Abstract
Introduction:
The risk of developing deep vein thrombosis (DVT) is high even after the period of bed rest following major general surgery including total joint arthroplasty (TJA). Mobile intermittent pneumatic compression (IPC) devices allow the application of IPC during postoperative exercise. Although ambulation included ankle movement, no reports have been made regarding the effects of IPC during exercise, including active ankle exercise (AAE), on venous flow. This study was performed to examine whether using a mobile IPC device can effectively augment the AAE-induced increase in peak velocity (PV).
Methods:
PV was measured by Doppler ultrasonography in the superficial femoral vein at rest, during AAE alone, during IPC alone, and during AAE with IPC in 20 healthy subjects in the sitting position. PV in AAE with IPC was measured with a mobile IPC device during AAE in the strong compression phase. AAE was interrupted from the end of the strong compression phase to minimize lower limb fatigue.
Results:
AAE with IPC (76.2 cm/s [95%CI, 69.0–83.4]) resulted in a significant increase in PV compared to either AAE or IPC alone (47.1 cm/s [95%CI, 38.7–55.6], p < 0.001 and 48.1 cm/s [95%CI, 43.7–52.4], p < 0.001, respectively).
Discussion:
Reduced calf muscle pump activity due to the decline in ambulation ability reduced venous flow. Therefore, use of a mobile IPC device during postoperative rehabilitation in hospital and activity including self-training in an inpatient ward may promote venous flow compared to postoperative exercise without IPC.
Conclusion:
Use of a mobile IPC device significantly increased the PV during AAE, and simultaneous AAE with IPC could be useful evidence for the prevention of DVT in clinical settings, including after TJA.
Keywords
Introduction
Among all types of major general surgeries, the risk of venous thromboembolism (VTE) is highest in major orthopedic surgery, particularly total hip arthroplasty (THA), total knee arthroplasty (TKA), and hip fracture surgery. 1 There have been a number of reports regarding the beneficial effects of pharmacological prophylaxis for VTE. However, such treatment sometimes causes serious bleeding due to its anticoagulant effect. 2 The risks associated with pharmacological methods can be avoided by using physical prophylactic methods, such as intermittent pneumatic compression (IPC), for VTE in patients at high risk of bleeding. 3
Physical prophylactic methods, including active ankle exercise (AAE) and IPC, have been reported to be efficacious in preventing venous stasis and VTE events. 2,4 –6 Several previous studies have shown that physical methods have the potential to reduce venous thrombotic events. 7 –10 In clinical settings, the use of a mobile IPC device has the potential for safe VTE prophylaxis in patients who have undergone total joint arthroplasty (TJA) and are at high risk of both venous thrombosis and bleeding. 11 The number of VTE events was significantly reduced by using a mobile IPC device in comparison with a non-mobile IPC device, 12,13 and such treatment is recommended in the Guidelines of the American College of Chest Physicians (ACCP) (9th edition). 1 A previous study compared the effects of a mobile IPC device and low molecular weight heparin for prophylaxis against VTE and deep vein thrombosis (DVT) in patients undergoing THA. The results indicated lower rates of venous thromboembolic events with both methods, 14 Furthermore, the mobile device also allowed the application of IPC during exercise, including ambulation and AAE. The risk of DVT is high after TJA and HFS, even after leaving the bed, and ambulation is generally recommended as a preventive measure in such patients. 2 As previous studies have demonstrated a relationship between plantar flexion torque and ambulation ability, including gait speed and stride length, DVT can also develop in walking patients due to a decline in ambulation following orthopedic surgery. 1,15,16 Therefore, the mobile IPC device would likely be beneficial for the prevention of DVT in walking patients. However, there have been no previous reports regarding the effects of IPC during exercise, such as AAE or ambulation, on venous flow.
This study was performed to examine whether IPC can augment AAE-induced increases in peak velocity (PV) in the femoral vein and thus prevent VTE and DVT.
Materials and methods
Study participants and characteristics
This study was approved by the institutional ethics committee. This prospective single-arm intervention study, with one experimental and two control conditions [AAE (heel raise) with IPC, AAE (heel raise) alone (AAE), and IPC alone (IPC)] in randomized order was performed in accordance with the principles of the Declaration of Helsinki. The study population consisted of 20 healthy young adult men and women (10 men and 10 women). Patients with a history of thrombophlebitis, arteriosclerosis obliterans, any thromboembolic events, bone fracture or inflammatory disease in the lower limb or pelvis, malignant tumor, cerebrovascular disease, heart failure, respiratory illness, kidney failure, or sensory disturbance in the lower limb were excluded. All eligible participants received standardized verbal and written information about the trial. Age, height, body weight, body mass index, and calf circumference were determined as basic characteristics (Table 1).
Subject characteristics.
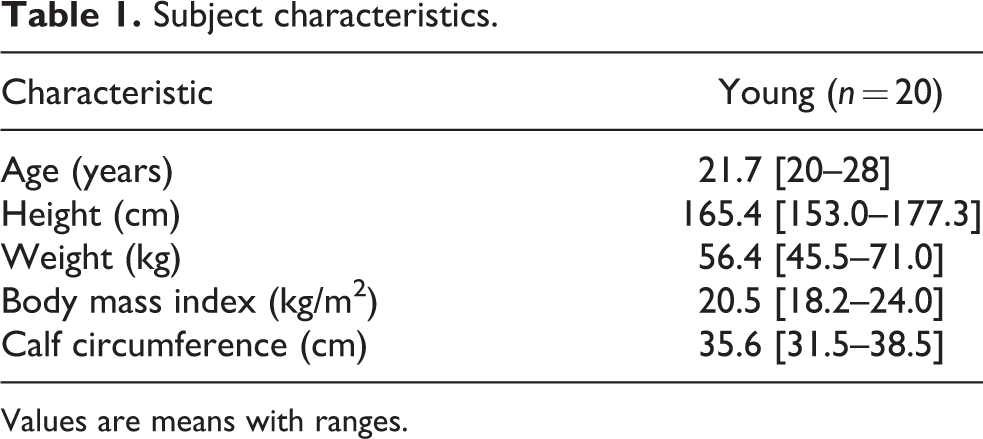
Values are means with ranges.
Measurement of blood flow with doppler ultrasound
Pulsed Doppler ultrasound was performed to measure PV in the femoral vein using a 7.5 MHz linear probe (EUP-L75; Hitachi Medical Corporation, Tokyo, Japan) interfaced with an ultrasound unit (HI VISION Avius; Hitachi Medical Corporation). First, the probe was placed vertically on the right thigh of the medial surface to display a cross-sectional image representing the superficial femoral vein, and defined using the color Doppler method. Then, the probe was turned on the surface of the thigh to display a longitudinal sectional image of the vein. Continuous waveforms were recorded for 8 s using the pulse Doppler method, and PV was obtained using the auto trace mode (Figure 1). The blood velocity in the femoral vein was measured at the front of the thigh, approximately 3 cm distal to the deep femoral vein junction. A Doppler angle relative to the femoral vein of <60° was maintained. The measurement point was marked with tape to allow measurement at the same point for each subject. To suppress motion artifacts in the ultrasonography, especially during AAE, an ulnar side of the examiner’s hand holding the probe was placed near the measurement point, avoiding strong compression on the femur. We considered the sample volume to be 80% of the apparent vessel diameter. One examiner performed all venous velocity measurements. The subjects were instructed to breathe naturally and refrain from taking deep breaths during the measurement to prevent marked respiratory changes in venous blood velocity. All participants wore shorts that did not put pressure on the lower limbs. Femoral vein blood velocity was then measured under one experimental and three control conditions: AAE (heel raise) with IPC, rest, AAE (heel raise) alone (AAE), and IPC alone (IPC).
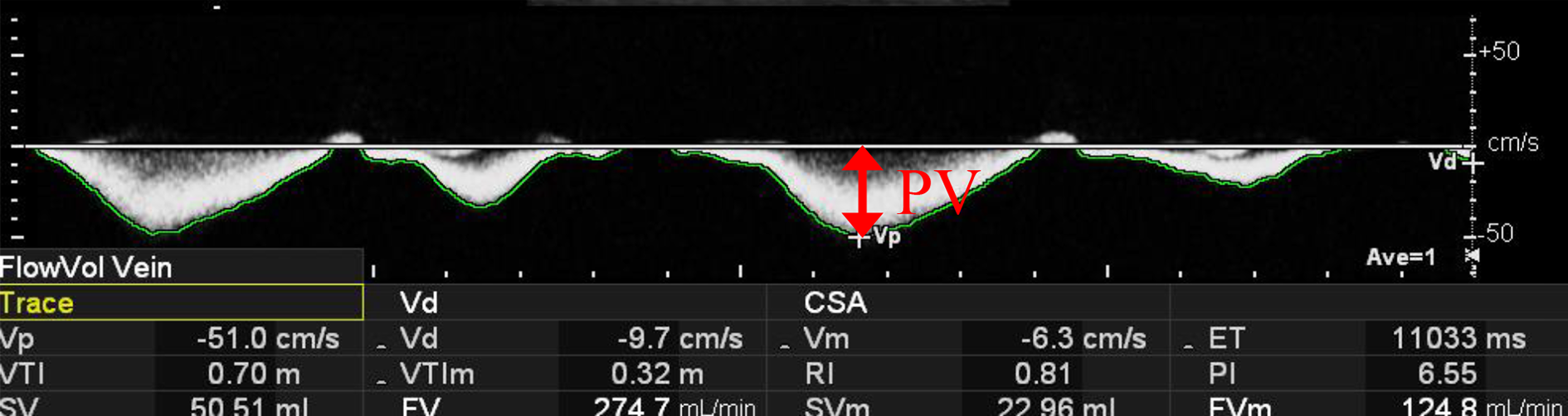
Continuous waveforms were recorded for 8 seconds using the pulse Doppler method and the peak velocity (PV) was obtained using the auto trace mode.
Procedures and measurements on each condition
The mobile IPC device used in this study (ActiveCare; Medical Compression Systems, Or Akiva, Israel) has a cycle of 60 s in which it compresses the lower legs afferently for 10 s each time with low pressure and high pressure, instantly decompresses, and maintains no pressure for 20 s after each compression phase. The subjects were instructed to relax without talking in a loud voice, to not fall asleep, and to minimize contraction of the muscles of the whole body as much as possible during the protocol.
AAE was defined as plantar flexion of the right ankle (heel raise) in a sitting position. The ankle was set at a neutral position at the start, and participants performed ankle movement from the neutral to the plantar-flexed position and back to the neutral position over a period of 2 s. As the following description of a blood flow measurement protocol of AAE with IPC, AAE was performed for 20 s in 1 min. The PV was obtained within 10 s and 10 s after the commencing of AAE until completion.
In AAE with IPC, AAE was commenced at the end of the weak compression phase of the mobile IPC device, and blood flow velocity was measured during AAE in the strong compression phase. AAE was continued until completion of the strong compression phase, measured once every 2 seconds, and then interrupted to minimize lower limb fatigue (Figure 2(a)).

Schematic of experimental protocol for blood flow measurement in active ankle exercise with intermittent pneumatic compression (a) and blood flow measure (b). Top, compression consisting of one cycle using the intermittent pneumatic compression device; bottom, active ankle exercise. Blood flow measurements were defined as the mean of three values.
Protocol
The protocol used in the present study is schematically shown in Figure 2(b). Baseline PV was measured after a 10-minute rest in the sitting position to allow steady-state blood flow to be reached. The baseline values were defined as the mean of three values taken when the measurement was within ±5% of the previous value. PV was measured in the first performing condition, after which the participant rested in the same position to allow sympathetic nervous activity to return to baseline. We obtained 3 PV for each condition, and statistical analyses were performed using the means of the 3 PV measurements. Once it had been confirmed that PV had returned to baseline, measurements were obtained in the second performing condition. Subsequent trials were performed in a similar manner. The order of conditions was selected based on a random number table for each participant to eliminate the potential for order effects.
Statistical analysis
Analyses were performed using the means of three PV (cm/s) measurements and the percentage increase in PV. One-way repeated measures analysis of variance (ANOVA) was performed, using the Greenhouse–Geisser correction to adjust for sphericity when Mauchly’s sphericity test revealed departure from sphericity, with Scheffé’s adjustment to compare the main effects of the different conditions on PV. All analyses were performed using IBM SPSS Statistics for Windows, ver. 24.0 (IBM Corp., Armonk, NY, USA). In all analyses, p < 0.05 was considered to indicate statistical significance.
Results
PV analyses were performed using data obtained from all 20 participants. Table 2 presents the PV in the femoral vein for all experimental and control conditions. Mauchly’s sphericity test showed departure from sphericity (p = 0.001). Greenhouse–Geisser epsilon was 0.73, correcting the degree of freedom of within-subjects factor from 3 to 2.20. Based on this correction, the main effect of the conditions for PV was significant (F = 93.47, p < 0.001). PV was significantly higher under the AAE and/or IPC conditions compared to the baseline values at rest. In the AAE with IPC condition, PV was significantly higher than those with either AAE or IPC alone. There was no significant difference in PV between AAE alone and IPC alone.
Peak velocity at rest and under three conditions.

The values are the means and 95% confidence intervals. All p < 0.001 between all conditions except between AAE and IPC (p = 0.99).
* AAE, active ankle exercise.
† IPC, intermittent pneumatic compression.
Figure 3 presents images of the typical wave forms of venous velocity during AAE in the sitting posture AAE alone, IPC alone, and AAE with IPC.

The typical wave forms of venous velocity during AAE in the sitting posture (a) AAE alone, (b) IPC alone and (c) AAE with IPC.
Discussion
The present study was performed to examine whether IPC could combine additively with exercise to promote venous flow. The experiments presented here indicated that PV in the femoral vein was higher with the combination of AAE and IPC than with either AAE or IPC alone.
PV is used as a surrogate outcome measure of physical thromboprophylaxis because higher PV was reported to indicate the efficacy of prophylaxis against thrombosis. 17,18 Such physical prophylaxis promotes blood flow and increases venous flow velocity and flow volume by mobilizing blood stored in the soleus vein and plantar venous plexus. Elevated venous flow velocity increases the shear stress on the endothelial cells lining the lumen, thus preventing VTE and DVT, and may also facilitate clearance of the valve sinuses. 19 The changes in sheer stress on the endothelial surface associated with IPC result in activation of the fibrinolytic system. 19 –21 The effects of AAE and IPC on PV in the lower limbs have been reported previously. 2,4 –6,22 The results of the present study indicated a 302% increase in PV associated with IPC compared with baseline obtained while sitting at rest. Murakami reported that use of a mobile IPC device (ActiveCare; Medical Compression Systems) resulted in a 42% increase in PV compared with resting in the supine position. 22 Thus, IPC showed a greater change in PV compared to resting in the sitting position than in the supine position. Stein also reported a greater change in PV associated with AAE from resting in the sitting position than in the supine position (500% and 118% increase, respectively). 23 Venous velocity differs depending on the measurement site. For example, PV during AAE in the femoral vein in the present study (47.1 cm/s) was higher than that in the popliteal vein in a previous study (28.4 cm/s). 24 This tendency corresponds to results from a report that concluded PV was higher in the femoral vein (44.8 cm/s) than in the popliteal vein (26.3 cm/s) during IPC to the thigh in the sitting position. 10
Observational data reported in 1989 suggested a symptomatic VTE event rate of ∼5% without prophylaxis after THA, TKA, and HFS. 25 More recently, the ACCP Guidelines estimated symptomatic DVT and PTE rates without prophylaxis in the immediate postoperative period for THA, TKA, and HFS as 2.8% and 1.5%, respectively. 1 In addition, the symptomatic DVT and PTE rates on prophylaxis with low-molecular-weight heparin were estimated as 1.25% and 0.55%, respectively. 1 These reductions in risk are due to improvements in medical practices, including early mobilization, more efficient and less traumatic surgical procedures, widespread use of IPC devices, better pain management allowing early mobilization, use of regional anesthesia, and frequent use of pharmacological prophylaxis. 26 –28 Nevertheless, the decrease in ambulatory ability after THA occasionally results in DVT, even after the period of bed rest in patients who have received pharmacological thromboprophylaxis, with symptomatic DVT incidence rates up to 90 days after THA and TKA of 0.7% and 0.9%, respectively. 1,15,29 –31 The shuffling gait of patients after major orthopedic surgery results in reduced calf muscle pump activity. We speculated that the reduction of ambulation ability causes insufficiency of the venous return mechanisms on ambulation, which consists of the calf muscle pump with calf muscle contraction and plantar venous plexus pump associated with weight-bearing activity. Of the two pumps, the calf pump is activated by the AAE. As a possible solution to assist the insufficient calf pump due to a decline in ambulation ability, we previously reported significant efficacy of applying resistance on venous velocity during AAE. 8 The results of the present study suggest that IPC during ambulation may represent another means of reducing the risk of DVT in walking patients with reduced ambulation ability, as IPC showed additive efficacy to the increased PV by activation of the calf muscle pump activity with AAE.
There have been no previous studies regarding whether IPC applied together with ambulation or AAE has a greater effect on PV compared to either AAE or IPC alone. The results of the present study indicated the additive efficacy of IPC and AAE on PV, which reached 76.2 cm/s in the sitting position and was increased to 48.1 cm/s with the application of IPC. Therefore, AAE with IPC, which increased PV to a greater extent than IPC alone, might be beneficial for preventing thrombosis. We recommend that patients perform AAE even with the use of a
Effective physical thromboprophylaxis requires a high degree of compliance, as the recommendation of the ACCP Guidelines (9th edition) is for IPC for more than 18 h/day. 1 High compliance was reported for the mobile IPC device not only in orthopedic inpatients (22.1 h/day) but also in outpatients, including walking patients (19.7 h/day) at high risk of DVT as well as no bleeding and non-inferior thromboprophylaxis effect compared to anticoagulants. 13 The present results support the usefulness of mobile IPC as reported in previous studies for walking patients at high risk of VTE.
This study had several limitations. First, AAE was substituted for ambulation, and blood velocity was measured in AAE with IPC. Thus, we cannot conclude that IPC would be efficacious on PV during ambulation, based on the observation that AAE with IPC further increased PV. In addition, the upright posture reduces venous return and thus central blood volume by pooling of blood in the lower parts of the body. 32 As stasis of blood flow is one of the three main factors associated with the formation of blood clots, the changes in blood flow produced by physical prophylaxis should also be measured during walking as well as in a standing posture. Second, blood velocity was measured only in the femoral vein, whereas DVT occurs at higher rates in the crural veins, such as the soleal vein and peroneal vein, than the femoral vein. 33 Some crural thrombi extend to the proximal veins as free-floating thrombi that can lead to fatal pulmonary thromboembolism. 34 Therefore, changes in blood flow produced by physical prophylaxis should also be measured in the crural veins. In addition, to prevent measurement selection bias, we could not obtain the opportunity to record additional duplex measurements of the flow. Third, the subjects were all healthy young adults, whereas patients undergoing operations with increased VTE risk, such as artificial joint replacement or hip fracture surgery, are usually older. 35 –38 Muscle has quantitative and qualitative differences between young adults and elderly people, and women have a higher risk of osteoporosis, which contributes to fracture risk. 39,40 Moreover, venous velocity during AAE with IPC postoperatively may not show changes corresponding to the results of the present study because patients show reduced venous flow following THA. 41 Therefore, the use of mobile IPC devices as physical prophylaxis for DVT can be recommended in ambulatory patients in hospitals after orthopedic surgery. Further studies are required to determine the differences between normal and decreased ambulation ability, efficacy, and adherence in a clinical context, and to follow-up the incidence rate of DVT.
Conclusion
IPC was associated with significantly higher PV during AAE occurring once every minute, than observed with either AAE or IPC alone, and simultaneous AAE with IPC could be useful for the prevention of DVT.
Footnotes
Author contributions
KS was the lead investigator of this study, conducted data collection, analyzed and interpreted data, and wrote the first draft of the manuscript. NT conceptualized this study, assisted in data interpretation, acquired funding and corresponding author. All authors debated the study design and protocol, conducted critical review and revision, approved the final version of the manuscript, and have accountability for all aspects of the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Japan Society for the Promotion of Science (JSPS; KAKENHI Grant No. JP17K10940).
