Abstract
The use of human tissue-derived autografts and allografts continues to be the gold standard in anterior cruciate ligament (ACL) repair. However, autografts and allografts have their own set of associated risks. Many alternative options, including synthetic replacements, have failed to demonstrate long-term success. In this study, sterile acellular porcine bone-tendon-bone (BTB) xenografts were created using a proprietary process and tested against BTB autografts in goats for 13 and 52 weeks. At 13 weeks, all xenograft-implanted animals (n = 9) had subjective hind leg motor function (HLMF) that was categorized as either normal (score = 0) or a slight limp (score = 1) compared with two of nine autograft-implanted animals having a moderate limp (score = 2). At 39 weeks, there was HLMF improvement with each autograft-implanted and xenograft-implanted animal having normal HLMF or only a slight limp. At 13 weeks, six of nine animals in each group achieved normal anterior drawer scores, which increased to nine of nine animals in each group by 39 weeks. Both autografts and xenografts exhibited minimal inflammation with excellent integration of the fibrous tendon portion of the graft to host bone, as evidenced histologically by Sharpey’s fiber formation. At 52 weeks, maximum mechanical load at failure for xenografts was 1092.0 ± 586.4 N compared with 1037.0 ± 422.6 N for autografts. These results demonstrate that a sterile acellular porcine BTB xenograft can perform equivalently to BTB autograft in a caprine model of ACL repair.
Introduction
The anterior cruciate ligament (ACL) is an essential structural component of a knee joint but also the most commonly injured ligament of the knee. ACL injury rates are higher in women and are increasing overall because of an aging population, 1,2 heightening the need for effective, convenient treatment options. Traditional nonsurgical approaches to managing ACL injuries can be effective; however, without surgical repair, the injured knee is generally unstable, so both the ACL and surrounding tissues are prone to further injury. 3,4 A damaged ACL that does not heal on its own usually requires surgical reconstruction to restore knee joint stability and an active lifestyle. 5,6
Although many surgical options are available for ACL reconstruction, the gold standard continues to be the use of autografts and allografts. 7 –11 However, complications with autografts include donor site morbidity, decreased range of motion, anterior knee pain, and joint weakness. 12,13 Allografts circumvent donor site morbidity but present their own risks, including disease transmission, infection, and host immunogenic response/rejection. 14,15 Allograft sterilization can reduce infection risk but can induce adverse biological responses to graft material and alter mechanical properties of the graft. 16 –21 Moreover, the supply of allograft tissue is limited. 22
A variety of graft alternatives have been or are currently being developed, including those derived from biodegradable natural polymers (collagen, silk, hyaluronic acid, and chitosan) and biodegradable synthetic polymers (polyglycolic acid, polylactic acid, and polycaprolactone). 23 –30 Application of nondegradable synthetic materials, such as the polypropylene Kennedy ligament augmentation device, polyethylene terephthalate Leeds-Keio ligament, and polytetrafluoroethylene Gore-Tex®, has been attempted with limited success. Although synthetic devices offer superior strength at fixation, they may degrade over time and often fail because of foreign body-induced inflammation with synovitis and effusion, typically requiring further surgical intervention. 31,32
Xenografts are becoming a viable alternative to autografts 32 –37 because of lack of donor site morbidity and adequate repair strength, but are subject to immunorejection, foreign body encapsulation, and degradation, which may result in a weak repair. 38 The ideal alternative to the autograft would be derived from an unlimited source, be nonimmunogenic, and fully integrate with the host tissues, ensuring a functional, strong ACL replacement.
For a xenograft to perform successfully as an ACL replacement, there are a few critical factors. The xenograft must be processed and sterilized in a manner that retains the native collagen structure, integrity, and strength. All cells, cellular debris, and DNA must be gently removed, and the galactose-α-1,3-galactose (α-gal) antigen must be reduced to lower the immunogenic response. Accomplishing these key attributes in a xenograft enhances graft integration and, ultimately, long-term graft performance. Tensile strength is another important factor to ensure xenografts can withstand the load required to stabilize the knee joint. Creep testing is critical to ensure that xenografts do not elongate and lose the appropriate designed tension under relevant surgical procedures and rehabilitation programs.
In this study, histologic, immunohistochemical, thermal, and biomechanical properties of a porcine patellar tendon that had been decellularized using a proprietary process were characterized. In addition, histologic host response, tissue integration, repair strength, and overall functionality were assessed in a caprine model of ACL replacement.
Methods
Study design
A caprine model of ACL repair was used based on its well-established use in ACL reconstruction techniques. Animals were cared for as described in the Guide for the Care and Use of Laboratory Animals, as published by the Institute for Laboratory Animal Research and in accordance with Standard Operating Procedures of the test facility. The study protocol was reviewed and accepted by the test facility Institutional Animal Care and Use Committee, certifying that all animals would be appropriately used, receive acceptable levels of care, and would not be subjected to pain or distress needlessly.
Unilateral ACL repair was performed on the right hind leg of 18 female goats to limit variability in a surgical procedure. Animals were approximately 3–5 years of age and weighing 40–50 kg with an untreated bone-tendon-bone (BTB) autograft for 3 months (n = 2), untreated BTB autograft for 12 months (n = 7), porcine BTB xenograft undergoing proprietary processing for 3 months (n = 2), or porcine BTB xenograft undergoing proprietary processing for 12 months (n = 7). Animals were allocated for either gross and histological joint evaluation or biomechanical testing.
Caprine and porcine BTB source animals were given glycopyrrolate (0.02 mg/kg) and buprenorphine (0.03 mg/kg) intramuscularly (IM) as well as flunixin meglumine (2.2 mg/kg) IM and cefotaxime (50 mg/kg) intravenously (IV) prior to surgery, with additional cefotaxime given every 90 min during surgery. Anesthesia was induced using a combination of dexmedetomidine (0.01 mg/kg) and ketamine (10 mg/kg) given IM. The animals were intubated and anesthesia/sedation was maintained by sevoflurane. Animals receiving a BTB implant were given fentanyl (2 μg/kg/h) transdermally over the first 3 days postoperatively.
Preparation of autografts
A unilateral anteromedial open approach was used to access the caprine right hind leg knee joint. An incision was made through the fascia over the medial aspect of the patellar tendon, exposing its lateral and medial margins. Incisions were made approximately 6–8 mm apart along the middle one-third of the patellar tendon, allowing for a partial-thickness resection. Osteotomies approximately 6 mm in diameter and 12 mm long were performed at the patellar and tibial insertions to harvest the BTB. Autografts were wrapped in gauze and soaked in saline with cefotaxime until ready for placement.
Preparation and characterization of xenografts
Porcine BTB xenografts were harvested from the middle one-third full thickness of the patellar tendon in right and left knee joints of 6-month-old pigs and subsequently put through a proprietary process to remove cells, cellular debris, and DNA, and to reduce the α-gal antigen. The xenograft was sterilized using supercritical carbon dioxide (NovaSterilis, Lansing, New York, USA). Cell removal was confirmed histologically via hematoxylin and eosin (H&E) staining, and α-gal antigen reduction was confirmed immunohistochemically using a biotinylated lectin I IB4 antibody (Sigma-Aldrich, St Louis, Missouri, USA). DNA clearance was confirmed using the Quant-iT™ PicoGreen® dsDNA assay kit (Invitrogen, Eugene, Oregon, USA) per manufacturer’s instructions. Differential scanning calorimetry (Q2000 thermal analyzer, TA Instruments, New Castle, Delaware, USA) was used to determine collagen integrity of the extra-cellular tendon matrix (ECTM). Mechanical tests were performed using an Instron 8874 servo-hydraulic tensile tester (Instron, Norwood, Massachusetts, USA) with custom grips and 5-kN load cell.
To confirm that ECTM strength was maintained following processing and sterilization, fresh frozen (control) and processed BTB xenografts were tensile tested to failure at a strain rate of 25%/s while recording maximum load (N), maximum stress (MPa), and stiffness (N/mm). Tendon thickness was measured at proximal, medial, and distal points using a custom jig, which applied a 0.12 MPa stress to the tendon for a total of 2 min, and a digital micrometer recorded the data. Bone plugs were loaded into a custom fixture with polyester resin (Bondo®; 3M, St Paul, Minnesota, USA), isolating the tendon portion for testing.
To confirm that BTB fixation strength was maintained following processing and sterilization, fresh frozen (control) and processed BTB xenografts were tested to failure at a strain rate of 25%/s while recording maximum load (N) and method of failure. The tibial bone plug was secured to a solid polyurethane block with a titanium Arthrex® interference screw, the patellar bone plug loaded and sealed into a custom fixture with polyester resin, and the entire BTB construct pulled to failure.
To understand the level of preloading necessary for the xenograft prior to implantation, a creep and relaxation test protocol was devised. BTB xenografts were subjected to 20 cycles of 0–50 N. On the 21st cycle, samples were held at a constant elongation for 30 min while recording decreases in applied load as the tissue relaxed. This test was used to model load relaxation in the xenograft following our intraoperative preconditioning protocol prior to implantation. Creep elongation is reported as average and standard deviation (SD), while a decrease in load is reported as percent load decrease.
ACL grafts are expected to perform under cyclic load extensively after surgery and during rehabilitation. It is important to understand the fatigue life of the graft in the period prior to full regeneration of the tissue and continued homeostasis of the graft following full regeneration and complete healing. We tested the cyclic fatigue life of the xenografts using Food and Drug Administration guidelines. 39 Samples were cycled until failure at 4 Hz and 37°C in a saline bath over three clinically relevant load levels similar to Dargel et al. 5 : descending stairs 350 N (n = 6), normal level walking 200 N (n = 5), and ascending stairs 100 N (n = 1). Tendon thickness was measured, as described above, to record maximum stress and number of completed cycles at failure. Data are reported on a per-sample basis.
Graft implant procedures
For animals receiving BTB grafts, an incision was made through the fascia over the medial aspect of the patellar tendon, exposing its lateral and medial margins. The native ACL was excised. ACL-bone insertion sites were used as a guide for tibial and femoral bone tunnel creation. Bone tunnels were drilled using a 4.5 mm bit and then widened to approximately 6.0 mm. Notchplasty was performed to create smoother tunnel openings using a 4-mm oval burr. Final bone tunnels measured approximately 12 mm deep. Approximately 30 min prior to placement, xenografts were trimmed to smaller than 6 mm. Using a custom preconditioning device, each xenograft was submerged in sterile saline at room temperature and subjected to a load of 10 lb (4.5 kilogram-force (kgf)) for 30 min. The proximal bone plug was passed sequentially through the tibial and then femoral bone tunnels and fixed within the femoral bone tunnel under 5 kgf (approximately 50 N) using at least one K-wire. Next, the graft was held under 5 kgf while the joint underwent 20 flexion cycles. Finally, the distal bone plug was fixed under 5 kgf (approximately 50 N) using at least one K-wire.
Isometric placement of the BTB was checked intra-articularly through the entire range of motion to ensure no graft impingement or deformation of greater than 2 mm. Anterior drawer motion at approximately 30–45° flexion was evaluated and recorded. Finally, the joint was closed sequentially in layers. Approximately 1 ml of bupivacaine and cefotaxime (400 mg/ml) were injected into the knee joint to provide postoperative analgesia and antibiotic prophylaxis.
In-life observations
Animal weights were recorded prior to surgery and monthly following surgery. Two separate assessments developed by the study surgeon with over 30 years of experience performing ACL reconstruction surgeries in this model were used. The first was a subjective hind leg motor function (HLMF) score that was used to assess limb function based on animal limp severity. Limbs were scored on a 0–3 point scale and evaluated preoperatively, once daily for the first 14 days, and then at 13, 26, 39, and 52 weeks postoperatively. The second was a semiquantitative knee joint function (KJF) assessment made up of a compilation of clinically relevant observations. KJF was performed preoperatively and postoperatively at 13, 26, 39, and 52 weeks and included scoring of the following: knee size (0 = normal/1 = slightly enlarged/2 = moderately enlarged/3 = greatly enlarged), range of motion (full extension to full flexion degree of motion, measured with a goniometer), patellar status (as judged by a normal, medial, or lateral subluxated placement), and anterior drawer motion (0 = normal (0–3 mm)/1 = slight drawer (4–6 mm)/2 = moderate drawer (7–9 mm)/3 = severe drawer (>9 mm)).
Following euthanization, synovial fluid from both knee joints was aseptically collected (as available) and characterized. Samples were sent for culture and cytology evaluation (IDEXX Laboratories, Westbrook, Maine, USA).
Termination
Animals were sedated using a combination of ketamine (15 mg/kg IM) and xylazine (0.1 mg/kg IM). After final knee evaluations, euthanasia was performed via overdose of euthanasia solution (sodium pentobarbital, approximately 100 mg/kg IV) in accordance with American Veterinary Medical Association guidelines. 40
Histology
Following termination and subsequent gross knee joint evaluation at 13 and 52 weeks, whole knee joints were excised en bloc, fixed in formalin for at least 4 days, decalcified, sectioned, and stained with H&E and Masson’s trichrome (MT). Cross sections of the implanted femoral and tibial bone plugs were taken to assess integration with the surrounding femoral and medial tibial cortex, respectively. Tendon integration was assessed using cross sections of the repaired ACL mid-substance as well as the bisected femoral and tibial insertion sites. Inflammatory response of the grafts was assessed using semiquantitative method by an external board-certified veterinary histopathologist: minimal (no or a few inflammatory cells), mild (local and <10% of areas), moderate (local and <30% of areas), and significant (spreading and >30% of areas).
Knee joint tensile testing
Knee joints from all animals surviving to 52 weeks were stored frozen at –80°C until tested. Joints were thawed, trimmed to isolate the reconstructed ACL, placed in a custom jig, and tensile tested to failure at 24.5 mm/min at 30° of flexion using an Instron 5865 electromechanical tensile tester outfitted with 5-kN load cell. Maximum load at graft failure was recorded and reported as mean ± SD.
Statistical analysis
Data were collected and analyzed to determine average and SD. Statistical significance was assessed by Student’s t-test, α = 0.05.
Results
Proprietary processing of xenografts successfully eliminated or reduced immunoantigens typically associated with graft rejection. Processed xenografts (n = 4) showed no cell remnants in the ECTM with significantly reduced presence of α-gal (Figure 1). DNA analysis revealed >99% DNA removal from processed xenografts (n = 4) compared with fresh frozen (control) xenografts. The collagen integrity of xenografts (n = 3) was confirmed by equivalent thermal denaturation profiles (onset and peak melting temperatures) for fresh frozen (control) and processed ECTM (Table 1), indicating negligible ECTM thermal property changes.

Histology assessment of xenograft ECTM before and after proprietary processing: (a) H&E stain of fresh frozen (control) ECTM with cells present (×100); (b) processed ECTM with no cells present and crimped collagen pattern (×100); (c) MT stain of processed ECTM with deep blue color; (d) lectin stain of fresh frozen (control) ECTM showing positive brown staining for α-gal antigen (×100); and (e) processed ECTM showing a significant decrease in α-gal antigen staining (×100). ECTM: extra-cellular tendon matrix; H&E: hematoxylin and eosin; MT: Masson’s trichrome.
In vitro assessment of fresh frozen (control) and processed porcine BTB xenografts.
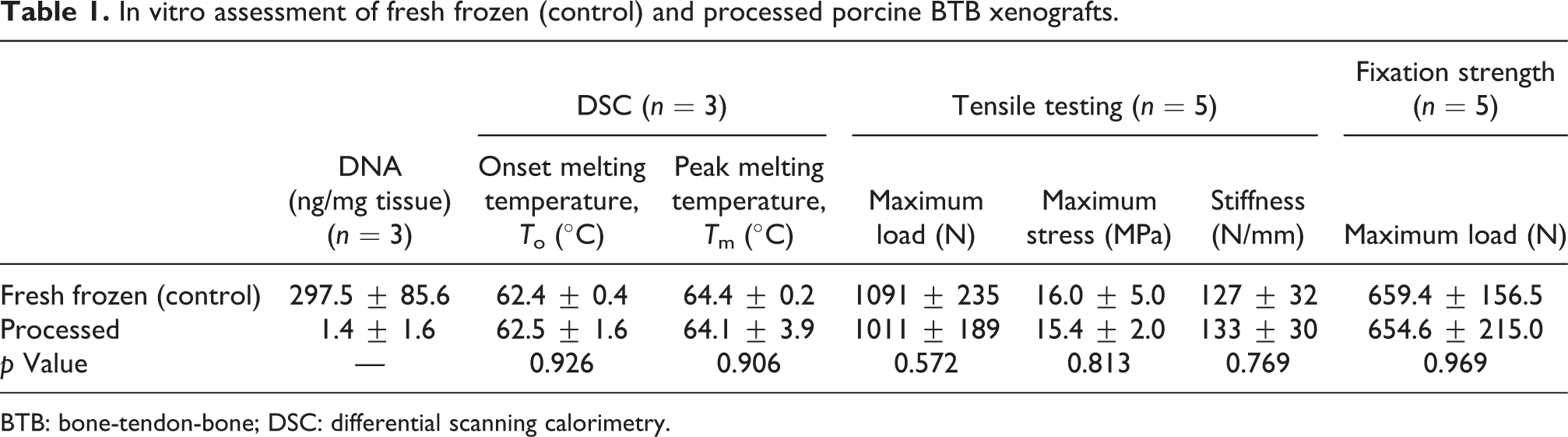
BTB: bone-tendon-bone; DSC: differential scanning calorimetry.
Tensile testing of fresh frozen (control) and processed xenografts showed no statistical differences in maximum load at failure, maximum stress at failure, or stiffness (Table 1). Fresh frozen (control) and processed xenografts also showed no statistical differences between groups in fixation strength (Table 1). All fixation samples tested failed at interference screw, suggesting that our processing and sterilization techniques do not affect xenograft mechanical strength.
To avoid laxity in xenografts following final fixation in the surgically repaired knee joint, cyclic creep followed by load relaxation testing was conducted. On average, processed xenografts elongated 4.4 ± 0.6 mm after 20 cycles at 50 N, which was followed by a 49% (±) decrease in load during relaxation (Figure 2). These results suggest that a preconditioning strategy to help reduce xenograft elongation and decrease load relaxation provides a more stable knee joint following surgery.

BTB xenografts were cycled from 0 N to 50 N 20 times. (a) On the 20th cycle, load and displacement were recorded for six distinct samples. (b) Following the 20th cycle, three distinct samples were pulled to a load of 50 N (21st cycle) and held at a constant elongation while the decrease in load (N) was recorded for 30 min. BTB: bone-tendon-bone.
Cyclic fatigue testing was performed at clinically relevant high (350 N, descending stairs), medium (200 N, normal level walking), and low (100 N, ascending stairs) loads on processed xenografts only. An inverse linear relationship was observed between maximum stress and number of cycles completed prior to failure. At 350 N, samples (N = 6) failed at approximately 340,000 cycles and an average stress of 5.0 ± 1.1 MPa with 9.5% (5.7 mm) deformation. At 200 N, samples (N = 5) failed at approximately 1.7 million cycles and an average stress of 3.3 ± 0.4 MPa with 7.2% (4.1 mm) deformation. At 100 N, a single sample failed at approximately 6.8 million cycles and stress of 1.6 MPa with 20% (13 mm) deformation (Figure 3). With an average of 1.5–2 million gait cycles annually, these data suggest that the xenograft can support the knee joint during rehabilitation while the tissue is integrating.

Cyclic fatigue testing of porcine BTB xenografts at three clinically relevant load levels: 350 N (n = 6), 200 N (n = 5), and 100 N (n = 1). BTB: bone-tendon-bone.
Micrometry measured preimplant harvested autograft ECTM as 48.2 ± 2.6 mm long and 2.9 ± 0.6 mm thick with a cross-sectional area of 14.6 ± 3.2 mm2 while preimplant xenograft ECTM was 67.3 ± 4.1 mm long and 6.2 ± 1.1 mm thick with a cross-sectional area of 30.8 ± 5.4 mm2. Following preconditioning, xenograft ECTM stretched to a final length of 69.5 ± 3.9 mm. Although xenografts were significantly longer and thicker than autografts, the same surgical procedure was used to fix each graft type. These graft lengths were sufficient for the current preclinical evaluation.
In-life observations
All 18 animals tolerated surgery well and were transferred to farm pasture within 3 weeks postoperatively. All animals resumed normal food consumption and continuous weight gain.
All animals receiving xenografts were observed with normal HLMF or only a slight limp (score 0–1) at 13, 26, 39, and 52 weeks (Table 2). Of the animals receiving autografts, two of nine exhibited a moderate limp (score 2) at 13 weeks, while the remainder exhibited normal HLMF or a slight limp. By 26 weeks, one of the remaining seven autograft-implanted animals exhibited a moderate limp, while the others had normal HLMF or only a slight limp. By 39 and 52 weeks, all animals received a score of 0–1.
Hind leg motor function.

HLMF: hind leg motor function.
HLMF was scored on a 0–3 point scale (0: normal/full use; 1: slight limp; 2: moderate limp; 3: not weight bearing/nonuse/severe limp). Results are reported as the number of animals scoring in the (0–1) range or (2–3) range in each group at each timepoint.
Observation at 13 weeks revealed the knee size of autograft-implanted animals to be slightly larger compared with xenograft-implanted animals, but this was expected given the effect of donor site surgery. By 39 weeks, all animals presented normal knee sizes (score 0), although by 52 weeks, knee size in both groups had become enlarged. Anterior drawer scores were similar between autograft- and xenograft-implanted animals at all timepoints with anterior drawer score improvement to normal/near normal levels by 39 weeks and lasting to 52 weeks for both groups.
Preoperative range of motion in operated knee joints (N = 18) from full extension to full flexion was 126.0° ± 7.3°. Postoperative range of motion in the operated knee joint was slightly reduced in both autograft and xenograft groups (N = 9 per group), averaging 117.8° ± 10.9° and 112.8° ± 8.7°, respectively. By 13 weeks and continuing to 52 weeks, both autograft and xenograft groups recovered to preoperative range of motion. Patellar status was normal in all animals in both autograft and xenograft groups at each postoperative timepoint.
Gross pathology
There were no signs of infection, arthritis, or excessive inflammation in any animal at either 13 or 52 weeks. Analysis of the synovial fluid collected from a representative sample of implanted animals from both groups (n = 11) indicated no etiologic agents and cytologically normal synovial fluid in all cases at both timepoints. Both autografts and xenografts appeared intact with satisfactory tension maintained at 13 and 52 weeks. Grafts from both groups appeared similar in gross appearance to native ligament with complete coverage by synovial-like tissue at both 13 and 52 weeks. There was extensive fibrosis surrounding the patellar tendon autograft harvest site at both 13 and 52 weeks, which was not present in the xenograft-implanted animals. Both autograft ECTM and xenograft ECTM were clearly distinguished from the posterior cruciate ligament with no adhesions visible at 13 or 52 weeks. There were no signs of degenerative articular damage in any animal at any timepoint (Figure 4).

Gross pathology of the caprine knee: (a) autograft and (b) xenograft at 13 weeks postoperative; (c) autograft and (d) xenograft at 52 weeks postoperative with arrow showing clear distinction of native posterior cruciate ligament.
Histopathology
The attachment of autologous bone graft and host bone was observed by staining with MT. At 13 weeks, integration of the autograft bone plug with the surrounding trabecular bone could be visualized with the tibial bone plug firmly attached to the outer surface of the tibial diaphyseal cortex. At 52 weeks, autograft bone was well integrated with the host bone, although there was some evidence of autograft bone plug resorption at the femoral and tibial bone insertion sites. In contrast, there was no apparent attachment of xenograft bone with the femoral and tibial host bone, with fibrous tissue encapsulation of the xenograft bone at the femoral bone tunnel and tibial cortex sites at both 13 and 52 weeks. Few inflammatory cells were observed to infiltrate into the autograft–host bone interface at either 13 or 52 weeks. However, xenograft implantation induced mild-to-moderate interface inflammation at 13 weeks, subsiding to minimal-to-mild by 52 weeks (Figure 5).

Masson’s trichrome staining of BTB autografts and xenografts to host bone integration at 13 weeks and 52 weeks: (a) Autograft bone plug integration within the femoral bone tunnel at 13 weeks showing good connection with trabecular femoral host bone. Asterisk showing a small portion of fibrovascular tissue/residual graft. (b) Xenograft bone plug integration within the femoral bone tunnel at 13 weeks with arrowheads indicating fibrovascular tissue surrounding the bone plug. (c) Autograft bone plug integration within the femoral bone tunnel at 52 weeks with arrows indicating bone plug. (d) Arrow showing xenograft bone plug at 52 weeks seated external to the femoral bone tunnel with arrowhead showing fibrous tissue within the femoral bone tunnel. (e) Autograft bone plug integration on the tibial cortex at 13 weeks with arrow indicating reactive bone present and arrowheads indicating a focus of fat necrosis with associated inflammation, likely related to trauma. (f) Xenograft bone plug integration on the tibial cortex at 13 weeks showing fibrovascular tissue surrounding the plug. (g) Autograft bone plug (asterisk) integration on the tibial cortex at 52 weeks showing excellent integration and remodeling with the cortex. (h) Xenograft bone plug (asterisks) at 52 weeks on the tibial cortex transected by the K-wire (arrows) and surrounded by a fibrovascular capsule. BTB: bone-tendon-bone.
MT staining revealed clear tendon remodeling for both autografts and xenografts following implantation. Both autograft and xenograft tendons were observed to integrate well at the femoral and tibial insertion sites at 13 weeks with clear formation of Sharpey’s fibers between the host bone and fibrous tendon tissue (Figures 6 and 7), which became more robust by 52 weeks. Within the bone tunnels, tendon graft collagen also became more organized and aligned by 52 weeks. Remodeling and integration appeared to occur more rapidly within the femoral bone tunnel than at the tibial insertion, but the rate of remodeling appeared to be similar for both autografts and xenografts.

Masson’s trichrome staining of BTB autograft and xenograft tendon to host bone integration at 13 and 52 weeks. (a, b) Integration and remodeling of autograft tendon and xenograft tendon to femoral insertion site at 13 weeks, respectively. (c, d) Integration and remodeling of autograft and xenograft tendon to femoral insertion site at 52 weeks, respectively, with arrows (d) indicating path of insertion site along base of articular cartilage. (e, f) Integration and remodeling of autograft and xenograft tendon to tibial insertion site at 13 weeks, respectively. (g, h) Integration and remodeling of autograft and xenograft tendon to tibial insertion site at 52 weeks, respectively, with arrows (h) indicating path of insertion site. BTB: bone-tendon-bone.

Masson’s trichrome staining of (a, c) autograft and (b, d) xenograft tendon to host bone integration at 13 and 52 weeks, respectively, with arrows indicating tight junction between tendon and host bone and Sharpey’s fiber formation. BTB: bone-tendon-bone.
Histology of tendon xenografts external to the bone tunnel demonstrated extensive repopulation of the previously acellular porcine tendon with fibroblast-like cells and vascular structure formation. Tendon tissue remodeling was significant and appeared complete with a fully aligned dense collagen bundle structure by 13 weeks. Inflammation of tendon grafts was minimal for autografts and mild-to-moderate for xenografts at 13 weeks but decreased to minimal levels for all grafts by 52 weeks. Loose connective tissue adhesions between the graft and posterior cruciate ligament were negligible.
Knee joint tensile testing
Tensile test failure typically occurred in the intra-articular mid-substance of the tendon graft with one sample from each group failing at or near the femoral insertion site. One xenograft could not be tested because of damage occurring to the tibia during tensile test sample loading. Maximum load for autografts (n = 5) and xenografts (n = 4) was 1037 ± 423 N and 1092 ± 586 N, respectively (p = 0.881; Figure 8). 41

Tensile strength of autograft and porcine xenograft-repaired knee joints following 52 weeks of implantation. No statistical difference between the groups was observed (p = 0.881). Dotted line represents the average of native caprine knee joint strength (N = 10) from Buma et al. 41
Discussion
Many groups have investigated alternative treatments for the repair or replacement of damaged ACL tissue. The gold standard in ACL repair continues to be the autograft, however, donor site morbidity still remains, and recovery can be difficult. Although the use of allografts has become a viable option, low success rates and poor incorporation with host tissue, especially in young patients, have been reported. 42 Following graft choice, there are numerous additional factors that can profoundly affect clinical outcomes in ACL reconstruction, including graft placement, 43 fixation technique, 44,45 tensioning, 46 –49 and physical rehabilitation. 50,51
An ideal strategy for eliminating critical autograft- and allograft-associated complications while maintaining long-term functional strength may be the use of a xenograft. 33,34,36 In this study, we evaluated the feasibility of sterile acellular porcine BTB xenografts (which maintain extracellular matrix integrity) compared with caprine BTB autografts in a caprine ACL repair model. We observed equivalent or superior outcomes for xenografts compared with autografts, especially with regard to in-life functional assessments at early timepoints. There were no significant differences between groups in gross observations or mechanical tensile strength of the knee explants. Histologic findings revealed that both autograft and xenograft tendon remodeling were similar to host tendon structure over 52 weeks with minimal-to-moderate inflammation, and significant tendon–bone integration with robust Sharpey’s fiber formation within the bone tunnels. Bone-to-bone integration was evident for autografts but not xenografts. Based on these findings, we believe that an acellular porcine BTB xenograft has the potential to be an ideal off-the-shelf material for ACL reconstruction. However, the results presented herein are based on a relatively small sample size and a short time frame (N = 6 for each test sample at 52 weeks). An animal study with a larger study population and longer time frame to mimic clinical outcomes is necessary prior to commercialization of any xenograft device.
We have thoroughly characterized our BTB xenografts prior to implantation by demonstrating an intact acellular ECTM that retains functional mechanical strength after processing and sterilization. Furthermore, we have characterized the creep and load relaxation of the xenograft allowing for the reduction of laxity observed within the reconstructed knee via a static preconditioning strategy. This is an important aspect that, when overlooked, can have detrimental effects on the initial graft tension and knee stability as well as overall strength of the repair. 46 –49,51 Previous studies conducted in our laboratory without adequate preconditioning of grafts resulted in unfavorable knee repair, including graft laxity, excessive connective tissue attachment to the intra-articular tendon grafts, a slower tendon remodeling process, and an overall weaker functional recovery (data not shown). Following our thorough mechanical characterization of BTB xenografts here, we have optimized the preconditioning procedure and were able to overcome the previous issues related to inadequate graft tension in this study.
Our BTB xenografts enabled a more rapid recovery of knee function than the autograft, as demonstrated by lower HLMF scores, similar KJF scores, equivalent knee tensile strength, and less swelling early on than autografts. We observed extensive fibrosis surrounding the autograft patellar tendon harvest site and greater intra-articular fibrosis than xenografts. These issues related to the graft harvesting procedure suggest the benefits of using an off-the-shelf product to eliminate donor site morbidity.
Clinically, one of the fundamental requirements for mechanical remodeling and complete recovery of the knee joint following ACL repair is rapid integration of tendon graft with host bone, as demonstrated by Sharpey’s fiber formation. We have shown Sharpey’s fiber formation in the bone tunnels of both autograft and xenograft tendons. These data indicate strong biocompatibility of our BTB xenograft, fabricated using techniques that gently remove cells, clear DNA, and reduce the α-gal antigen while maintaining critical extracellular matrix components. Using a carefully selected processing method that removes specific antigenic materials while not inducing matrix damage allows for greater regenerative potential of the xenografts, evidenced by the excellent tendon to host bone integration as a result of minimal inflammation. Stone et al. 34 have previously demonstrated feasibility of a porcine BTB xenograft in a primate model. They were able to create an immunochemically modified and sterilized porcine xenograft that functionally incorporated into the surgical site without adversely affecting the biomechanical properties of the graft.
It is also interesting to note that autografts became thoroughly remodeled to form a completely new tendon with newly deposited and reorganized collagen, rather than maintaining the original graft collagen fibers, as demonstrated by MT staining. The detailed pathology and molecular mechanism of remodeling involved in this ligamentization process are unknown at this time. Further study and a better understanding of the different mechanisms involved for autografts and xenografts will help us design a more regenerative construct for ACL repair in the future.
Although xenografts did not demonstrate bone-to-bone healing as extensively as autografts, the overall strength of repair did not appear to be adversely affected. Since the bone plug is designed to function merely as an anchor for graft fixation, and not for mechanical load-bearing, it plays a minimal role in knee function once adequate tendon graft integration in the femoral and tibial bone tunnels has been established. Furthermore, the bone graft plugs are designed small enough to ensure that they do not cause bone weakness. Based on the data presented here, our BTB xenograft is a feasible alternative for restoring ACL function despite the less favorable bone-to-bone integration. In the case of autografts, it was surprising to observe bone plug resorption at 52 weeks despite substantial bone-to-bone integration. Although the mechanism of bone resorption and its functional necessity in bone remodeling is not clear, it is not anticipated to adversely affect ACL function for the reasons addressed above.
Taken together, our results suggest that a properly processed and sterile acellular porcine BTB xenograft can perform equivalently to BTB autograft in a caprine model of ACL repair. The use of such a xenograft provides significant advantages over autografts and allografts and should be considered as a viable alternative to the current standard of care in ACL repair.
Footnotes
Acknowledgments
We would like to graciously thank Dr Vince Mendenhall at Wake Forest Innovations, Preclinical Translational Services, for his technical expertise, and Dr Balakrishna Haridas from Device & Implant Innovations, Inc., for his invaluable knowledge and support. Editorial assistance was provided to the authors by Evidence Scientific Solutions, Philadelphia, PA, and funded by Allergan plc, Dublin, Ireland (prior to its acquisition by AbbVie). All authors are former employees of LifeCell Corporation and are employees of AbbVie.
Author contributions
JAL processed test samples, performed mechanical testing, designed and executed in vivo study, wrote the abstract/introduction/methods/results sections, and edited the discussion section. AH and MS advised on in vivo study design. NR and AD designed and performed experiments and analyzed the data. GM advised on the project and performed the cyclic fatigue testing and data analysis. HX designed the in vivo study, interpreted the histology results section, and wrote and edited the discussion. All authors contributed to the manuscript and approved the final version for submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by USAMRMC proposal 09104003.06, award W81XWH-09-2-0127.
