Abstract
Purpose:
This study aimed to investigate how fibroblastic and chondrocytic properties of human meniscal fibrochondrocytes are affected in culture conditions according to the type of meniscal pathology and localization, and to provide basic information for tissue-engineering studies.
Methods:
Primary fibrochondrocyte cultures were prepared from meniscus samples of patients who had either traumatic tear or degeneration due to osteoarthritis. Cultures were compared in terms of mRNA expression levels of COL1A1, COL2A1, COMP1, HIF1A, HIF2A, and SOX9 and secreted total collagen and sulfated sGAG levels according to the type of meniscal pathology, anatomical localization, and the number of subcultures.
Results:
mRNA expression levels of COL1A1, COMP1, HIF1A, HIF2A, and SOX9 were found to be increased in subsequent subcultures in all specimens. COL1A1 mRNA expression levels of both lateral and medial menisci of patients with traumatic tear were significantly higher than in patients with degenerative pathology, indicating a more fibroblastic character. P1 subculture of lateral and P3 or further subculture of medial meniscus showed more fibroblastic characteristics in patients with degenerative pathology. Furthermore, in patients with degenerative pathology, the subcultures of the lateral meniscus (especially on the inner part) presented more chondrocytic characteristics than did those of medial meniscus.
Conclusions:
The mRNA expression levels of the cultures showed significant differences according to the anatomical localization and pathology of the meniscus, indicating distinct chondrocytic and fibroblastic features. This fundamental knowledge would help researchers to choose more efficient cell sources for cell-seeding of a meniscus scaffold, and to generate a construct resembling the original meniscus tissue.
Introduction
Meniscus injuries are a significant clinical problem among orthopaedic pathologies since they might lead to painful and restrictive consequences. The engineering of biologically-driven tissues is one of the most promising choices to repair the meniscus, restore meniscus function, and prevent the onset of osteoarthritis. 1 Autologous meniscus cells isolated from the surgical debris of patients with traumatic or degenerative meniscus pathology are seen as a potential cell source because they have a limited immune response and an appropriate phenotype. 2 –4 In animal models, some studies have shown that culturing autologous cells on a scaffold after meniscal biopsy might be a method for obtaining a tissue similar to the original meniscus. 5 However, due to the distinctive features of inner and outer meniscus zones, seeding one type of cell on a scaffold might not yield optimal results for obtaining a biologically driven tissue with characteristics and biomechanics similar to the original meniscus.
Fibrochondrocytes are the primary cells that maintain the structure of the meniscus, and they exhibit both fibroblastic and chondrocytic characteristics. Both extracellular matrix content and cell properties differ throughout the inner and outer meniscus zones. Fibroblast-like cells in the outer third of the meniscus produce mostly collagen type I, while chondrocyte-like cells located at the inner two-third of the meniscus produce mainly collagen type 2 and proteoglycan. 6 Vascular and neural elements are found in the outer region with more fibroblastic characteristics, conveying better healing capacity, while the middle and inner regions of the meniscus are avascular, aneural, and alymphatic, with reduced healing capacity that imbues the meniscus with greater viscoelasticity. 7 This study aimed to investigate how fibroblastic and chondrocytic properties of human meniscal fibrochondrocytes are affected in culture conditions according to the pathology (traumatic tear or degeneration) and anatomical region (lateral or medial) of the meniscus. The information obtained from this study can help us shed light on the lack of information about cell selection in meniscus tissue engineering with autologous cell-seeded meniscal scaffolds, which is important in terms of generating a construct resembling the meniscus both histologically and biomechanically.
Materials and methods
Meniscus samples were obtained from surgical debris of patients who underwent total knee arthroplasty due to Kellgren-Lawrence Grade III–IV bi- or tri-compartmental osteoarthritis and patients who underwent partial meniscectomy due to acute (<3 months) traumatic meniscal tear. All patients had weight-bearing knee anteroposterior and lateral radiographs. The patients with traumatic meniscus tear had magnetic resonance imaging of the knee. To rule out any possibility of a degenerative tear in the traumatic group, the patients who had pain started immediately after a recent particular knee injury with no preceding history of knee pain were diagnosed as acute traumatic meniscus tear. None of the patients with traumatic tear had an accompanying anterior cruciate tear. Patients who had a concomitant rheumatological disease or inflammatory arthritis, unicompartmental osteoarthritis, previous surgical history of the affected knee, or infectious disease at the time of surgery were excluded from the study. Informed consent was obtained from all patients. This study was approved by the local Ethics Committee (Istanbul University Clinical Research Ethics Committee, Approval number: 116871).
All meniscus samples obtained during surgery were noted as either medial or lateral in origin according to their anatomical locations. Patients were separated into two groups based on the meniscal pathology as traumatic or degenerative. All patient demographics are given in Table 1.
Demographics of patients.
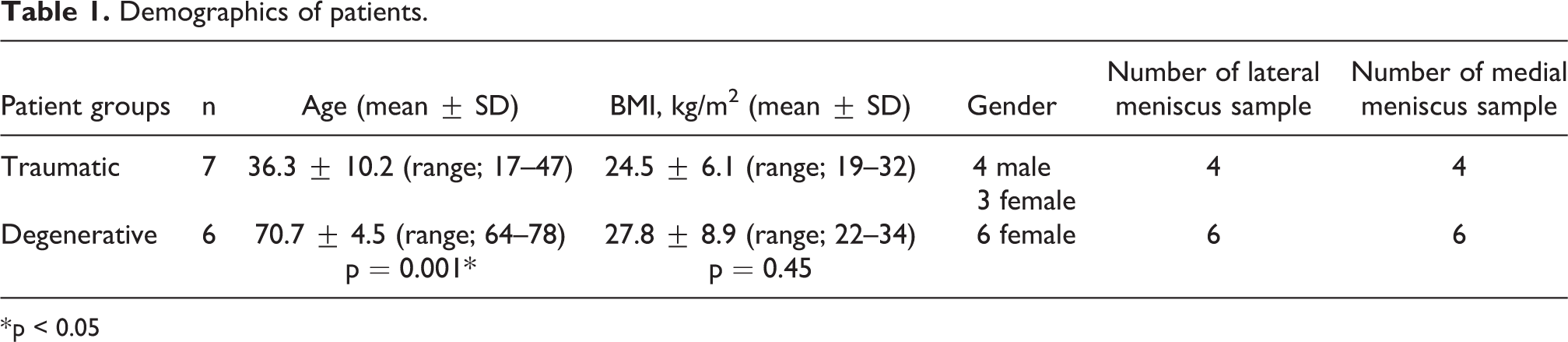
* p < 0.05
All meniscus samples were immediately transferred to the cell culture laboratory in ice-cold HBSS (Gibco, 14025050) containing 1.5% antibiotic-antimycotic (Gibco, 15240-062). The menisci of the patients who underwent total knee arthroplasty were cut into two pieces from the middle point of meniscal width as inner and outer portions. All meniscus samples obtained from a traumatic tear belonged to the inner portion of the meniscus. All meniscus samples were cut into small pieces approximately 1 mm3 in size using scalpels. Pieces were incubated at 37°C in a 5% CO2 humidified hood for 24 h in the culture medium, comprising DMEM(Gibco, 41965039), 10% fetal bovine serum(Gibco, 10270106), 25 μg/ml L-ascorbic acid (Sigma, A4544), 1% nonessential amino acids(Gibco, 11140050) and 1% PSF containing 0.2% collagenase II(Gibco, 17101015) for enzymatic digestion. The next day, cells were dissociated using cell strainers (Falcon, 352350). Culture medium containing collagenase II was replaced with fresh culture medium, and then cells were plated to flasks coated with 1:50 collagen I (Gibco, A1064401) at 25% confluence and labeled as subculture 0 (P0). The media was replenished every 3 days. When cells reached 85–90% confluence, the cells were detached from the flask with 0.05% trypsin-EDTA solution (Sigma, T4049) and subcultured at 25% confluence. The cells were subcultured three times, and each subculture was labeled as P1, P2, and P3, successively. The experimental design was shown in Figure 1.
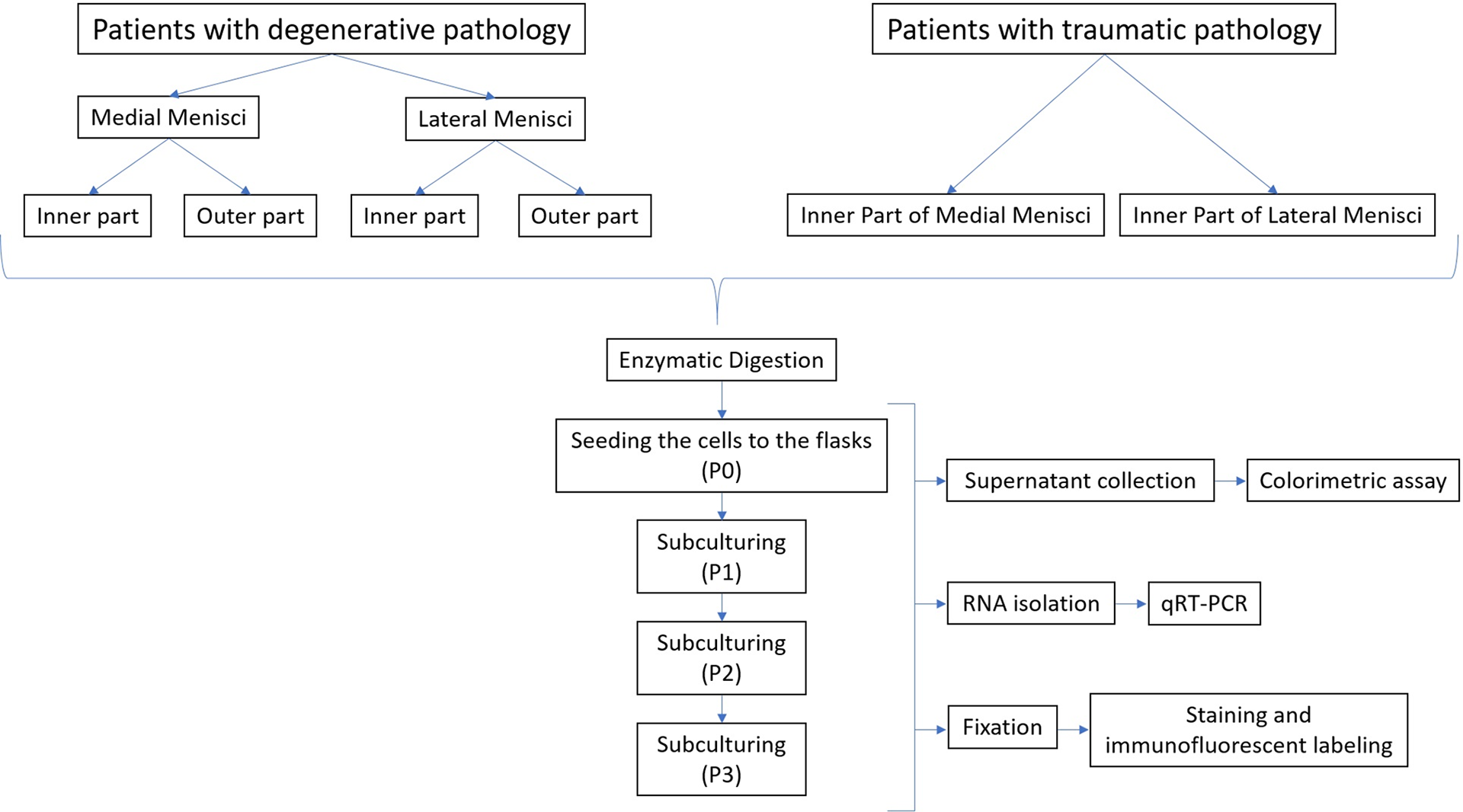
Experimental design. All experimental procedures were schematized in order.
RNA was isolated from cultured meniscal fibrochondrocytes using the PureLink RNA isolation kit according to the manufacturer’s protocol (Ambion, 12183018A). 15 ng of RNA from each sample was used for cDNA synthesis. cDNA synthesis was performed using the iScript cDNA synthesis kit according to the manufacturer’s protocol (BioRad, 1708891). mRNA expression levels of COL1A1 (UPL Probe No. 1, Roche, 04 684 974 001), COL2A1 (UPL Probe No. 9, Roche, 04 685 075 001), COMP1 (UPL Probe No. 38, Roche, 04 687 965 001), HIF1A (UPL Probe No. 66, Roche, c04 688 651 001), HIF2A (UPL Probe No. 4, Roche, 04 685 016 001), and SOX9 (UPL Probe No. 61, Roche, 04 688 597 001) were analyzed by qRT-PCR using Universal Probe Library (UPL) probes and the LightCycler 480 Probe Master Mix kit (Roche, 04707494001) with a LightCycler 480 machine (Roche Applied Biosystems). RPL13A (UPL Probe No. 28, Roche, 04 687 604 001), B2 M (UPL Probe No. 42, Roche, 04 688 015 001), and HPRT1 (UPL Probe No. 73, Roche, 04 688 961 001) were used as endogenous reference genes for normalization. qRT-PCR was performed in triplicate for each sample using following profile: denaturation at 95°C for 10 min, 45 cycles of amplification at 95°C for 10 sec, 60°C for 30 sec, 72°C for 1 sec, and cooling at 40°C for 10 sec. Reaction mixture including no cDNA used as negative control.
Culture supernatant collected during each subculturing was used for the measurement of collagen and sGAG levels. Collagen and sGAG levels were determined by colorimetric assay using the Sircol soluble collagen assay kit(Biocolor Life Science Assays, S1000) and Blyscan glycosaminoglycan assay kit(Biocolor Life Science Assays, B1000), respectively. Each sample was tested in duplicate.
For staining and immunofluorescence labelings, cells were fixed with 3.7% paraformaldehyde in phosphate buffer, pH 7.4. Cresyl violet staining was performed, and the cells were observed by light microscopy to identify cell morphology of both medial- and lateral-derived menisci in cell culture conditions. Double immunofluorescent labeling for the fibroblastic marker TE-7 and the chondrocytic marker SOX9 was used to determine cellular characteristics of fibrochondrocytes. Immunofluorescent labeling was done using TE-7 (CBL271, Millipore) primary antibody at a dilution of 1:100 and SOX9 (AB5535, Millipore) primary antibody at a dilution of 1:200 as previously described. 8 Negative controls for immunofluorescent stainings which omit primary antibodies and include only anti-rabbit seconder antibody tagged with Alexa Fluor 488 (A11034, ThermoFisher) and anti-mouse seconder antibody labeled with Alexa Fluor 568 (ab175703, Abcam) were also included (data was not shown). The Leica Application Suite Image Overlay Software (Leica Microsystems Ltd, Heerbrugg, GE) was used to obtain overlay images. Assessments were also repeated with the Leica TCS SPE confocal system.
Collagen and sGAG levels were calculated with the formula obtained from the graphic of distribution drawn using the collagen and sGAG levels in the standards and OD values. Ct values obtained from qRT-PCR were used in the formula *ΔCt = 2(Geometric mean of reference genes − Ct target gene) to determine relative expression levels of target genes, and Microsoft Office Excel was utilized for calculations and graphics as previously described. 9
Analysis of all comparisons was performed using three different methods: Between-subculture analysis was performed in both patient groups and all meniscus types. Between-meniscus type analysis was performed in both patient groups and all subcultures. Between-patient group analysis was performed in all meniscus types and all subcultures.
A correlation analysis was also performed. For both comparison and correlation analysis, raw data of each group were analyzed on GraphPad InStat DTCG 3.06 (GraphPad Software, Inc. San Diego USA). Depending on the normality of data, either a one-way ANOVA followed by Tukey-Kramer multiple comparisons test or Kruskal-Wallis followed by Dunn’s multiple comparisons test was performed to compare more than two groups. Unpaired t-test, unpaired t-test with Welch correction, or Mann-Whitney U test was performed to compare two groups. Depending on the normality of data, either Pearson’s or Spearman’s correlation was performed. P-values less than 0.05 were considered statistically significant. Data are presented as mean (SD). Power analysis was performed to determine the minimum sample size required at a significance level α = 0.05 and power = 0.8, based on data from previous studies. 10 –12 The analysis revealed that a minimum sample size between 4 and 7 samples was required (allocation ratio 1:1).
Results
There was no statistically significant difference in either collagen or sGAG levels between lateral and medial menisci in any patient groups or any subcultures (Supplementary figure 1). COL1A1 mRNA expression levels were used to define fibroblastic characteristics, and COL2A1, COMP1, HIF1A, HIF2A, and SOX9 levels were used to define chondrocytic characteristics. COL2A1 mRNA expression was not included in the statistical analysis given the low levels of expression (cycle threshold was higher than 35) of subcultures. mRNA expression levels of COL1A1, COMP1, HIF1A, HIF2A, and SOX9 were found to be increased in subsequent subcultures compared to the levels at P0 in each meniscus type (inner and outer parts of both lateral and medial meniscus) in both patient groups (traumatic and degenerative) (Figure 2, Supplementary Table 1). The gene expressions differed between medial & lateral menisci and between patient groups (Figure 3, 4) (Supplementary Table 2, 3). It was found that COL1A1 mRNA expression levels of both lateral and medial menisci of patients with traumatic tear were significantly higher than in patients with degenerative pathology (Figure 4, Supplementary Table 2). The inner part of lateral menisci from patients with degenerative pathology showed higher COMP1 expression levels at P2, higher HIF2A expression at P3, and higher SOX9 expression at P0 compared to medial (Figure 3, Supplementary Table 2). P1 subculture of lateral and P3 or further subculture of medial meniscus with degenerative pathology showed increased COL1A1 levels (Supplementary Table 3). The subcultures of lateral meniscus with degenerative pathology presented more increased COMP1 and HIF1A expression levels (Supplementary Table 3).

Comparisons of subcultures in lateral and medial menisci of both patient groups. *p < 0.05, **p < 0.01, ***p < 0.001

Comparisons between lateral and medial menisci in subcultures of both patient groups. *p < 0.05, **p < 0.01, ***p < 0.001

Comparisons between patient groups (traumatic vs degenerative pathology) in subcultures of inner parts of both lateral and medial menisci. *p < 0.05, **p < 0.01, ***p < 0.001
A correlation analysis was also performed for collagen & sGAG levels, collagen & COL1A mRNA expression levels, and sGAG & COMP1 mRNA expression levels in all subcultures of each patient group. A positive correlation between collagen and sGAG levels was found when inner and outer parts of lateral menisci of patients with degenerative pathology were analyzed together at P2 (n = 6; r = 0.9094, r2 = 0.8270; 95% CI: 0.002099 to 0.009398, p < 0.05). When inner and outer parts of lateral menisci of patients with degenerative meniscus were analyzed together, a positive correlation between collagen level and COL1A1 mRNA level was found only when data from all subcultures pooled (n = 15; r = 0.6871, r2 = 0.4721; 95% CI: 0.01340 to 0.05971, p < 0.01). A positive correlation between sGAG level and COMP1 mRNA level was found when inner and outer parts of lateral menisci of patients with degenerative meniscus were analyzed together only when data from all subcultures pooled (n = 15; r = 0.6618, r2 = 0.4380; 95% CI: 0.1459 to 0.7619, p < 0.01).
Immunofluorescent labeling revealed that both fibroblastic and chondrocytic characteristics were present in all cells regardless of their morphologies in all subcultures of both menisci of both patient groups (Figure 5, 6).

Morphology and cell characteristics of meniscal cells in cell culture conditions. Morphologically fibroblast-like (short-arrow) and chondrocyte-like (long-arrow) cells were observed with cresyl-violet staining (20X). (Asterix: dedifferentiated chondrocyte-like cell) Immunofluorescent labelings revealed that both characteristics were present in all cells regardless their morphology (63X, confocal microscopy images).
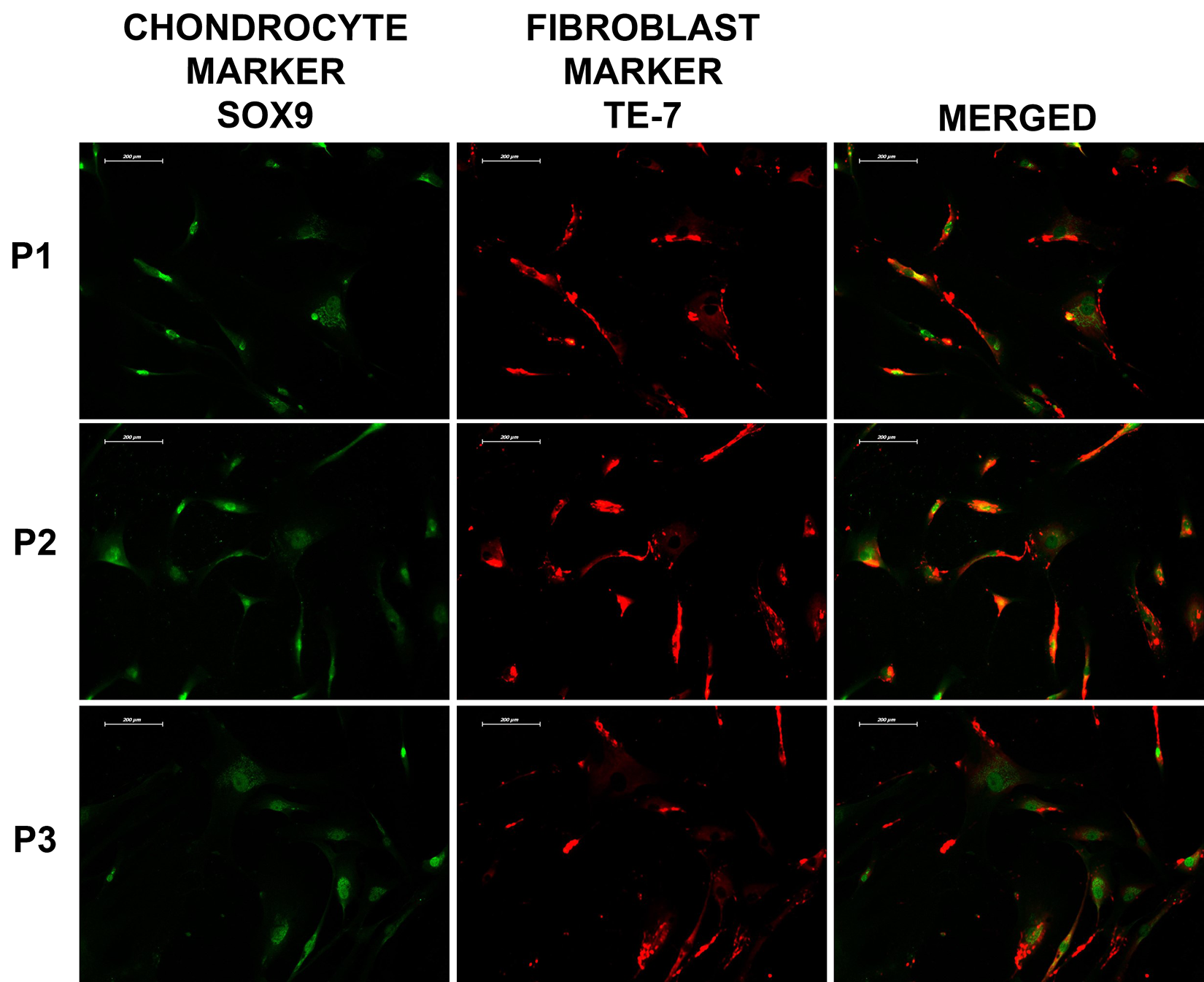
Immunofluorescent labelings of fibroblastic and chondrocytic markers. Both fibroblastic and chondrocytic characteristics carrying cells were observed in all subcultures of both menisci of both patient groups, 20X.
Discussion
The total collagen or sGAG levels did not differ significantly in any of the comparisons but mRNA expressions showed significant changes according to the number of subcultures, the type of meniscal pathology, and the anatomical localization of the menisci. The culture of menisci with traumatic tear showed more fibroblastic character compared to the menisci with degeneration due to osteoarthritis. The lateral menisci of the patients with degenerative pathology presented more chondrocytic pattern compared to the medial menisci. These results can guide us to decide which subculture and what type of meniscus can help us seeding a scaffold optimally to obtain a biologically driven tissue with characteristics and biomechanics similar to the original meniscus. Our proposal for histologically-favorable cell-seeding of a meniscus scaffold is summarized in Figure 7.

The summary of the best options for cell-seeding of a meniscal scaffold.
There was no significant difference in total collagen or sGAG levels in any of the comparisons. This result may point out that total collagen levels (including type I–V), or sGAG, might not be a discriminating factor for the choice of subculture number in tissue engineering. However, this situation may differ with regard to specific collagen types like collagen type I and II. No significant difference in total collagen or sGAG levels can also be a result of the low number of samples.
The underlying pathology can affect the characteristics of a meniscus. Osteoarthritis is a pathology affecting the whole joint through the changes in joint biology and the release of degradative metalloproteinases, cytokines, chemokines, etc. Therefore, meniscal pathology occurs as part of the global response of the joint in both the osteoarthritis-affected and unaffected joint compartments with similar histopathologic changes between joints with and without significant cartilage erosion. 13,14 Several studies strived to define the characteristics of the meniscus excised from osteoarthritic knee or meniscal tear to provide useful information for cell-based strategies since surgical debris is a valuable source for such regenerative plan. 14 –17 Kodama et al. reported that the menisci degenerated due to osteoarthritis showed more chondrogenic potential. 16 Brophy et al. compared the gene expressions of traumatic and degenerative tears and found that degenerative tears have higher COL1A1 expression, but they did not analyze any subcultures of the meniscal tissue. 18 In our study, when patient groups were compared for COL1A1 mRNA expression levels from both lateral and medial menisci within the same subculture numbers, it was found that expression levels in patients with traumatic tear were significantly higher than in patients with degenerative pathology. This situation might be interpreted as cells from patients with traumatic tear increasing their COL1A1 levels during subculturing (P3 or later), thus maintaining their fibroblastic characteristics at an increasing rate, which is more suitable for cell-seeding to the outer portion of a meniscus scaffold. In patients with degenerative pathology, the same situation seems to require earlier subcultures (such as P1) for lateral meniscus or further subcultures such as P3 for medial meniscus.
Another variable that might affect the quality of culture is the anatomical site of the meniscus from which the sample originates. Animal studies show that lateral and medial menisci cells might behave differently from each other in the culture microenvironment. 19,20 Fuller et al. showed that the characteristics and pathological response of medial versus lateral and inner compared with outer zones differs. 19 Kremer et al. showed that lateral menisci of horses have more chondrocytic features with increased rates of collagen II and sGAG. 20 In our study, considering higher COMP1 expression levels at P2, higher HIF2A expression at P3, and higher SOX9 expression at P0 in the inner part of lateral menisci from patients with degenerative pathology showed increased chondrocytic characteristics compared to medial, indicating a more chondrocytic phenotype of the lateral meniscus, which is also consistent with the results of the study by Kremer et al. 20 This aspect might make the inner part of lateral menisci a better choice for cell seeding to the inner portion of a meniscus scaffold rather than medial menisci. Increased load on lateral meniscus compared to medial might lead to degeneration over the years, leading to an increase in chondrocytic character and potentially explaining our results. In patients with a traumatic tear, there was no distinctive difference between lateral and medial meniscus regarding the expression of mRNAs. Furumatsu et al. also showed that inner meniscus cells carry a more chondrogenic phenotype compared to outer meniscus cells. 21 Our results also showed a more chondrogenic phenotype in cells obtained from the inner portion of the menisci compared to the outer portions in patients with degenerative pathology, indicating a better choice for cell seeding of the inner part of a scaffold.
When results for the correlation analysis between COL1A1 and total collagen levels were considered, we solely found a positive correlation between the total collagen level of lateral menisci of patients with degenerative pathology and COL1A1 mRNA only when data from all subcultures pooled. An explanation for this finding might be that COL1A1 encodes collagen type I but that the measured collagen level includes types I to V and the levels of different types of collagen other than type I might be increasing or decreasing.
Genes such as COL1A1, COL2A1, aggrecan (ACAN) and SOX9 are commonly used to determine the phenotype of meniscal cells however, it was recently reported that COL2A1 and ACAN may not be suitable markers for meniscal cells since they have significantly low expression levels in the meniscus. 22,23 Some studies have found a dramatic downregulation of COL2A1 mRNA, 24,25 while some was unable to detect the expression in cell culture, 26 including ours. It is important to note that some conditions such as injury, degeneration, cell culture environment and extracellular matrix component that is used during culturing may change gene expression profile. Besides, COL2A1 expression might depent on some other factors including SOX5, SOX6, interleukin 1β (IL-1β), interferon γ (IFN-γ) other than SOX9, a suggested transcription factor of COL2A1. 27,28
Most studies define fibroblastic or chondroblastic characteristics by morphological assessment of the cells in culture. 15,29 On the other hand, our study and a limited number of studies evaluated gene expression and extracellular matrix component profiles in addition to cell morphology. 17 Our study took assessment one step further by evaluating markers for fibroblastic or chondrocytic characteristics, the end products of gene expression, using immunofluorescent labeling. Most studies involving animal menisci report that fibrochondrocytes have altered gene expression profiles, decreased chondrocytic characteristics, and exhibit increased fibroblastic features in culture conditions. 30,31 In the present study, all morphologically defined fibroblast-like cells, chondrocyte-like cells, and dedifferentiated chondrocyte-like cells were shown to express both SOX9, a chondrocytic character marker, and TE-7, a fibroblastic character marker, by IF. Given these findings, at least for the meniscal cell cultures, it might not be useful to evaluate cells by their morphology to define their true character. Tissue engineering studies for menisci should consider that meniscal cell cultures generally maintain stable chondrocytic characteristics defined by COMP1, HIF1A, HIF2A, SOX9, and sGAG levels in P0-P3 subcultures, regardless of the type of meniscal pathology.
Based on COL1A1 and collagen levels, subcultures at P3 or later might be preferable for tissue engineering in patients with a traumatic tear, regardless of the anatomical region of the meniscus. In patients with degenerative pathology, the choice of subcultures for tissue engineering depends on the anatomical origin of the meniscus. The P1 subculture of both inner and outer part of lateral and P3 or later subculture of both inner and outer part of medial meniscus might be preferable for the outer portion of a scaffold due to the subcultures having more fibroblastic characteristics. Again, in patients with degenerative pathology, subcultures of inner part of lateral meniscus, rather than medial, might be a better choice for cell seeding to the inner portion of a scaffold due to enhanced chondrocytic characteristics.
This study has several limitations. Firstly, the patient groups were relatively small and studies with higher patient numbers can provide more knowledge for the field. However, most of the available studies analyzing human menisci or cartilage in cell culture also included a similar or lower number of patients due to the difficulty in obtaining and analysis of surgical debris. 2,15,21,29,32 Besides, the patients with degenerative pathology had a higher mean age, therefore some of the biological changes could have developed due to the senescence of the meniscal cells. 33 The average body mass index of the patients with degenerative pathology was higher which might affect the loads on the menisci thus changing the microenvironment, however, the difference was not statistically significant. Also, there was no comparison to normal menisci, since our main aim was to analyze the potential role of meniscal surgical debris in cell-based strategies. Besides, it would be ethically inappropriate to obtain healthy meniscal tissue from a healthy individual. To our knowledge, there are no studies in the literature comparing human meniscus cultures based on the type of meniscal pathology (traumatic tear versus degeneration due to osteoarthritis) and side of the anatomic origin of the meniscus to question their potential for seeding inner and outer portions of a cell scaffold.
Conclusions
Our results indicated that all cell types in all cultures presented both chondrocyte and fibroblast characteristic markers with immunofluorescent labeling, regardless of their morphology. However, the mRNA expression levels of the cultures showed significant differences according to the localization and pathology of the meniscus. Following mRNA expression levels of chondrocytic/fibroblastic markers would be a better choice for defining the characteristics of the cells. This fundamental knowledge would enable researchers to choose more efficient cell sources and the subculture numbers for optimal cell-seeding of a meniscus scaffold.
Supplemental material
Supplemental Material, sj-doc-1-osj-10.1177_23094990211000168 - Gene expression profiling of primary fibrochondrocyte cultures in traumatic and degenerative meniscus lesions
Supplemental Material, sj-doc-1-osj-10.1177_23094990211000168 for Gene expression profiling of primary fibrochondrocyte cultures in traumatic and degenerative meniscus lesions by Nuri Aydın, Bedri Karaismailoğlu, Merve Alaylıoğlu, Duygu Gezen-Ak, Büşra Şengül, Esin Candaş, Selma Yılmazer and Erdinç Dursun in Journal of Orthopaedic Surgery
Supplemental material
Supplemental Material, sj-doc-2-osj-10.1177_23094990211000168 - Gene expression profiling of primary fibrochondrocyte cultures in traumatic and degenerative meniscus lesions
Supplemental Material, sj-doc-2-osj-10.1177_23094990211000168 for Gene expression profiling of primary fibrochondrocyte cultures in traumatic and degenerative meniscus lesions by Nuri Aydın, Bedri Karaismailoğlu, Merve Alaylıoğlu, Duygu Gezen-Ak, Büşra Şengül, Esin Candaş, Selma Yılmazer and Erdinç Dursun in Journal of Orthopaedic Surgery
Supplemental material
Supplemental Material, sj-doc-3-osj-10.1177_23094990211000168 - Gene expression profiling of primary fibrochondrocyte cultures in traumatic and degenerative meniscus lesions
Supplemental Material, sj-doc-3-osj-10.1177_23094990211000168 for Gene expression profiling of primary fibrochondrocyte cultures in traumatic and degenerative meniscus lesions by Nuri Aydın, Bedri Karaismailoğlu, Merve Alaylıoğlu, Duygu Gezen-Ak, Büşra Şengül, Esin Candaş, Selma Yılmazer and Erdinç Dursun in Journal of Orthopaedic Surgery
Supplemental material
Supplemental Material, sj-tif-1-osj-10.1177_23094990211000168 - Gene expression profiling of primary fibrochondrocyte cultures in traumatic and degenerative meniscus lesions
Supplemental Material, sj-tif-1-osj-10.1177_23094990211000168 for Gene expression profiling of primary fibrochondrocyte cultures in traumatic and degenerative meniscus lesions by Nuri Aydın, Bedri Karaismailoğlu, Merve Alaylıoğlu, Duygu Gezen-Ak, Büşra Şengül, Esin Candaş, Selma Yılmazer and Erdinç Dursun in Journal of Orthopaedic Surgery
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received financial support for the research, authorship, and/or publication of this article: This study was funded by Scientific Research Projects Coordination Unit of Istanbul University-Cerrahpasa (Project No: 45372, 47186, 27781).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
