinomycetemcomitans), Porphyromonas gingivalis (P. gingivalis), and Fusobacterium nucleatum (F. nucleatum) have been officially designated as major pathogen agents of periodontitis and play important roles in the development of periodontitis.8,9
Calcified dental plaque composed of calcium phosphate mineral salts and surrounded by a nonmineralized bacterial layer is called dental calculus. Dental calculus facilitates bacterial involvement through its porous structure and acts as a predisposing factor in the development of periodontal diseases by causing the release of toxic agents produced by bacteria into the periodontal region.9,10 Several cross-sectional and longitudinal studies have shown the relationship between dental calculus and periodontal diseases.11–13
Dental calculus formation is the result of the deposition of calcium phosphate crystals on the organic matrix from saliva and gingival fluid when dental plaque is not eliminated. 14 Dental plaque provides a base for the formation of calculus by supplying the organic matrix required for the calcification of the deposits. Previous theories regarding the formation of calculus relied on the local increase of calcium and phosphate ions thought to cause the initiation and formation of dental calculus. Currently, the adopted hypothesis is that bacteria contribute to the setup of suitable physical conditions required for the deposition and crystallization of calcium salts in the calcification of dental plaque.15,16
Although it is known that A. actinomycetemcomitans, P. gingivalis, and F. nucleatum play an important role in periodontitis, there has been no study on their potential role in dental calculus formation. The aim of this study was to determine whether these bacteria would form calcification in vitro and to examine the mechanism of action. This study also intended to determine the in vitro calcification of S. mutans, S. sanguinis, and S. gordonii, which contribute to the formation of dental plaque.
Materials and Methods
Setting and Design
The study consisted of an in vitro observation and was conducted from September 2014 to November 2014 at the Faculty of Science and Dentistry of the University of Atatürk. The study was approved by the Local Ethics Committee of Faculty of Dentistry, Atatürk University.
Bacteria and Media
Table 1 lists the bacteria used in this study and their sources. Bacteria were incubated by preparing each young culture in a medium containing its specific enriching factors. For this purpose, an extract of trypticase soy broth with yeast was prepared for the incubation of P. gingivalis, S. mutans, S. sanguinis, S. gordonii, A. actinomycetemcomitans, and F. nucleatum. 17 A brain heart infusion was prepared for the incubation of Corynebacterium matruchotii (C. matruchotii). 18 An anaerobic jar system (Oxoid Anaero Gen3.5L AN0035A; Thermo Scientific, Hampshire, United Kingdom) was used to provide the anaerobic environment for incubation.
Microorganisms
2Atatürk University, Science Faculty, Department of Biology, Laboratory of Molecular Biology and Bacteriology.
B2 and B4 media were selected to enable the observation of active calcification, as discussed in many previous studies.19,20 Mineral salt basal (MSB), a special buffer solution, was used to observe passive calcification. 21 In addition, 15–0.25 percent calcium acetate was added to the three-fourths of the media where the microorganisms showed the best development and were later modified to allow the observation of calcification (modified media). Table 2 lists the contents of the media used.
Inoculation
Bacteria were inoculated onto the solid media through line inoculation in the liquid media using a loop. The fluid form of the B2 medium was used for the liquid media and was left to incubate at 37 °C with a 120 rpm rotation, whereas the solid cultures were incubated without shaking. The cultures were examined under a light microscope (LM) to observe calcification after 3, 5, 7, 14, 28, and 40 days. If the microorganism showed calcification, it was not reevaluated on the next day of examination. However, for microorganisms that did not show calcification, examination continued until the last day. The experiments were conducted in a triplicate under aseptic conditions.
Media and Their Contents
To form nearly a 1 × 108 CFU/ml ratio in MSB, 2 ml of 140 g/l was added in drops to 10 ml of the bacteria suspended from the young cultures, and the turbidity–precipitation formation was observed. A bacteria-free buffer was used as the negative control. The turbidity–precipitation formation was measured using spectrophotometry at a 540 nm wavelength as the percent transmittance.
Results
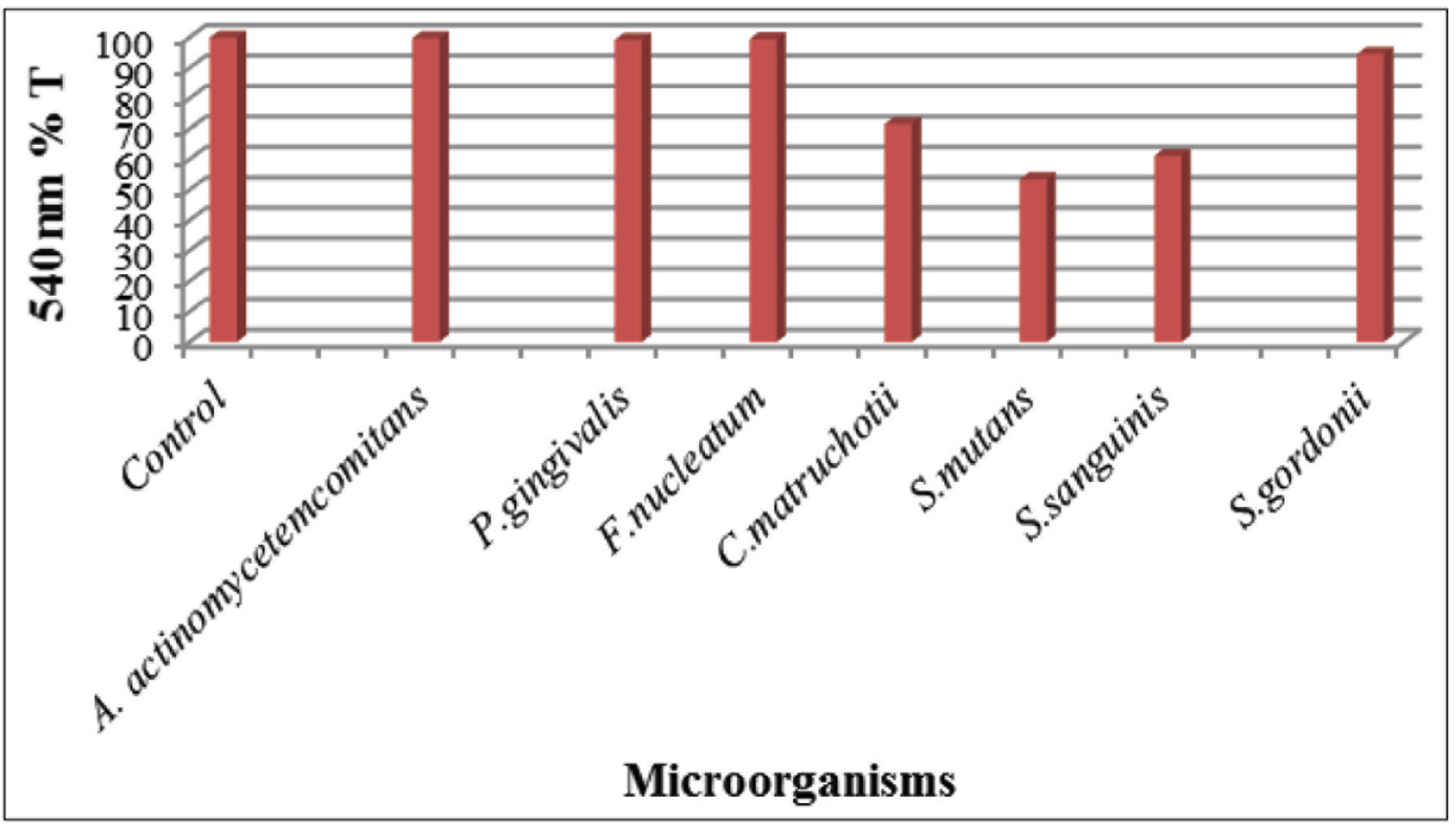
The state of the presence of calcification is shown in Table 3. Figure 1 shows the graph of the results of the experiments using MSB for passive mineralization. Figure 2 shows the appearance of the crystals under the LM in the solid media, whereas Figure 3 shows the data for the liquid medium. Figures 2 and 3 are provided to demonstrate the crystals seen directly under the LM.
In the modified medium, no detection of calcification was performed because the blood and hemin used as enriching agents blocked detection under the LM. Therefore, we could not conclude the presence or absence of calcification for bacteria in the modified medium.
In the B4 medium, all bacteria showed calcification. In the B2 solid and B2 liquid media, S. mutans, S. sanguinis, and S. gordonii demonstrated calcification (Figures 2 and 3). In MSB, C. matruchotii, S. mutans, S. sanguinis, and S. gordonii showed calcification. It was determined that bacteria showing calcification in MSB have a lower transmittance than both the control group and the bacteria that did not show calcification (Figure 1).
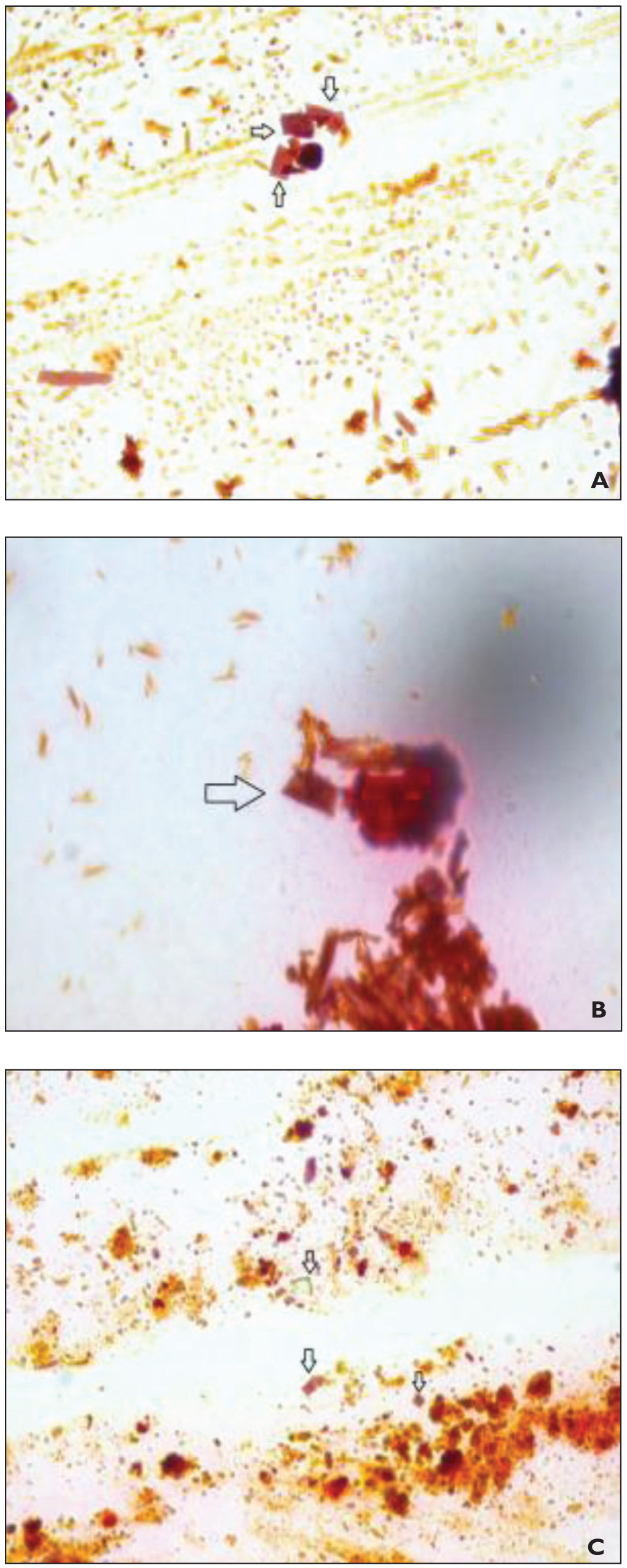
Mineralization on Liquid and Solid Media

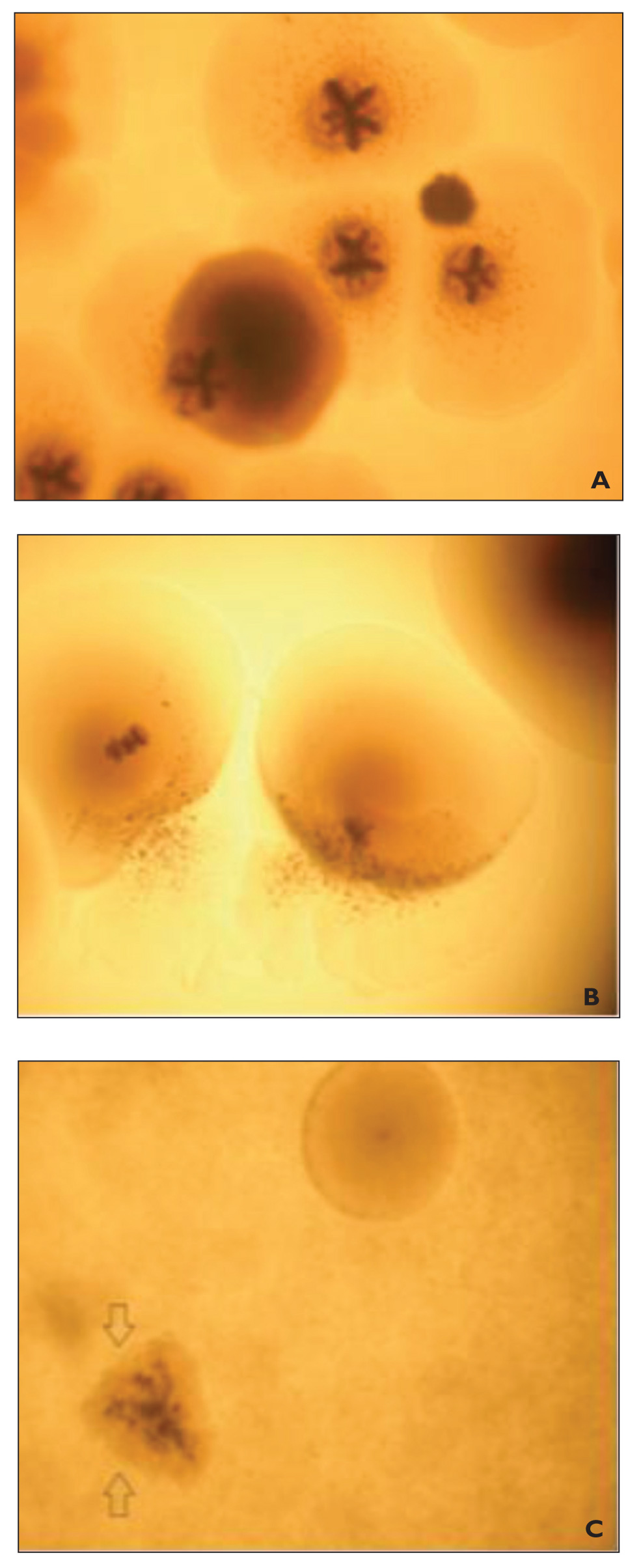
LM Images of the B2 Liquid Medium: (A) Crystals of S. Mutans in the Liquid Medium (100 × 10 LM), (B) Crystals of S. Sanguinis in the Liquid Medium (100 × 10 LM), and (C) Crystals of S. Gordonii in the Liquid Medium (100 × 10 LM). Arrows (") Indicate the Calcification Nuclei of Bacteria
Discussion
Dental calculus forms when calcium and phosphate ions in saliva begin to precipitate and change into a mineral form in the presence of an alkaline medium. The precipitation continues in the presence of calcification nuclei. 22 Bacteria that develop in alkaline conditions are believed to contribute to the formation of dental calculus and pose a risk for periodontal diseases. 23 It is known that bacteria are significant in the formation of dental calculus, whatever their role in plaque formation or the precipitation of calcium and phosphate ions. 13 Studies on the mechanism of dental calculus formation have indeed noted the role of bacteria, but they have not fully explained the relationship between their presence and the development of dental calculus.24–26
In this study, S. mutans, S. sanguinis, and S. gordonii, which are involved in the formation of dental plaque, and A. actinomycetemcomitans, P. gingivalis, and F. nucleatum, called periodontopathogens, were investigated. Calcification studies were conducted for the first time in the present study on the oral bacterium C. matruchotii. 25 Bacteria other than periodontopathogens were also included to test the calcification ability and reliability of the media selected for this study.
Although none of the bacteria showed calcification in the B4 medium, S. mutans, S. sanguinis, and S. gordonii showed calcification in the B2 medium; this medium harbors a higher rate of nitrogen sources with higher nutrient values and is more conducive to bacterial development. In accordance with our findings, it was reported that S. sanguinis increases the pH value of dental plaque, which causes the calcium ions in the medium to precipitate. 27 It was also concluded that S. mutans leads to a decrease in oral pH that aids in calcification formed in the cytoplasm, and calcification began in the extracellular region following the calcification of the cellular wall. 28 This study posits that S. mutans, S. gordonii, and S. sanguinis may be important for the calcification of the molecules they produce as a result of their metabolism or due to an organic molecule present in their cell structure.
It has been reported that C. matruchotii exhibits calcification only in the calcium present in the medium, which is not a suitable environment for sustainability. 29 It was thought that C. matruchotii could not sustain its viability and, therefore, degenerated and calcified. 29 In our study, C. matruchotii was inoculated on a content-rich medium and maintained bacterial viability. Therefore, in the studied medium, bacteria did not degenerate, did not lose their viability, and were not calcified. This inconsistency of the results of the present study with others can be explained as described above.
These study findings could not be subjected to direct comparisons with any earlier similar investigation because, to the best of our knowledge, this is the first study to examine the in vitro calcification of periodontopathogenic bacteria. It was observed that periodontopathogens had no effect on the formation of calcification in all media. In this study, the reason for using a liquid medium was to simulate the increased gingival fluid in the subgingival region in the presence of periodontitis. This study also included B2 as a liquid medium because this medium contains higher amounts of the nutrient factors required by microorganisms. The results obtained from the liquid medium were found to be the same as those from the solid medium. It was also observed in the examination following inoculation that S. mutans, S. sanguinis, and S. gordonii produced calcification in the liquid medium as well as in the solid, whereas A. actinomycetemcomitans, P. gingivalis, and F. nucleatum did not form calcification in the liquid medium. In addition, C. matruchotii, which was detected in the initial calcification studies and, therefore, included in this study as a test bacterium, did not show any calcification in the liquid medium. The reason for these findings may be that all the conditions are suitable for this bacterium to maintain its viability and thus not become calcified. This is the first study to examine the in vitro calcification of related microorganisms in the liquid medium.
The MSB buffer system was used to determine the passive calcification characteristics of microorganisms. This system consists of a phosphate buffer solution containing dipotassium phosphate, monopotassium phosphate, and magnesium sulfate. 29 MSB was chosen as a good buffer to provide the necessary pH stability, and the phosphate present in it combines with calcium to form particles.
CaCl2 was used to form a saturated calcium solution that dissolves well in MSB and produces Ca2+ . The main reason for allowing the ratio of CaCl2 added to the MSB to reach 20% was not to increase saturation excessively to prevent self-precipitation. S. gordonii, S. sanguinis, S. mutans, and C. matruchotii exhibited calcification in the MSB buffer system. Acidic phospholipids present in the cell membrane play a key role in the formation of calcification by C. matruchotii. 30 No calcification could be detected under the LM in the solid or liquid media, as the bacteria were not fully degenerated and calcified. Although calcification started in the cell membrane, the bacteria were able to sustain viability in both the solid and liquid media. In MSB, C. matruchotii showed calcification due to the presence of Ca2+, which causes full degeneration.
Streptococci are calcified in MSB may be because of the precipitation of cell membrane components that bond to free Ca2+. No calcification was observed for A. actinomycetemcomitans, P. gingivalis, and F. nucleatum in the solid or liquid MSB media. These findings suggest that these bacteria did not calcify, and they showed secondary colonization in the channels and lacunae in dental calculus.
Dental calculus develops when nonmineralized biofilms, which are rich in oral bacteria, become mineralized. 31 The complex structure of biofilm, which entraps large amounts of oral bacteria, human proteins, viruses, and food remnants, could not be simulated in this in vitro study. However, we suggest that microorganisms that show active mineralization need a biofilm to maintain their viability, whereas those that demonstrate passive mineralization have no need for a biofilm. Further and detailed studies on this subject are needed to confirm our results.
The study has some limitations. First, the study was conducted in vitro and could not mimic the one-to-one oral environment. The second is that healthy observation could not be made in the modified medium because of the blood and hemin. Despite these limitations, it was concluded that S. mutans, S. sanguinis, and S. gordonii take part in the formation of dental plaque in addition to participating (both actively and passively) in the formation of dental calculus. C. matruchotii was determined to cause only passive mineralization. Periodontopathogenic bacteria could not be shown to participate, either actively or passively, in the formation of dental calculus. According to this result, periodontopathogenic bacteria are located in the channels and lacunae in dental calculus during its formation. These pathogens, which do not play a role in the formation of dental calculus, release toxins into the environment regardless of whether they are present and viable in dental calculus and cause infection to develop, which can lead to periodontitis.
The present study reaffirmed the theory on the effect of bacteria in the formation of dental calculus. Further investigations and research are needed to understand the mechanism of bacterial calcification and to identify other bacteria involved in the calcification process.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Scientific Research Project Fund of Atatürk University under the project number 2013/264.
