Abstract
Background and Aim:
Regeneration potential of platelet-rich fibrin (PRF) and platelet-rich plasma (PRP) in oral tissues regeneration has revolutionized the clinical dentistry in recent years. The in vivo and in vitro research on PRP and PRF has opened a new avenue in the dentistry and medicinal care. It is a minimally invasive and cost-effective technique that will raise the bar of clinical dentistry. The results presently documented are overwhelming; the potential to use PRF and PRP in the clinical dentistry have improved dental quality, satisfaction, and clinical outcome. The aim of the review was to recapitulate the regenerative and healing potential of platelet extracts in different disciplines of clinical dentistry.
Materials and Methods:
The attempt was to answer the following question: what are the various clinical oral health applications for PRF and PRP available in the published literature? The dual functions (tissue sealants and drug delivery systems to carry biomolecules and chemotactic growth factors) have helped the practitioners to treat the complicated oral conditions and tissues regeneration.
The PRF and plasma has emerged as a powerful therapeutic tool for management of soft and hard tissue healing and regenerative procedures, including implant osseointegration. More research is mandatory to use these platelets concentrate to their full potential in various clinical applications.
Conclusion:
PRF is an emerging therapeutic tool in the management of oral soft and hard tissue healing and regenerative procedures.
Clinical Significance
Platelet extracts (platelet-rich fibrin [PRF] and platelet-rich plasma [PRP]) are known for their natural healing potential and have shown promising results in oral surgical procedures, periodontal regeneration, and bone tissues surgery.
Introduction
Platelets are nonnuclear secretory cells derived from the bone marrow that were discovered by Bizzozero in the nineteenth century as protein-producing cells. 1 Upon activation, the platelets produce secretory granules and synthesize proteins. Platelets contain a variety of protein molecules ranging from membrane proteins, cytokines, regulatory proteins, and bioactive peptides. 2 Until now, there is an argument whether to categorize platelets into cell fragments or whole cells. It is a well-documented fact these human platelets initiate and control wound healing through the activation of various biomolecules, including growth factors, adhesion molecules, angiogenesis, proliferation, and activation of associated cells, such as macrophages, fibroblast stem cells, and neutrophils.1,2 A number of key biomolecules and cellular products have been listed below. 3
Platelet-derived growth factors (PDGF)
Interleukin 1
Platelet-derived endothelial growth factors
Basic fibroblast growth factor
Transforming growth factors (TGF-1 and TGF-2)
Vascular endothelial growth factor
These characteristics have attracted researchers to explore platelet concentrates as a tissue regenerative material in modern medicinal and dental care applications.3,4 The regenerative potential of the platelets was introduced in the late 1970s. 5 Further innovations led the categorization of platelet concentrates into platelet-rich plasma (PRP) and platelet-rich fibrin (PRF). These platelet concentrates are suspension of the growth factor in the platelets. 6 The PRP has been readily used in orthopedic applications, 4 in soft tissue injuries, and for oral surgical procedures. 7 PRP works on the mechanism by the activation of the osteoprogenitor cell in host bone and graft. Choukran et al., specifically in oral and maxillofacial surgeries, first introduced PRF as a refined version of the platelet concentrate in the early 2000. 8 It is a matrix of autologous fibrin having more advantages, i.e., easier preparation and no chemical manipulation than PRP. 9 Recently, platelet products have been explored for a range of clinical applications for the oral and maxillofacial surgery. 10 Therefore, focusing on the preparation, advantages, and disadvantages, and the prospects in regeneration of oral tissues in context of clinical applications, the aim of this article is to summarize the regenerative potential of PRF and PRP in various disciplines of dentistry.
Materials and Methods
The attempt was to answer the following question: What are the various clinical oral health applications for PRF and PRP available in the published literature?
Search Protocol
Search strategy included the use of following different terms in combinations: PRP, PRF, oral disorders, oral health, periodontal disease, socket healing, dental implant, osseointegration, regeneration, soft tissue, and hard tissue. The MEDLINE/PubMed, Scopus, and Embase databases were used to search for appropriate research articles from 2000 to, and including, July 2019. The studies with the following criteria were eligible for inclusion: human studies, experimental studies, and intervention: PRF or PRP being used as a treatment modality; reference list of potentially relevant research articles; and articles published only in the English language.
Titles and abstracts of articles were obtained using the search mechanism described above. The author for eligibility initially screened these articles. The full text based on the initial eligibility was evaluated against the criteria and letter to the editors and unpublished articles were excluded. Hand searching was not carried out. The present narrative review followed the scale for the quality assessment of narrative review article guidelines. All the included articles were summarized and the summary data is presented in the concentrated topical headings in the manuscript below.
Platelet-rich Plasma
PRP is a concentrate fibrin glue and an easily available source of growth factors to support hard and soft tissue healing.9,11 The normal composition of blood consists of 94% red blood cells (RBCs), 5% platelets, and 1% white blood cells. In contrast, PRP is composed of 95% platelets. 12 The platelet gel, PRP, is used as a carrier for growth factors in a high concentration on the site of bone defect or on the area requiring augmentation. Once the platelets in the PRP gets activated, it releases growth factors, which play a vital role in rapid wound healing. 11 PRP plays a dual role: act as an adhesive graft material and a hemostatic. The concentration of platelets in the PRP ranges from 500,000 to 1 million µL. 11 In terms of components, the PRP comprised of growth factors (PDGF and TGF) and alpha granules of platelets (vitronectin, fibronectin, plasma adhesion molecules, and fibrin).13,14 There are two methods available for the preparation of PRP: general purpose and platelet-concentrating cell separator. 12
General Purpose Cell Separator
This technique requires hospital settings in which a large amount of blood (approximately 450 ml) is taken from the host in a bag containing anticoagulants, such as citrate–phosphate–dextrose. 9 Initially, the blood is centrifuged at speed of 5600 revolutions per minute (rpm) to separate plasma poor platelet (PPP) and PRP from RBCs.5,11 Now, again this blood is centrifuged at a low speed of 2400 rpm to allow further separation of PRP from the RBCs and the leukocytes. After obtaining around 30 ml of the PRP in this technique, the remaining RBCs and PPP can be returned back to the patient’s circulation.5,11
Platelet-concentrating Cell Separators
This method for preparation of PRP is almost the same as the general purpose cell separator; however, it differs in terms of the centrifugation speed and the duration. Initially, the venous blood is taken and mixed with an anticoagulant to avoid activation and degranulation of the platelets.5,12 Now, this blood is given a soft spin in a centrifugal machine, which separates blood into three layers. Bottom most layers consist of blood, 55% middle buffy layer of PRP contributing 5% and acellular topmost layer of 40% poor plasma protein (PPP). Now, the operator transfers this mix into a sterile syringe without anticoagulant. This sterile syringe goes through centrifugation at a high speed.7,12 This process allows the PRP to settle at the bottom with only few RBCs. The rest of the acellular PPP (the top layer) is discarded. The remaining (PRP) is shaken well followed by the incorporation of thrombin and calcium chloride prior to the application.5,7,9
PRP for the Regeneration of Oral Tissues
Clinical evidences highlighting the current and potential role of PRP for the regeneration of various oral and dental tissues has been discussed.
PRP in Regeneration of Periodontal Tissues
The periodontal regeneration is mainly though the guided tissue regeneration that uses various membranes’ barriers and biomolecules to promote the regeneration of periodontal tissues. 15 Several studies given in Table 1 have explored the PRP for periodontal regeneration; however, the analysis concluded that the data is heterogeneous and evidences supporting the periodontal regeneration are inadequate.16,17 Mechanism of PRF in mediating the inflammatory and healing processes in periodontal conditions are yet to be studied on cell-based models. 16 More clinical studies evaluating these paradoxical effects of the PRP in the periodontal regeneration needs to be performed.16,18,19,20
PRP in Implant and Osseointegration
There is a strong association of PRP for improving the soft and hard tissues regeneration and healing potential (Table 2). PRP can be employed as a valid adjunct to many procedures in the oral and dental surgery. The PRP in the alveolar socket after tooth extractions is certainly able to improve soft tissue healing and positively influence the bone regeneration.21,22,23 However, the efficiency of the PRP in regeneration is still limited. More clinical studies are required in order to define the proper outcomes related to the PRP in the tooth extraction.
Studies Reporting the Outcome of PRP for Periodontal Regeneration

PRP in Soft Tissues and Bone Tissue Surgery
PRP has shown a strong effect in the management of fractures, bone augmentation, mandibular reconstruction surgeries, implants, and sinus lifting (Table 3). The performance of the PRP in combination with biomaterials is promising, but the results are dependent on the type of biomaterial used for these applications.
PRP and Implant Osseointegration
A lack of evidence exists on the use of PRP during implant placement and peri-implantitis defects. 24 A study by Georgakopoulos et al. 25 highlights that the PRP placement around the dental implant improves the bone formation, resulting in better implant stability and treatment outcomes. Furthermore, a study by Thor et al. highlights the improved implant stability and the survival rate of the implant when used along with the PRP. 26 The PRP exhibits early implant stability and loading. 27 The efficiency of the PRP in implant dentistry is still limited. More clinical studies are required in order to define clinical relevance of the outcome measurements related to the PRP and the implant (Table 4).
Studies Reporting the Outcome of PRP for Alveolar Bone Regeneration
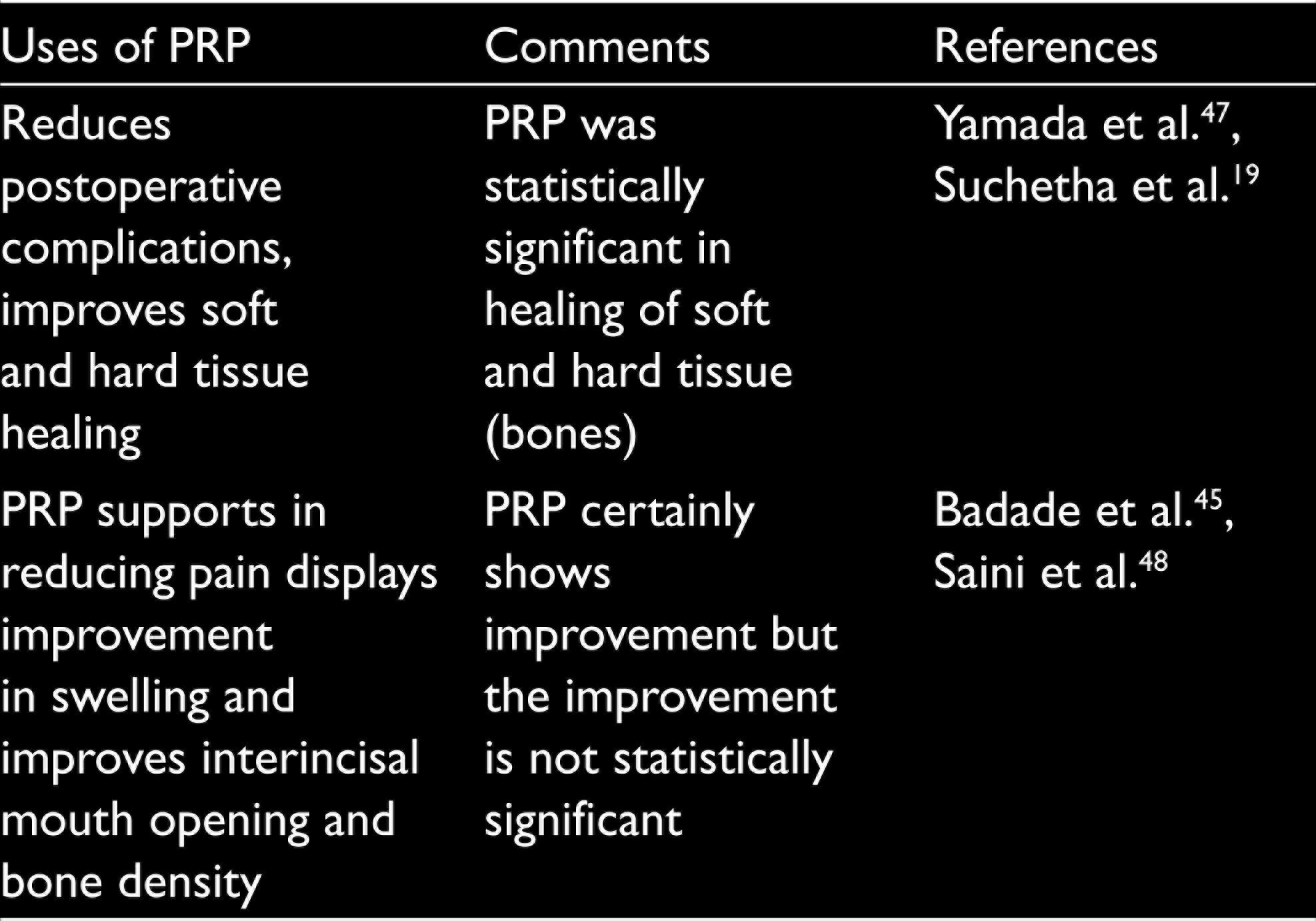
Various Studies Reporting the Outcome of PRP for Soft Tissues and Bone Regeneration
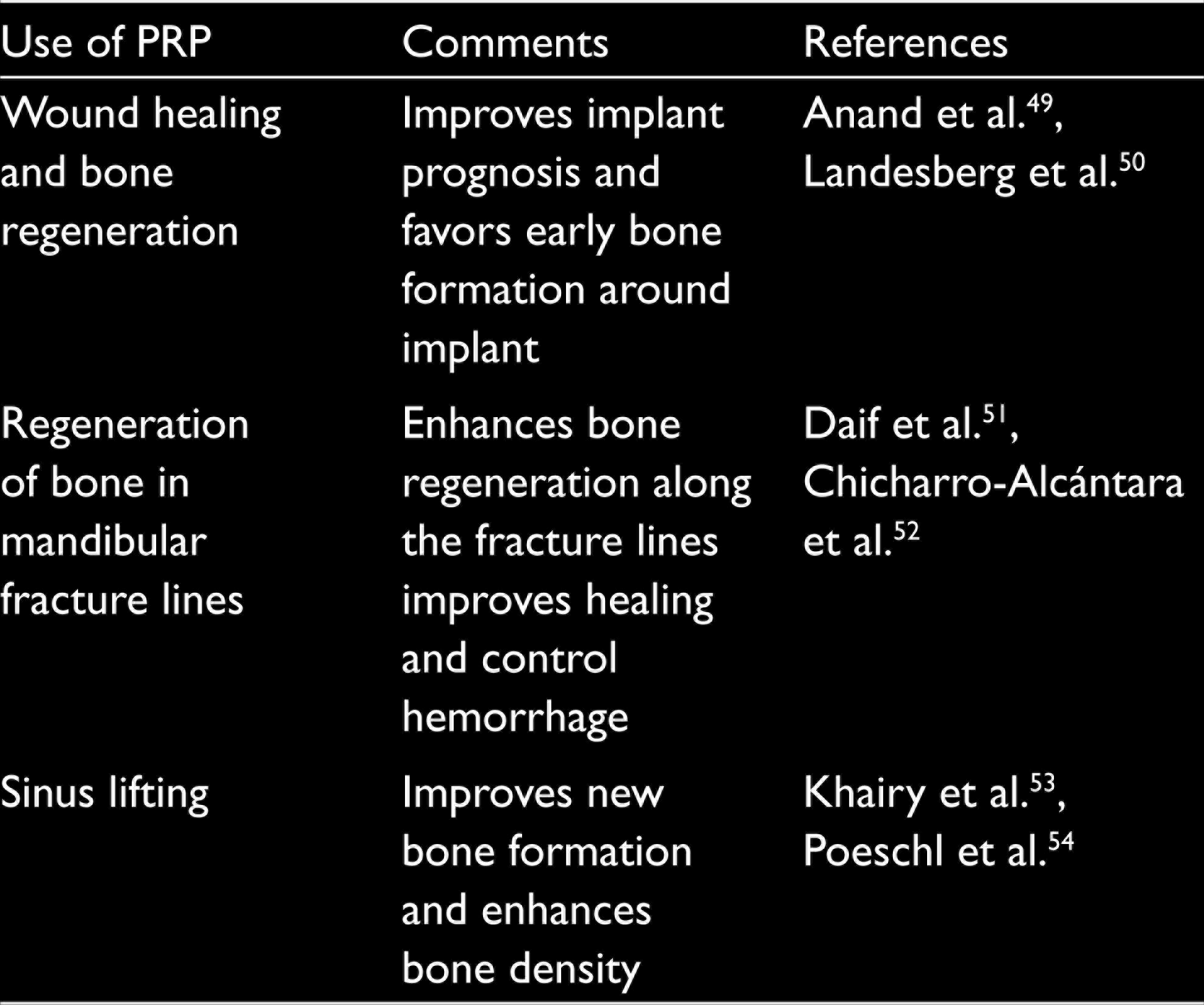
Various Studies Reporting the Outcome of PRP for Implant Surgery
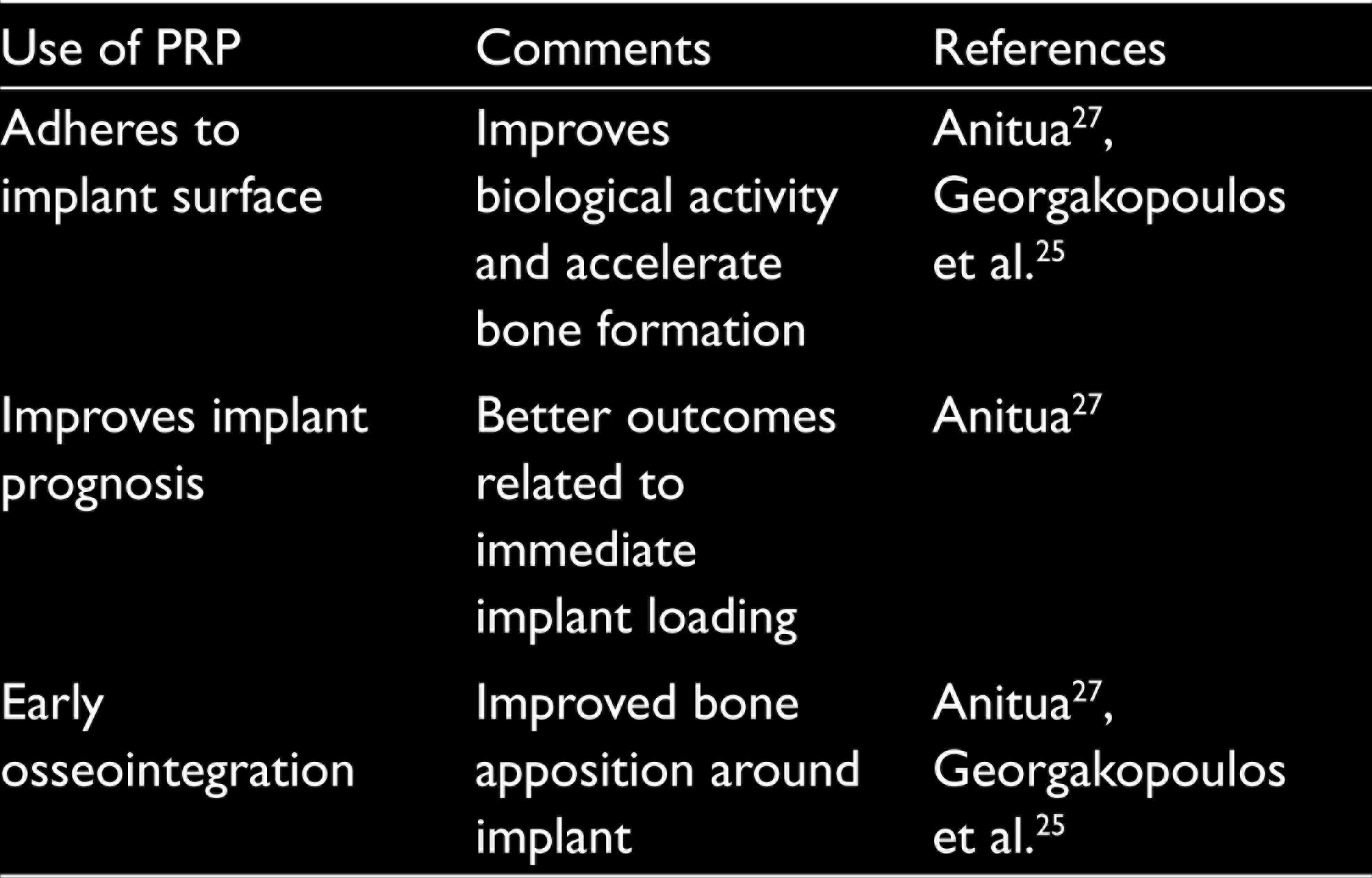
Contraindication of Using PRP
There are a number of medical conditions restricting the use of PRP, for instance, in patients with bleeding and hematological diseases. Bovine thrombin used in the PRP may lead to the formation of antibodies against a number of clotting factors (V and XI) and thrombin, which may result in complicated coagulopathies. 28
Platelet-rich Fibrin
PRF is a second-generation platelet concentrate that is composed of cytokines, leukocytes, stem cells, platelets, and a tetra molecular structure, which act as a scaffold, supports microvascularization, and works as a transport for carrying cells, which are important for tissue’s regeneration. 29 This strong fibrin has comparatively better mechanical properties and slower remodeling properties, which resemble to blood clot. The PRF has remarkable regenerative properties for soft and hard tissues without inducing any kind of inflammatory reactions. 30 Dohan et al. demonstrated the antibacterial and immunological properties of the PRF, which may lead to degranulation of leukocytes and activate angiogenesis.14,31
PRF over PRP
While comparing PRF and PRP, the key advantages of the PRF are easy manipulation, cost effectiveness, no biochemical handling of patient’s blood samples, supportive to the immune system, and relatively better wound healing potential due to lower degree of polymerization. In addition, using the PRF involves lower risks than the PRP due to absence of bovine thrombin and anticoagulants and better efficiency for cell migration and proliferation. 32 The PRF reduces patient’s discomfort during early period of wound healing and can be used alone or in combination with various bone grafts. 33 The fibrin matrix in the PRF is better organized and thus it is more efficient in direct stem cell migration, bone healing, hemostasis, wound healing, and graft stabilization. Release of growth factors in vitro and in vivo results put forward the use of the PRF clinically. 34 Dohan et al. 14 through in vitro studies claimed that the PRF has better healing properties over the PRP. Similarly, Wiltfang et al. from different clinical trials have demonstrated that the PRF shows promising results over the PRP. 35 A study by Kawamura and Urist reported that PRF works as a supportive matrix for carrying morphogenetic proteins. 36
Preparation of PRF
The methodology for the synthesis of PRF has been established. Briefly, venous blood (5 ml) is drawn into two sterile vacutainer tubes without anticoagulant and bovine thrombin.31,37 These tubes are centrifuged using 2700 to 3000 rpm for 10 minutes. This process divides the venous blood into three distinct layers: the uppermost straw-colored acellular plasma layer, the middle layer containing the fibrin clot, and the bottom layer containing the RBCs. 38 The uppermost acellular plasma layer is removed and the middle layer below 2 mm of dividing line, which is PRF, is collected. There is a certain limitation of this approach; since there are no anticoagulants, the clinical success of this procedure is dependent on speedy blood collection and centrifugation well before the activation of clotting cascade. 39 It is reported that the fibrin clot is transformed into a membrane by compression between sterile gauzes. 40 Other noticeable disadvantages of the PRF included requirement of faster handling for prognosis and limited quantitative of autologous blood. 9 The synthesis of the PRF is a technique of sensitive procedure and require trained professionals. 41 Glass tubes are necessary for the propagations of clot polymerization. 42 Considering the favorable features of the PRF, a number of studies have explored it for various tissue regeneration applications in dentistry (Table 4). Although the outcome of most of the studies is very promising, further research and more clinical trials are needed in order expand its applications in dentistry (Table 5).
Various Studies Reporting the Usage of PRF in Other Dental Applications

Conclusions and Future Prospects
The PRF and plasma have emerged as a powerful therapeutic tool for management of soft and hard tissue healing and regenerative procedures, including implant osseointegration. The dual functions (tissue sealants and drug delivery systems to carry biomolecules and chemotactic growth factors) have helped the practitioners to treat the complicated oral conditions and tissues regeneration. The in vivo and in vitro results have opened a new avenue in the dentistry and medicinal care. This minimal invasive and cost-effective technique will raise the bar of clinical dentistry practiced at present. Still, more research is required to use these platelets concentrate to their full potential for various clinical applications.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
