Abstract
Aim:
Certain individuals are more prone to dental caries than others are. Caries risk may be related in the secretion of certain defensive salivary proteins including secretory IgA (sIgA) and proline-rich acidic protein (PRAP-1). In this study, we investigated the relationship between PRAP-1 and sIgA leading to the susceptibility of development of dental caries in adults and explored the differences in the levels of sIgA and PRAP-1 between men and women.
Materials and Methods:
Unstimulated saliva samples were collected from 28 patients with high caries risk and 32 control subjects with low caries risk according to caries management by risk assessment guidelines. sIgA and PRAP-1 levels in clarified saliva samples were measured using the enzyme-linked immunosorbent assay.
Results:
According to our results, sIgA and PRAP-1 levels did not demonstrate statistically significant differences as a function of caries risk or gender, even when potential confounding variables such as age and numbers of teeth were taken into consideration. Estimates of effect size, however, revealed small- to medium-sized effects and suggest that significant results may have been found if larger sample sizes were used.
Conclusion:
The results of this study indicate that caries risk and salivary levels of sIgA and PRAP-1 do not appear to be significantly associated. Statistically significant findings could emerge if the sample size was larger.
Keywords
Introduction
Dental caries is a multifactorial disease, requiring bacteria, teeth, time, and fermentable nutrients to initiate and progress.1,2 It has been shown that certain individuals are more prone to dental caries than others. 2 This variability in risk may be due to certain health issues and medications that affect salivary flow and the composition of microbes in the oral cavity 3 in addition to the hardness of one’s teeth and lifestyle habits such as the amount of fluoride exposure and sugar intake.1,2,4 The difference in susceptibility to dental caries can also be related to the composition of saliva and its functioning capability. 5
The composition of saliva includes multiple secreted proteins and compounds that enable us to buffer and control the proliferation of caries-causing microorganisms and prevent the outcome of disease.3,4,6
Proline-rich proteins (PRPs) and secretory IgA (sIgA) are commonly found in the salivary secretions of the oral cavity.3,4,6 PRPs and sIgAs are parts of our intraoral immune defense mechanisms that are supposed to help to prevent intraoral diseases such as caries by limiting the harmful actions of certain bacteria such as Streptococcus mutans and Lactobacilli.7,8
Our oral cavity is populated by a wide range of bacteria, fungi, and microorganisms.1,2 Without a proper immune response, our oral cavity is prone to developing diseases such as dental caries and periodontitis as a result of shifts in the oral microbiome. 9
Dental caries is a unique infection of hard tissue. PRP and sIgA are well-known secretory proteins that are involved with health of oral cavity and are related to their function and quantity. 10 Salivary sIgA is an immunoglobulin that agglutinates cariogenic bacteria and prevents their binding to the tooth/mucosal surface and ease its clearance.4,6
The PRP family of proteins can make up to 80 percent of salivary proteins and is mostly produced by the parotid gland. Similar to sIgA, PRP acts as an aggregative protein and inhibits bacteria binding to the tooth surfaces. PRPs are divided into acidic and basic PRPs.4,6,10 Acidic PRPs mostly act by forming pellicle on an outer tooth layer, thereby forming a diffusion barrier to protect the enamel surface. Acidic PRPs can also bind calcium and apatite crystals to control calcium ion precipitation. 10 Basic PRPs do not bind to the pellicle layer but serve as a protective protein by binding to tannins and bacteria within the oral cavity. 9 In order for caries to progress, it requires specific bacteria on a tooth in the presence of fermentable nutrients for a sufficient amount of time.1,2 When all of these criteria are met, the pH in the oral cavity drops below 5.5 which will allow enamel/dentin demineralization to occur.1,2 Therefore, it is important for sIgA and PRP to be secreted in sufficient quantity to displace bacteria and prevent them from attaching to the tooth. 10
Despite what is known about their potential protective effects, there appears to be a paucity of research that directly examines whether or not levels of sIgA and PRPs have any association with caries risk in adults. As such, this study was undertaken to examine sIgA and acidic PRP levels as a function of caries risk and gender. In general, it was expected that higher caries risk would be associated with higher levels of both sIgA and acidic PRP, while no significant differences as a function of gender would be found.
Materials and Methods
Study Design
A total of 60 adult patients aged between 18 and 70 years volunteered to give saliva samples at the University of Detroit Mercy School of Dentistry, Department of Advanced Education in General Dentistry Clinic. The patients were divided into two categories: high caries risk and low risk caries, according to caries management by risk assessment (CAMBRA) guidelines. A total of 27 patients were diagnosed as having high caries risk and 33 patients were classified as low caries risk in accordance to CAMBRA guidelines.11,12 Caries risk was confirmed using clinical and radiographic measurements. All patients presented with either healthy periodontium or stable periodontal disease. 13 Patients taking pain medications or any medication that may have affected their salivary flow, lost all their posterior teeth, and/or younger than 18 years were excluded from the study.
Saliva Collection
Specimens were collected from patients in resting conditions, with an expected secretion rate between 0.1 and 0.3 mL/min. 14 Unstimulated saliva samples were collected in sterile urine cups during morning appointments. Patients refrained from eating, drinking, or smoking for one hour before their appointment. Saliva was left at the floor of the mouth to be pooled and then collected in intervals of 30 seconds for 10–15 minutes until 5–10 mL saliva was collected. Samples were stored at –80 °C until ready to be tested. Once all samples were collected, 1 mL of each sample was centrifuged at 16,000 × g for 5 minutes to clarify and remove epithelial cell debris, bacteria, and food residues.
Determination of sIgA and PRAP-1 Concentration in Saliva
Clarified saliva was analyzed using commercially available enzyme-linked immunosorbent assay kits according to the manufacturers’ guidelines. Each kit consisted of all reagents needed to evaluate total levels of salivary sIgA (30-SECHU-E01 ALPCO Diagnostics, Windham, NH, USA) and human proline-rich acidic protein kit (PRAP -1) (MBS765465 MyBioSource, San Diego, CA, USA). Absorbance values were collected at 490 nm (Spectra Max M3, Molecular Devices, Sunnyvale, CA, USA) to determine the concentration of the samples. All samples were within the detectable assay range of the sIgA kit (22.2–600 ng/mL) and PRAP-1 kit (78–5,000 pg/mL).
Statistical Analysis
Planned statistical analyses included the following: (a) descriptive statistics (means, standard deviations) for the total pooled sample, males, females, caries risk groups (high vs low risk) and sex by caries risk groups (females with high risk, females with low risk, males with high risk, and males with low risk) on all variables (i.e., age, number of teeth, sIgA, and PRAP-1), (b) bivariate correlations, and (c) inferential statistics including parametric (e.g., analysis of variance) and nonparametric (e.g., Mann-Whitney and Kruskal-Wallis tests) analyses. Analyses of covariance (ANCOVAs) would be used if there were indications that the age and number of teeth may be confounding variables. All analyses were completed using SPSS software.
Results
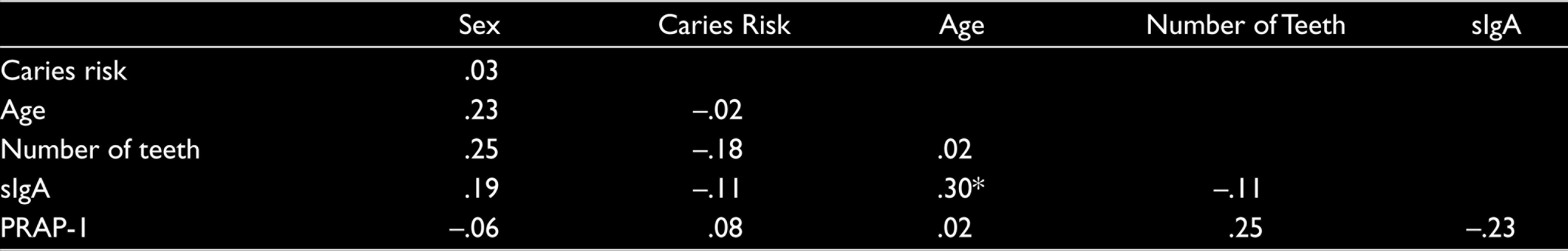
Correlations between all study variables using the entire sample (N = 60) are reported in Table 1. Descriptive statistics for age, number of teeth, sIgA, and PRAP-1 for the entire sample as well as subsamples broken down by sex (male and female), caries risk (low and high) and sex by caries risk (female low, female high, male low, and male high) are given in Table 2. Also reported in Table 2 are the results of one-way analyses of variance (ANOVAs) examining age, number of teeth, sIgA and PRAP-1 as a function of sex, caries risk, and sex by caries risk, respectively.
Examination of the findings reported in Table 1 reveals one statistically significant correlation between age and sIgA (r = .30, P < .05). Although no other significant correlations were obtained, it was noted that correlations between sex and age (r = .23), sex and number of teeth (r = .25), sex and sIgA (r = .19), caries risk and number of teeth (r = –.18), and the number of teeth and PRAP-1 (r = .25) were sufficiently large so as to suggest that the age and number of teeth may need to be taken into account when completing tests of group differences.
Bivariate Correlations Between All Study Variables for Entire Sample (N = 60)
Descriptive Statistics and One-Way ANOVA Results Examining Age, Number of Teeth, sIgA, and PRAP-1 as a Function of Sex, Caries Risk, and Sex by Caries Risk
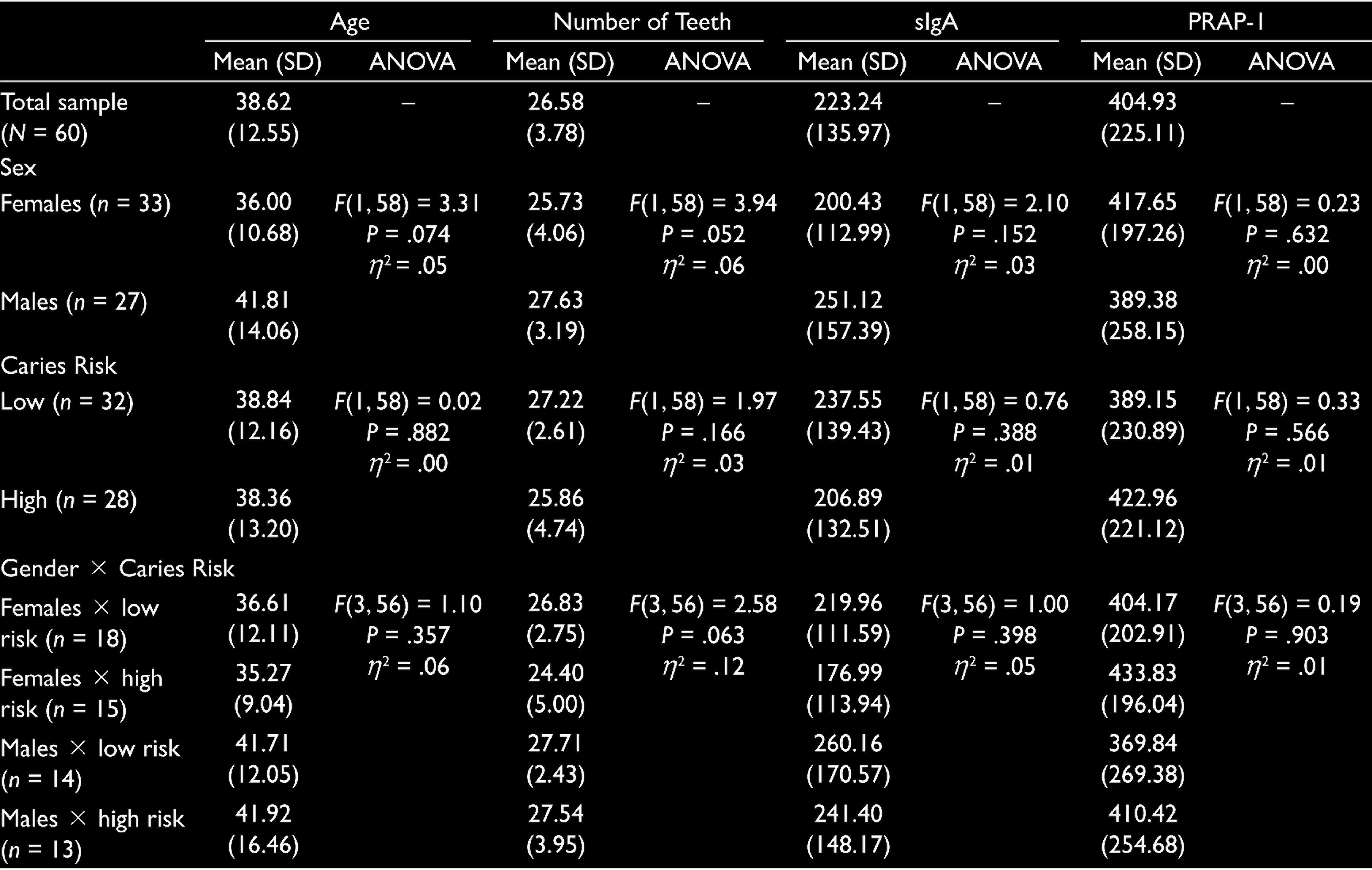
Inspection of the results given in Table 2 reveals that all 12 ANOVAs are not statistically significant. This suggests that the age, number of teeth, sIgA, and PRAP-1 do not vary as a function of sex, caries risk, and the combination of sex and caries risk. Examination of effect sizes, however, reveal small to medium effects in all analyses, except those involving PRAP-1 as a function of sex, and age as a function of caries risk. In both cases, the effect size estimates are less than .00 (reflective of no effect). Given this pattern of results, it was surmised that the nonsignificant findings may be the product of insufficient statistical power due to the relatively small group sizes. The calculation of observed power for all ANOVAs confirmed that the analyses demonstrate low power (e.g., observed power ranges from .039 to .603; minimum acceptable power for statistical analyses is .80).
In addition to the ANOVAs, Mann-Whitney tests with sex and caries risk, and Kruska-Wallis tests with sex by caries risk were also computed. All tests emerged nonsignificant.
Finally, because the age and number of teeth may be influencing the effect of gender and caries risk on sIgA and PRAP-1 levels, ANCOVAs were calculated where sIgA and PRAP-1 were examined as a function of sex (male and female), and caries risk (high and low), while controlling for the age and number of teeth as covariates. For the analysis using sIgA as the dependent variable, nonsignificant main effects for sex (F[1, 54]= 1.93, P = .170, partial η2 = .03) and caries risk (F[1, 54] = 1.25, P = .269, partial η2 = .02) were found. The interaction between sex and caries risk was also found to be nonsignificant (F[1, 54] = 0.28, P = .602, partial η2 = .01).
For the ANCOVA with PRAP-1, similar findings were obtained; main effects with sex (F[1, 54] = 1.38, P = .245, partial η2 = .03) and caries risk (F[1, 54] = 1.06, P = .307, partial η2 = .02) were nonsignificant as was their interaction (F[1, 54] = 0.08, P = .773, partial η2 = .00). The calculation of the observed power revealed that both analyses had low power (e.g., values ranging from .048 to .275).
Discussion
Salivary sIgA and PRAP-1 proteins are considered to be defenses against oral bacteria.10,15,16 These salivary antimicrobial systems act as protective mechanisms against oral bacteria that cause hard and soft tissue destruction.10,16–18 The initiation of caries is associated with an increase in the microbiome.16,17 Thus, it would be expected that higher levels of these two proteins in the saliva should be found as a preliminary reaction to caries. 15
We did not find a positive significant association between the level of dental decay and sIgA, similar to the findings of Shifa et al. 19 However, the patients in the Shifa et al. study were children aged 3–6 years. Conflicting results were reported by Gornowicz et al., 20 who found a positive correlation with dental caries and sIgA. These differences may be due to differences in caries risk assessment (decay-missing-filled [DMF] index vs CAMBRA), patient populations (children vs adults), smaller sample size, and lack of exclusion criteria. 20
In another study, Pandey et al. 21 showed a low level of IgA in patients they considered to be at a high caries risk and suggested low IgA is associated with a high risk of developing dental caries and that this association might be predictive of future caries status. However, this study was done on children having dissimilar caries status, whereas our study consisted of adult patients.
SIgA is the most common salivary antibody in oral cavity. 16 Reduced salivary IgA levels are shown following an increase in the number of decayed teeth,21,22 which suggests it might play a role in the oral cavity defense mechanism.
In contrast to our results, Szkaradkiewicz-Karpinska et al. 10 showed a severe caries group had twice the content of PRAP-1 compared to the minimal caries group. The reason behind it is the chemical structure of PRAP-1 binding to oral bacteria that can colonize the dental surfaces and then causing dental decay. However, that study used the decayed, missing due to caries, and filled teeth that applied to each tooth specifically with the level of PRAP-1 in adult patients, whereas we only measured the number of decayed teeth.
Both the Gornowicz et al. 20 and Szkaradkiewicz-Karpinska et al. 10 studies used the DMF index; however, they did not state why these teeth were missing. Teeth may be missing for a number of reasons not related to caries, including periodontal disease, orthodontics treatment, or trauma. In addition, teeth may be filled for esthetic reasons rather than decay. In the current study, patients with low caries risk according to CAMBRA did not have filling or decay within the last two years.11,12
These saliva samples collected during their 6–12 months recall visit. We used caries risk as a variable that influences the level of sIgA and PRAP-1.
According to the our analyses, gender had no effect on the level of sIgA or PRAP-1, which was consistent with the results of Jafarzadeh et al. 16 who did not find any significant differences in the salivary IgA and IgE levels between females and males. However, other studies done by Eliasson et al. 23 and Prodan et al. 24 showed significant gender differences, with a lower sIgA level in women that in men. These results may be different since these studies had larger sample sizes and the patients were caries free with a healthy periodontium in the Prodan et al. study. 24
Differences in our findings and others may be due to samples sizes and varying caries risk criteria. In addition, few studies have been performed on saliva in adult patients. Additional studies with larger sample sizes, similar patient populations using the same guidelines for caries risk, and sample collection procedures are needed to investigate the relation of salivary proteins to dental decay.
Conclusion
The results of the current study seem to indicate that sex, caries risk, and levels of sIgA and PRAP 1 do not have a statistically significant influence on, or relationship with, each other. However, there is a reason to argue that statistically significant findings could emerge if the sample size was larger, which especially appears to be especially the case for sIgA. We believe that salivary sIgA and PRAP-1 may increase as a result of hard tissue inflammation and infection that occurs in dental decay. Further studies with larger sample sizes and similar methodology are needed to investigate the level of salivary levels of sIgA and PRAP-1 in adult with caries.
Ethics Approval and Consent to Participate
The authors declare that they have obtained the University of Detroit Mercy Internal Review Board (IRB# 1718-46) approval. The consent to participate was part of the IRB application. The participants had to consent in writing before participating in the study.
Footnotes
Acknowledgements
We would like to acknowledge Mr Jamal Alhabeil for his technical help and support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
