Abstract
Purpose:
To evaluate the integrity of lateral soft tissue in varus osteoarthritis knee by comparing the mechanical axis under varus stress during navigation-assisted total knee arthroplasty before and after compensating for a bone defect with the implant.
Methods:
Sixty-six knees that underwent total knee arthroplasty were investigated. The mechanical axis of the operated knee was evaluated under manual varus stress immediately after knee exposure and after navigation-assisted implantation. The correlation between each value of the mechanical axis and degree of preoperative varus deformity was compared by regression analysis.
Results:
The maximum mechanical axis under varus stress immediately after knee exposure increased in proportion to the degree of preoperative varus deformity. Moreover, the maximum mechanical axis under varus stress after implantation increased in proportion to the degree of preoperative varus deformity. Therefore, the severity of varus knee deformity leads to a progressive laxity of the lateral soft tissue. However, regression coefficients after implantation were much smaller than those measured immediately after knee exposure (0.99 vs 0.20). Based on the results of the regression formula, the postoperative laxity of the lateral soft tissue was negligible, provided that an appropriate thickness of the implant was compensated for the bone and cartilage defect in the medial compartment without changing the joint line.
Conclusion:
The severity of varus knee deformity leads to a progressive laxity of the lateral soft tissue. However, even if the degree of preoperative varus deformity is severe, most cases may not require additional procedures to address the residual lateral laxity.
Introduction
Soft tissue balancing is an important factor for achieving a good clinical outcome in total knee arthroplasty (TKA). Inadequate soft tissue balancing may lead to a number of complications that include instability, abnormal polyethylene wear, aseptic loosening, altered patellofemoral biomechanics, and pain. 1 –6 Patients undergoing TKA may therefore require some kind of mediolateral soft tissue balancing in order to provide a stable soft tissue envelope. 7,8 The development of lateral laxity is generally believed to be subsequent to medial tightness and correlates with the severity of the arthritic varus deformity. 9 –12 If the preoperative varus deformity is severe, the surgeon may have to consider the need for a semi-constrained implant or lateral ligament repair for postoperative lateral instability before surgery. However, little data exist as to the degree of laxity that depends on the severity of preoperative varus deformity. A reason for the scarcity of information may be a result of technical difficulties in obtaining an accurate evaluation for the laxity of lateral structures by a manual stress test due to bone defects and cartilage loss in the medial compartment. When evaluating the mechanical axis of the knee joint in a manual stress test, it is difficult to determine whether the axis is in varus under a manual stress test, owing to the laxity of the lateral ligament itself or a defect of bone and cartilage in the medial compartment. To compensate for medial bone and cartilage loss, Bellemans et al. 9 attempted to evaluate the coronal laxity of the lateral compartment under a manual stress test by filling in the medial compartment with an intercalary shim before bone cutting. However, even with the use of an intercalary shim, neutral coronal alignment could not be achieved in varus deformities exceeding 10°; thus, coronal laxity could not be evaluated in severe varus deformities.
With recent advances in computerized navigation software, the quantitative measurement for soft tissue balancing and intraoperative identification of the mechanical axis under stress are available in real time. 13 –16 Furthermore, navigation software allows for precise bone cutting and osteotomy with equal thickness to the implant and preservation of the physiological joint line. Because an appropriate thickness of the implant is compensated for the bone defect and cartilage loss in the medial compartment without changing the joint line by using this technology, it is possible to evaluate cases of varus deformity over 10° that Bellemans was unable to evaluate.
Establishing a correlation between the extent of preoperative varus deformity and the lateral laxity will aid in preoperative planning and soft tissue balancing in TKA procedures. We hypothesized that varus deformity of the knee leads to progressive stretching of the lateral structures. The purpose of this study was to examine this hypothesis by comparing the mechanical axis under varus stress immediately after knee exposure and after implantation during navigation-assisted TKA.
Materials and methods
This study adhered to the ethical guidelines of the 1975 Declaration of Helsinki. The study protocol was approved by our institutional review board, and all patients provided written informed consent.
Study design and patients
A total of 123 consecutive knees (105 patients) undergoing navigation-assisted TKA with osteoarthritis classified as Kellgren-Lawrence (KL) grade 3 or higher at our facility from May 2015 to May 2017 were included in this study. Patients with osteonecrosis, rheumatoid arthritis, valgus knee, posterior cruciate ligament (PCL) resection, cases in which the thickness of the tibial implant and insert differed from the thickness of the osteotomy, and cases exhibiting a postoperative hip-knee-ankle (HKA) angle of more than 3° varus and valgus recorded on the navigation screen in neutral position were excluded from this study. Based on this exclusion criteria, the following 57 knees were excluded: rheumatoid arthritis, 2; valgus knee, 5; PCL resection, 10; insert thickness, 37; postoperative HKA angle of more than 3° varus and valgus, 3. Because the absence of PCL affects the stability of coronal alignment, 17 10 cases were excluded. In the 37 cases excluded for insert thickness, the joint line was considered to have changed because a thicker insert than the osteotomy was selected. Three cases with postoperative HKA angle of more than 3° varus and valgus were excluded because the knees were not corrected to neutral alignment. The 66 remaining knees (59 patients) were enrolled in this study.
Surgical technique
Patients were under the care of three experienced surgeons who routinely used computer navigation in their standard TKA practice. All implants used in this study were cruciate-retaining Triathlon TKA (Stryker Orthopedics, Mahwah, NJ, USA). The Stryker imageless navigation system (Precision version 4.0; Stryker Orthopedics, Mahwah, NJ, USA), which does not require intraoperative fluoroscopy or preoperative CT images, was used for computer-assisted implantation. The system uses kinematic acquisition of the hip center and anatomical acquisition of the knee and ankle centers.
After knee exposure with the para-patellar approach, active patient trackers were fixed onto the femur and tibia with two bi-cortical anchoring pins, respectively. Care was taken so as not to interfere with knee motion; thus, pins were placed away from the surgical wound. After the registration of anatomical landmarks using standard navigation workflows, an anterior cruciate ligament resection was performed as a preliminary minimal medial release, whereby a periosteal sleeve was elevated approximately 5 mm distal to the joint line that included a part of the deep medial collateral ligament (MCL) fibers from the medial aspect of the tibia. Next, a complete removal of peripheral osteophytes from the medial and posteromedial aspects of the proximal tibia and distal medial femoral condyle was performed.
The modified gap technique was applied to all TKAs. First, a distal femoral cut was made perpendicular to the mechanical axis from femoral head to the center of knee joint with an extramedullary cutting block positioned under the guidance of navigation. In addition, a proximal tibial cut was also made perpendicular to the mechanical axis with an extramedullary cutting block. The amount of bone cutting at the lateral femoral and tibial condyle were the same thickness as the implant. Once the extension gap was created, it was measured with a seesaw-tensioner which is similar to a lamina spreader with a torque meter, tensor, and sliding rule. Thirty pounds of torque was applied to the tensioner. Then, the flexion gap was measured at 90° flexion. The posterior cut of the femur was determined according to the value of tensioner. Implantations were fixed with cement. The implantation gap was measured with a force of 30 pounds using a tensioner, and the thickness of the insert was defined as same as the value of the medial gap in the extended position. All patients received patellar resurfacing.
Evaluation criteria
Preoperative variables including age, sex, affected side, HKA angle, KL classification, maximum extension and maximum flexion were recorded.
The femoro-tibial mechanical angle was measured immediately after surgical knee exposure in neutral position without applied stress, and loading in the navigation screen was investigated and defined as “the degree of preoperative varus deformity.” This value did not account for changes from the neutral position but merely presented the value of the femoro-tibial mechanical angle showed in the navigation screen. To evaluate the laxity of lateral structures, the femoro-tibial mechanical angle in maximum manual varus stress was recorded on the computer navigation at two points immediately after knee exposure and after implantation. The femoro-tibial mechanical angle in maximum manual varus stress immediately after knee exposure was defined as “the degree varus immediately after knee exposure.” Furthermore, the femoro-tibial mechanical angle in maximum manual stress after implantation was defined as “the degree varus after implantation.” These measurements were performed after partial closure of the arthrotomy and were rounded off to the nearest 0.5°. In knees with a flexion contracture, the measurements were performed at maximum extension. All measurements were conducted by three surgeons with more than 15, 20 and 25 years of experience in TKA, respectively. The correlation between the degree of preoperative varus deformity and the degree varus immediately after knee exposure was investigated. In addition, the correlation between the degree of preoperative varus deformity and the degree varus after implantation was investigated.
Interobserver agreement in the measurement of the mechanical angle in maximum manual valgus stress was assessed by having multiple examiners evaluate 25 knees selected at random in this study using intra-class correlation coefficients (ICC).
Statistical analysis
The regression analysis was used to investigate the correlation between the degree of preoperative varus deformity and mechanical angle in maximum manual varus stress at two points. Gradient a1 and correlation coefficient r were calculated by performing a single linear regression analysis with the x-axis defined as the degree of the preoperative varus deformity and y-axis defined as the mechanical angle in maximum manual varus stress at two points.
The level of statistical significance was set at p = 0.05, and all calculations were performed using SPSS version 12 (SPSS Inc, Chicago, IL, USA).
Results
The patient characteristics of 66 knees (59 patients) are shown in Table 1. The mean age was 76.1 (63–96) years, comprising of 44 females and 15 males. The mean KL classification was 3.8, and the mean HKA angle was 12.4 ± 4.8°. The mean flexion contracture was 6.0 ± 8.9°. The mean baseline degree of preoperative varus deformity was 10.5 ± 3.8° varus (4–20.5° varus) at extension.
Patient characteristics.

Linear regression analysis between the degree of preoperative varus deformity and the mechanical angle in maximum manual varus stress recorded on the navigation screen are shown in Figure 1. The degree of varus immediately after knee exposure increased in proportion to the degree of preoperative varus deformity: y = 0.99x+3.25, R2 = 0.87, p < 0.001 (y: degree varus, immediately after knee exposure; x: degree of preoperative varus deformity). The degree of varus after implantation also increased in proportion to the degree of preoperative varus deformity: y = 0.20x+0.51, R2 = 0.17, p = 0.001 (y: degree varus, after implantation; x: degree of preoperative varus deformity). However, regression coefficients after implantation were much smaller than those measured immediately after knee exposure (0.99 vs 0.20).
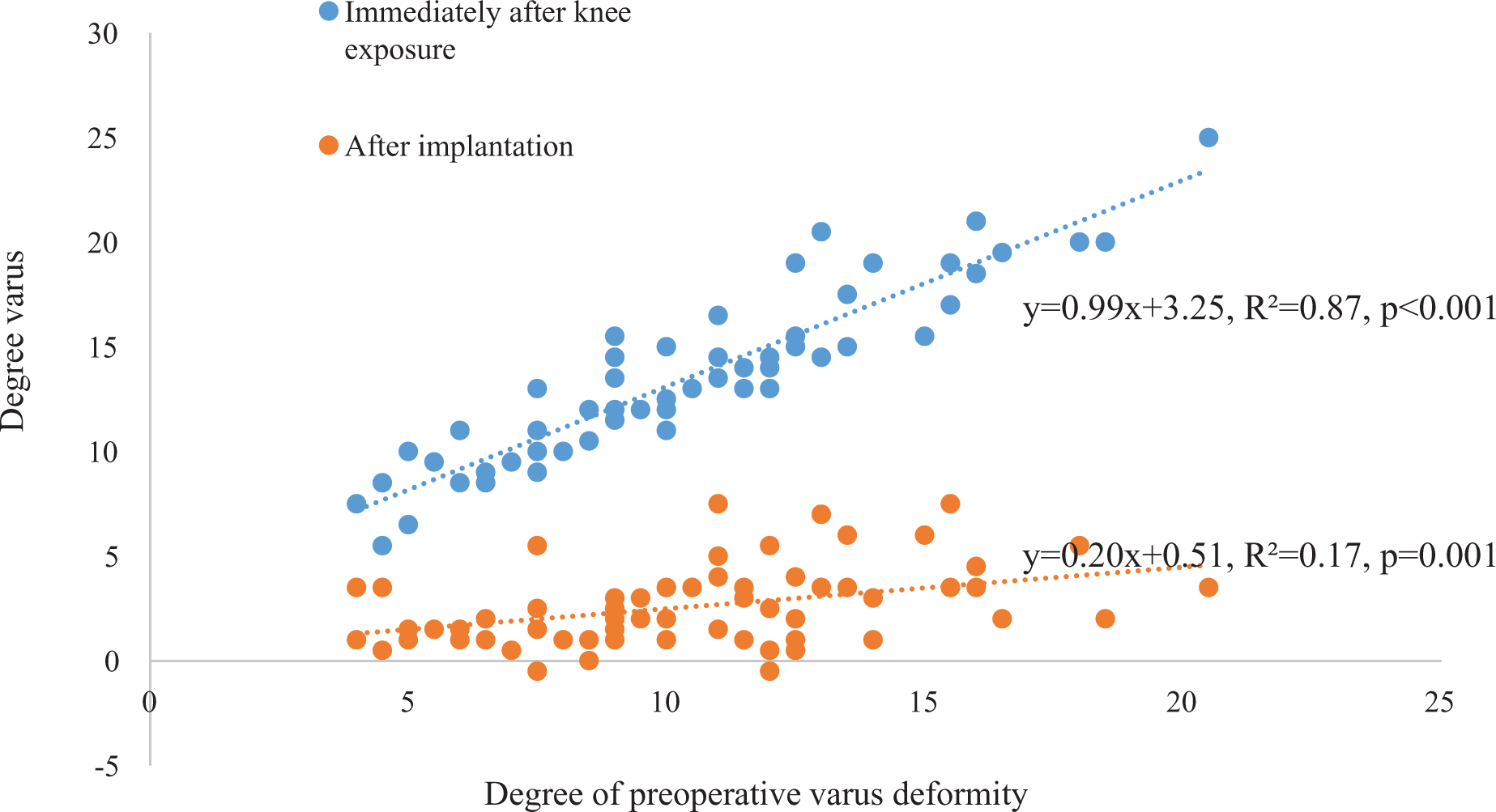
Linear regression analysis between the degree of preoperative varus deformity and mechanical angle in maximum manual varus stress in the navigation screen.
ICC of mechanical angle in maximum manual valgus stress based on the data of 25 knees in this study was 0.88.
Discussion
In clinical practice, most surgeons believe that severe varus deformity of the osteoarthritis knee would lead to progressive laxity of the lateral soft tissue in addition to tightness of the medial soft tissue envelope. To achieve mediolateral soft tissue balancing in the TKA procedure, it is important for knee surgeons to understand the degree of lateral soft tissue laxity according to the degree of varus deformity; therefore, this study may help surgeons recognize the influence of lateral factors on coronal laxity.
The main points of our study are summarized in Figure 1. The line of the degree varus immediately after knee exposure indicates the degree of lateral laxity as well as the degree of varus deformity. In contrast, the line of the degree varus after implantation indicates only the degree of lateral laxity, since the postoperative varus deformity is corrected by implantation. The maximum mechanical axis under varus stress after implantation increased in proportion to the degree of preoperative varus deformity. This means that the progression of varus deformity of the knee would lead to the progressive laxity of the lateral soft tissue. However, in the linear regression analysis, the regression coefficients measured after implantation were much smaller than those measured immediately after knee exposure. The reason for this difference is assumed to be due to the preoperative bone defect and cartilage loss in the medial compartment. Because of the bone defect and cartilage loss in the medial compartment, there was a strong tendency for varus angulation on manual stress tests immediately after knee exposure as the preoperative varus deformity progressed. Once an implantation with appropriate thickness is achieved without changing the joint line at the bone defect and cartilage loss in the medial compartment, that tendency showed a dramatic decrease. The maximum mechanical axis under varus stress after implantation can be estimated from the degree of preoperative varus deformity by using the regression formula between both criteria. If the degree of preoperative varus deformity is 20°, the maximum mechanical axis under varus stress after implantation is estimated to be approximately 4.5°; accordingly, we expect a reduction in varus deformity of roughly the same value. Based upon available data in the literature, 2–4° of mechanical axis deviation under varus stress after implantation were considered normal. 18 –23 Murer et al. 24 reported that 6.1° was the cut-off value which indicate the need of revision due to instability by stress radiographs. Thus, even if the degree of preoperative varus deformity is close to 20°, the laxity of the lateral soft tissue was negligible after implantation. To obtain good soft tissue balancing even within a 20-degree varus deformity in TKA procedures, most cases may not require special attention to the lateral laxity.
Studies on the relationship between lateral laxity and the progression of varus osteoarthritis are scarce in the literature and their results are controversial. Okamoto et al. 11 compared the intraoperative joint gap, amount of resected bone, and intraoperative soft tissue laxity on the lateral side according to the severity of preoperative varus deformity. The lateral soft tissue gap was larger in the severe varus group compared to the mid and moderate varus group. Ishii et al. 25 evaluated preoperative coronal laxity by using radiographs with applying a force of 150 N. The distribution of lateral laxity in knees with advanced osteoarthritis were wider than that of controls. However, lateral laxity indicated that 90% of patients were within a clinically “balanced” range. Conversely, Creaby et al. 26 investigated differences in passive frontal plane laxity of asymptomatic control knees to those with mild, moderate, and severe medial femoro-tibial osteoarthritis by using a dynamometer. There was no difference in varus angular laxity. Ishii et al. 25 stated that lateral laxity did not change dramatically with the progress of osteoarthritis. However, a limitation of these three studies is that their quantitative results are affected by the medial compartment. Okamoto et al. 11 reported that a greater amount of medial soft tissue release was often performed when severe preoperative varus was observed, and the degree of medial release varied according to the case; therefore, the degree of medial release may have influenced the joint gap. Ishii et al. 25 and Creaby et al. 26 found that the osteochondral defect affected their results because the alignment was evaluated by stress imaging while the medial bone and cartilage defect still remained. Thus, an accurate evaluation of the lateral laxity in osteoarthritis knee was difficult due to medial bone defect and cartilage loss. To compensate for medial bone defects, Bellemans et al. 9 used an intercalary shim. A specialized intercalary shim with incremental thickness was placed into the medial femoro-tibial compartment until obtaining a neutral mechanical alignment on the navigation screen prior to bone cutting. However, the coronal alignment could not be corrected to neutral alignment despite the use of an intercalary shim in varus deformities over 10°, and coronal alignment with severe varus deformities could not be evaluated. On the other hand, in our study, the degree of medial release was the same in all cases and accurate implantation with the use of navigation assistance enabled the evaluation of varus deformities over 10° via a manual stress test after implantation, which preserved the joint line.
In the present study, the following three conditions were fulfilled in order to ensure an accurate assessment of the relationship between the lateral laxity and the degree of preoperative varus deformity. First, the thickness of the lateral femoral and tibial bone cutting needs to be equal to the implant thickness without changing the joint line. In this study, implantation was performed with appropriate thickness by achieving bone cuts of the same thickness using navigation assistance. Second, the thickness of the insert must be correct. Even if lateral laxity is initially observed, lateral laxity may disappear when a thick insert is selected. To address this issue, our study excluded cases in which a thicker insert was chosen over bone cutting. Third, postoperative neutral alignment needs to be obtained regardless of the degree of varus deformity. Lateral laxity cannot be accurately evaluated if the varus of the knee joint remains or the alignment is valgus after surgery. In this study, three cases (4.5%) which the degree of postoperative alignment was over 3° varus were excluded from this study.
The present study used the femoro-tibial mechanical angle in neutral position without applied stress and loading in place of preoperative HKA as an indicator of the degree of preoperative varus deformity. The HKA complex is in a loaded and stressed state. 27 Because of the weight load, varus deformations are likely to appear more severe than normal. For this reason, it could not be used as an indicator of the original degree of varus deformation.
There are some limitations in our study that must be considered. Firstly, our protocol relied on manual stress testing which was not quantitative. Contact between the femur and tibia may affect the results. Although all measurements were conducted by surgeons with extensive experience were obtained during the procedure with confirmation of ICC, the data values may vary. Secondly, a case of flexion contracture with an inability to achieve full extension was included in this study. A flexion contracture may affect the lateral instability. In addition, evaluations in full extension may differ from those in flexion with contracture. However, cases of knee osteoarthritis with severe varus deformity are associated with flexion contractures and could not be excluded from the study. Thirdly, measurements were made under anesthesia which may be overestimated due to the relaxation of the muscle. Although an evaluation of postoperative laxity should have been examined to assess the condition of the knee, a consistent evaluation was not obtainable due to the unavailability of the navigation system. Despite this limitation, we believe the results of this study are valid and useful for surgeons dealing with the varus osteoarthritis knee.
The severity of varus knee deformity leads to a progressive laxity of the lateral soft tissue. However, even if the degree of preoperative varus deformity is severe, most cases may not require additional procedures to address the residual lateral laxity.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
