Abstract
Objective:
Despite the wide use of tranexamic acid (TXA) in the perioperative period of total hip arthroplasty (THA), whether the hemostatic state changes after the application of intravenous (IV)-TXA are still unknown. The aim of this study was to investigate whether IV administration of TXA changes the blood coagulation following primary THA via thrombelastography (TEG) analysis and conventional laboratory tests.
Methods:
A total of 174 patients who underwent primary THA from September 2016 to July 2018 were selected. They were randomly divided into two groups, 86 patients with IV administration of 15 mg/kg TXA and 88 controls without TXA usage. Demographic data, TEG paremeters,
Results:
There were no differences with regard to TEG or conventional laboratory tests between the two groups (p > 0.05). The total blood loss and drain blood loss in the TXA group were significantly lower than those in the control group (p < 0.05). The transfusion rates and the volume of blood transfusion of the control group were higher than those of the TXA group (p < 0.05).
Conclusion:
The administration of IV-TXA resulted in a significant reduction in total blood loss, transfusion volumes, and transfusion rates without the increase of thromboembolic complications. Moreover, it was confirmed that TXA would not change the coagulation via the TEG analysis.
Introduction
Total hip arthroplasty (THA) is an efficient treatment that can relieve pain, improve hip function, and correct deformity in patients with end-stage hip osteoarthropathy. However, considerable blood loss during surgery, subsequent acute anemia, and a substantial risk of blood transfusion are still major concerns for joint surgeons. 1 Besides, THA is also complicated by fibrinolysis that continues through the postoperative period.
Venous thromboembolism (VTE), including deep venous thrombosis (DVT) and pulmonary embolism (PE), is a grave threat to patients undergoing total joint replacement. Reports have shown that the rates of VTE after total joint arthroplasty can be as high as 60% without proper prophylaxis. 2
Tranexamic acid (TXA) is a synthetic analog of amino acid lysine, which can inhibit fibrinolysis by competitively blocking lysine-binding sites of plasminogen, resulting in decreased proteolytic action on the fibrin monomers and fibrinogen, which ultimately results in clot stabilization. TXA has gained popularity in reducing perioperative blood loss, particularly after the publication of a trial in high-risk cardiac surgery. 3 Many previous studies have demonstrated the efficacy of TXA in decreasing blood loss and blood transfusion rate after THA. 4 –6 No increased risk of DVT or PE has been reported in several meta-analysis studies on the use of TXA in arthroplasty, even at higher and repeated dosages. 7 –9 Despite the evidence from several studies that shows no associated increased VTE complications due to TXA, theoretical concerns do exist because of a greater risk of thromboembolic complications with the use of TXA due to its antifibrinolytic activity. 10
Most commonly, routine laboratory-based coagulation tests (e.g. prothrombin time (PT)/international normalized ratio (INR), activated partial thromboplastin time (aPTT), fibrinogen), and platelet numbers are being used to assess the patients’ coagulation status. 11 However, the value of these tests has been questioned; for coagulation tests are determined in plasma rather than whole blood; no information is available on platelet function; and the assays are performed at a standard temperature of 37°C rather than the patient’s temperature.
Thrombelastography (TEG) was first described by Hartert in 1948 as a method to assess the global hemostatic function from a single blood sample. 12 TEG is a rapid and readily available whole-blood assay potentially capable of identifying both hypocoagulable and hypercoagulable states. 11,13 –15 It offers an all-around analysis of a patient’s coagulation status which was completely different from plasma-based tests, including the contribution of platelets, coagulation factors, fibrinogen levels, erythrocytes, and leukocytes. 16 Furthermore, the clot development can be visually displayed in real-time and the coagulation analysis can be done at the patient’s temperature. Hypercoagulability can be reflected by the changes of TEG parameters, that is, reaction (R) time and clotting (K) time decrease, but α-angle and maximum amplitude (MA) increase. 17 Hypercoagulable trauma patients who will develop VTE can be identified by TEG but not be found from PT and aPTT. 18,19
The objective of this study was to determine if there was a difference in the TEG profiles after the use of TXA in primary THA patients and to assess whether the use of TXA leads to the changes of thrombosis frequency. We hypothesized that (1) the application of TXA does not increase the risk of thrombosis and (2) TXA can reduce blood loss with no significant hemodynamic abnormalities.
Patients and methods
This study received approval by the Ethics Committee of our hospital, and all patients provided written informed consent for participation. From September 2016 to July 2018, all consecutive patients diagnosed as having osteoarthritis or osteonecrosis of the femoral head undergoing primary unilateral cementless THA at our hospital were recruited in this prospective clinical trial. Patients were excluded if they had an allergy to TXA, had a history of hypercoagulation, hemophilia, DVT, PE, or lower limb fracture in the previous 6 months, had been treated with warfarin, heparin, or estrogen before surgery; or had hepatic or renal dysfunction. Before surgery, patients were divided into two groups by a computer-generated randomization method: the TXA group and the control group. Patients assigned to the TXA group were given intravenous (IV)-TXA at 15 mg/kg of bodyweight 30 min before skin incision. Calculated dose of TXA was mixed in 100 mL of isotonic sodium chloride solution and given as a slow IV injection. Meanwhile, the control group only received the same dosage of normal saline at the same time. The patients, attending surgeons, anesthesiologists, and outcome assessors were blinded to the randomization.
Surgical procedure and perioperative management
All THA procedures were performed by the same senior surgeon using the posterolateral approach. After the prosthesis was implanted, joint capsule, gluteus medius, piriformis, gluteus maximus, deep fascia, and skin were closed subsequently. A closed-suction drain was inserted into the hip joint. The drain was clamped for 2 h and then released, and it was removed 48 h after the surgery. Routine preoperative investigations included a full blood-cell count, blood urea, electrolytes, chest radiograph, electrocardiography, echocardiogram, and pulmonary function test. Routine plasma-based coagulation tests, TEG, and routine blood test were performed on each patient before operation, on the postoperative days 1 and 7, respectively.
Prophylaxis against VTE was a combination of mechanical and chemical thromboprophylaxis at our center. As mechanical prophylaxis, intermittent pneumatic compression devices were applied after anesthesia resolution. As chemical prophylaxis, a half-dose of low molecular-weight heparin (2000 IU in 0.2 mL; Clexane; Sanofi-Aventis, Paris, France) was first subcutaneously administered 6 h postoperatively and then at 24-h intervals with a full dose (4000 IU in 0.4 mL) until hospital discharge. After discharge, 10 mg/day of rivaroxaban (Xarelto; Bayer, Leverkusen, Germany) was prescribed for 15 days for prophylaxis against VTE if no bleeding events occurred. The patients were examined daily in the hospital for any clinical symptoms of VTE. Doppler ultrasound of both lower limbs was performed routinely to detect DVT 1-day preoperatively, 1-week postoperatively, and at 30-day follow-up, or when there was a clinically suspected DVT during the follow-up. VTEs were defined by the development of either PE or DVT during the study period. DVTs were defined by the presence of symptomatic DVT confirmed by venous duplex. PE was defined by a clot detected by helical computerized tomography angiography of the chest which was based on clinical suspicion of unexplained tachycardia, tachypnea, chest pain, and/or hypoxia.
Allogeneic blood transfusion was indicated if the hemoglobin drops to less than 7.0 g/dL or if any symptoms suggestive of anemia such as dyspnea or tachycardia persist in spite of volume replacement therapy. 4 Accordingly, 1 U of packed red blood cells was administered and the patients’ symptoms should be monitored.
Data collection
Demographic data such as age, sex, weight, height, and the surgery time were collected. The intraoperative blood loss, the amount of postoperative drainage, and blood transfusions were also recorded to estimate the blood loss. The total blood loss and changes in hemoglobin, hematocrit, and platelet concentration, amount of drainage and intraoperative blood loss, transfusion frequency, and number of blood units transfused were compared between the two groups. The incidence of DVT and other complications that occurred during hospitalization were also recorded.
Routine CIs include PT, aPTT, INR, thrombin time (TT), and
Perioperative total blood loss was calculated by the Gross equation (Females’ total blood loss in milliliters = [0.3561 × H 3 + 0.03308 × W + 0.1833] × [Hctpreop−Hctpostop], males’ total blood loss in milliliters = [0.3669 × H 3 + 0.03219 × W + 0.6041] × [Hctpreop−Hctpostop], H (m): Height, W (kg): Weight, Hctpreop and Hctpostop (mL): the hematocrit (Hct) values before surgery and the lowest postoperative level). 21 If a reinfusion or an allogenic transfusion was performed, then the total blood loss is equal to the loss calculated from the change in the hematocrit plus the volume transfused. The hidden loss was determined by subtracting the visible blood loss from the calculated total blood loss and then plus the volume transfused. 22
Statistical analysis
All data analyses were performed using SPSS version 22.0 (IBM, Chicago, Illinois, USA). Continuous data with a normal distribution were presented as mean ± standard deviation. Continuous variables were compared between the two groups with the use of the Student’s t-test. One-way analysis of variance was used for comparison of continuous variables among the three time points of each group. The Pearson χ 2 test and the Fisher exact test were performed to analyze the qualitative variables. A p-value <0.05 was set as statistically significant.
Results
A total of 174 patients with a mean age of 58.9 years (range, 27–83 years) were enrolled in the study. Among these patients, 88 patients were randomly assigned to the control group, while 86 were in the TXA group. The mean age, body mass index, and gender distribution were similar between the two groups
Baseline demographics (mean ± SD).

SD: standard deviation; TXA: tranexamic acid; BMI: body mass index; DVT: deep vein thrombosis.
In the control group, 8 of the 88 enrolled patients had postoperative VTE, including 1 proximal DVTs and 7 DVTs in the calf, whereas in the TXA group, 9 of the 86 enrolled patients had VTE, including 2 proximal DVTs and 7 DVTs in the calf. All of the 17 patients confirmed with VTE were asymptomatic. The difference in the incidence of VTE between the two groups was not significant (p = 0.803).
The transfusion rate, the amount of postoperative blood transfused, total blood loss, and the amount of drainage were significantly lower in the TXA group than those in the control group. However, there was no significant difference between the two groups with regard to the hidden blood loss, visible loss, or the calculated blood loss ( Table 2 ).
Perioperative blood loss outcomes (mean ± SD).

SD: standard deviation; TXA: tranexamic acid.
*p < 0.05.
The TEG variables and conventional laboratory values of the two groups were shown in
TEG variables and conventional laboratory values (mean ± SD).
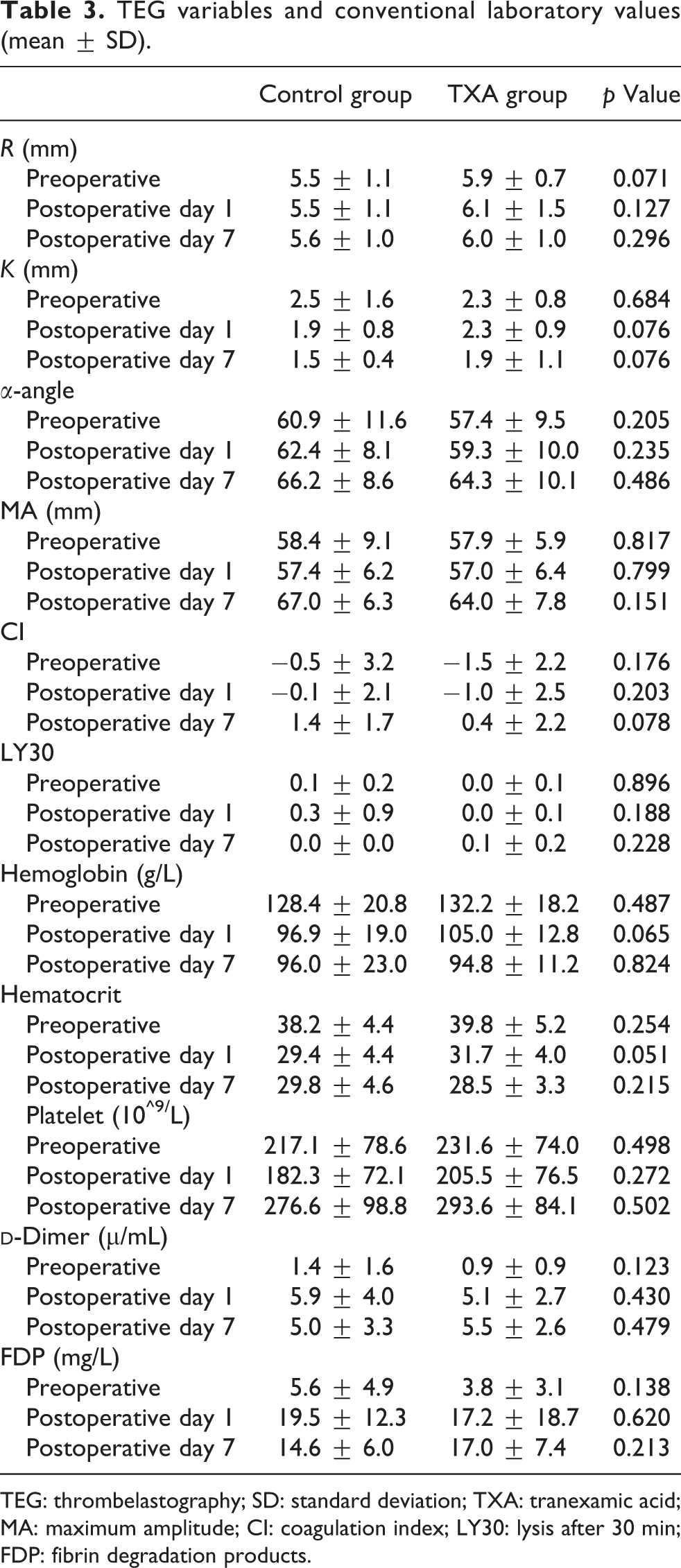
TEG: thrombelastography; SD: standard deviation; TXA: tranexamic acid; MA: maximum amplitude; CI: coagulation index; LY30: lysis after 30 min; FDP: fibrin degradation products.
Discussion
Postoperative blood loss and subsequent blood transfusion were still major concerns for THA. 23 In the present study, it was found that blood transfusion and total blood loss both decreased without the increase of the thrombosis rates after the application of TXA in patients undergoing primary THA. There was no significant difference in TEG variables or other laboratory tests between the two groups. To the author’s knowledge, this study provides the first evidence of the hemostatic changes for the application of IV-TXA with the use of TEG.
TXA has been used in arthroplasty for over 20 years to decrease blood loss and transfusions. 24 Previous reports have demonstrated the benefits of TXA in reducing blood loss without an increase in thromboembolic complications. 25,26 In Wang’s study, 5 a total of 124 patients undergoing THA were randomized into three groups: control (placebo), 10 mg/kg IV-TXA, and 15 mg/kg IV-TXA. They found that IV-TXA at 10 mg/kg can significantly reduce the total blood loss and mitigate the decrease in hemoglobin and hematocrit after THA, but it did not significantly reduce the need for transfusions. In contrast, a dose of 15 mg/kg reduced both bleeding and transfusion requirements. Based on Wang’s results, 5 in the present study, patients assigned to the TXA group were given IV-TXA at 15 mg/kg of bodyweight. We also found that IV administration of TXA at 15 mg/kg could reduce the postoperative drainage, the amount of postoperative blood transfused, and the total blood loss but not hidden blood loss, which was inconsistent with Good’s results. 27
Despite the benefits of TXA use in THA, the consequent prevention of clot dissolution, while beneficial for the hemorrhaging trauma patients, may potentially heighten risk of VTE by promoting thrombus formation. 28 In Myers’s study, TXA was associated with more than threefold increase in the odds of VTE (adjusted odds ratio, 3.3; 95% confidence interval, 1.3–9.1; p = 0.02), and TXA was regarded as an independent risk factor for VTE. 29 In the present study, in contrast, it was found that TXA does not increase the rates of thromboembolic events after THA. However, the hemodynamic evidence of whether there were hemodynamic changes after TXA application is still lacking.
Although routine plasma-based coagulation tests are easily available and cheap, they are not ideal for quantitative comparison between patients with TXA use and those without due to a discreet variation compared to normal values and a high dependency on the reagent used.
30
In the present study, no significant difference was observed in terms of the aPTT, PT, INR, TT, and
TEG can provide the entire phase of the dynamic changes in viscoelasticity property of the clot, 31 and it was regarded as a better indicator of hypercoagulable state after injury than plasma PT or aPTT. 32 Moreover, clot strength, fibrinolysis, hypercoagulability, and platelet dysfunction can also be reflected by TEG. 33 In Doran’s study, 34 it was found that 10% of all patients were coagulopathic according to standard laboratory testing, compared with 64% detected by an abnormal TEG on admission (p = 0.0005). Van et al. also thought that TEG on hospital admission could distinguish trauma patients in hypercoagulable state. Yang et al. 35 reported that TEG was effective to distinguish hypo- and hypercoagulability in patients following THA. Despite the wide use of TXA in the perioperative period of THA, the published reports mainly concentrated on the prevalence of DVT, the blood loss, the mortality, etc. 36 –39 After the administration of IV-TXA, whether the blood coagulation will be changed, especially evaluated by TEG has not been figured out. Hypercoagulability is defined as CI >+3. In both groups, CI reached the highest level 7 days postoperatively, so did MA value. Despite the hypercoagulable tendency in both groups, we identified that all the TEG variables in the TXA group were comparable with those of the control group. Even evaluated by TEG, the change of patients’ coagulation state was not significantly influenced by TXA.
There were several limitations in this study. First, the Doppler ultrasound was only performed the day before operation, 7 days after the operation, and at 30-day follow-up. VTEs that happened later were not obtained. However, the seventh day after lower extremity surgery was the peak time for clinically important thrombosis. 40 So it may be reasonable to take this follow-up interval for collecting VTE data. Second, the population of the study was relatively small, thus the possibility of type II error increased. Third, we estimated blood loss using the Gross equation method. Even though this equation theoretically includes both intraoperative and hidden blood loss, we recognized that true calculated blood loss may be influenced by factors such as hemodilution and fluid shifts. Fourth, the favorable dose of TXA remains controversial. As previous reports demonstrated that the use of repeat doses, higher doses, or variation in timing of administration did not significantly reduce blood loss or risk of transfusion. 41 Wilde et al. 42 also reported that one dose of TXA was as effective as two doses for decreasing blood loss and transfusion rates after THA and TKA without an increase in complications. Thus, in the present study, 15 mg/kg IV-TXA prior to skin incision was applied in this investigation. Finally, we acknowledge the marked limitation that the half-life of TXA systemically was not taken into account. The half-life of IV trenaxemic acid is approximately 3 h. The ideal time point to obtain the TEG profiles is within this time frame and before the administration of thromboprophylaxis. However, due to concerns of the harm caused by the frequent blood draws, the blood tests including TEG profiles were performed on the first day after surgery, when it was more than 10 h since the end of the operation, and it was not considered as a primary variable. In addition, considering the postoperative seventh day which was the peak time for clinically important thrombosis, there was no difference with regard to either the incidence of VTE or the blood coagulation via the TEG analysis between the two groups at the first day after operation and seventh day after operation.
In conclusion, IV administration of TXA may be reliable to significantly reduce total blood loss without altering TEG values or increasing the risk of thrombosis. However, further research regarding the exact dosage and timing of combined IV, oral, and topical TXA use will be necessary in the future.
Footnotes
Authors’ contributions
X-cZ, M-jS, and SP contributed equally to this work.
XZ and K-jG designed the study and drafted the manuscript. X-cZ, M-jS, and SP performed the experiments and the statistical analysis. MR, F-cZ, G-cZ, and YP participated in the study design and data collection. XZ and K-jG interpreted the data and critically reviewed the paper. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has received support from the National Natural Science Foundation of China (81902244 and 81672184), the Research project of Jiangsu Provincial Health Department (H201528), Jiangsu Provincial Medical Youth Talent (QNRC2016801), and Xuzhou Science and Technology Innovation Project (KC19063).
