Abstract
Purpose:
Giant cell tumor of bone (GCTB) is a local aggressive bone tumor, histologically classified as intermediate malignancy. Recently, the RANKL inhibitor, denosumab, was developed as a novel and effective treatment option for GCTB. Since the risk of preoperative use of denosumab with curettage had been previously reported, this study aimed to investigate the relationship between recurrences and clinicopathological features associated with adjuvant denosumab treatment in GCTB.
Methods:
A total of 87 GCTB cases were treated at our institution. We reviewed 66 patients with conventional-type GCTB occurring in the extremities and analyzed 78 surgical treatments, including curettages and resections, with clinicopathological features and denosumab treatment.
Results:
GCTB lesions, including 66 primary and 12 recurring, underwent surgical treatment like curettage and resection. Recurrence-free survivals in 78 GCTB surgeries were 78.7% in 3 years and 71.9% in 5 years. In the resected cases of GCTBs, there was no recurrence either with or without denosumab. In curettage cases, 3-year recurrence-free survivals were 0.0% (n = 3) in preoperative treatment of denosumab, 66.7% (n = 6) in postoperative treatment, and 76.6% (n = 43) in no treatment. Interestingly, three preoperative treatment cases demonstrated low MIB-1 index despite 100% recurrence. The other clinicopathological factors did not contribute much to the risk of recurrence in curettage cases.
Conclusion:
Our findings revealed the use of denosumab in GCTB, prior to curettage, to possibly increase the risk of local recurrence. Together with previous reports, our finding might provide information for beneficial treatment of GCTB.
Brief introduction
Giant cell tumor of bone (GCTB) has been classified by the World Health Organization as an intermediate malignancy; it is a locally aggressive osteolytic bone tumor with frequent local recurrence. 1,2 Metastases to the lung, including implantation and malignant transformations are described to be rare. 3 Local recurrence rates in curettage and resection have been reported to be 16–50% and 0–12%, respectively. 4 –12 To reduce its local recurrence and preserve the adjacent joint, adjuvant treatments with high-speed burr, 4 phenol, 6,10 ethanol, liquid nitrogen, 6 and polymethyl methacrylate 4,6,10 have been reported. Histologically, GCTB is composed of mononuclear stromal cells highly expressing receptor activator of nuclear factor kappa-B ligand (RANKL), along with some rounded mononuclear cells expressing RANK, and evenly distributed osteoclast-like multinucleated giant cells expressing RANK. 13 These cellular components play a significant role in the osteolytic process, leading to bone destruction, by interacting with various factors. Denosumab, an anti-RANKL monoclonal antibody, exerts its effects by binding to RANKL, inhibiting bone destruction, and eliminating giant cells. 14 Several studies have reported denosumab to be an effective treatment option for aggressive and difficult cases of GCTB. 15 –20 However, despite the reported benefits, adjuvant treatment using denosumab has recently been shown to be associated with a risk of recurrence in curettage treatment of GCTB. 12,21 A phase 3 trial of adjuvant treatment using denosumab in surgical settings has been running in Japan since 2017; however, the study is still ongoing due to difficulty of collecting entries. 22 In surgical setting, the utility of adjuvant treatment using denosumab in GCTB remains controversial. Recently, we had described some immunostaining factors (MIB-1 and GPX-1) that were associated with the prediction of local recurrences of GCTB. 23 Other research groups had also reported various immunostaining factors (p53, cyclin D1, β-catenin, and Ki67 (MIB-1)) useful for predicting the recurrence of GCTB. 24,25 Therefore, to determine the utility of adjuvant therapy using denosumab in GCTB, the current study retrospectively analyzed the associations between recurrence status and clinicopathological factors, including denosumab and immunostaining factors, using 66 GCTB cases that occurred in the extremities, excluding the malignant ones.
Materials and methods
Patients
Clinicopathological data of GCTB were retrospectively collected and analyzed using a protocol approved by the institutional review boards (IRBs) at Juntendo University, Tokyo, Japan. Of the 87 GCTB cases treated at Juntendo University hospital from January 2003 to May 2019, 21 were excluded, due to locations on trunk, histological malignant transformation, development of distant metastasis, or being missing in follow-up. Finally, 66 cases and 78 surgical treatments were statistically analyzed with clinicopathological factors, including age, gender, sites, Campanacci grade (I, II, III), local adjuvant chemotherapy during surgery, pathological fracture at presentation, timing of denosumab (pre-curettage vs. post-curettage, pre-curettage vs. curettage only, post-curettage vs. curettage only), and immunostaining (Figure 1). The details of administration method regarding denosumab are shown in Supplemental Table 3B.
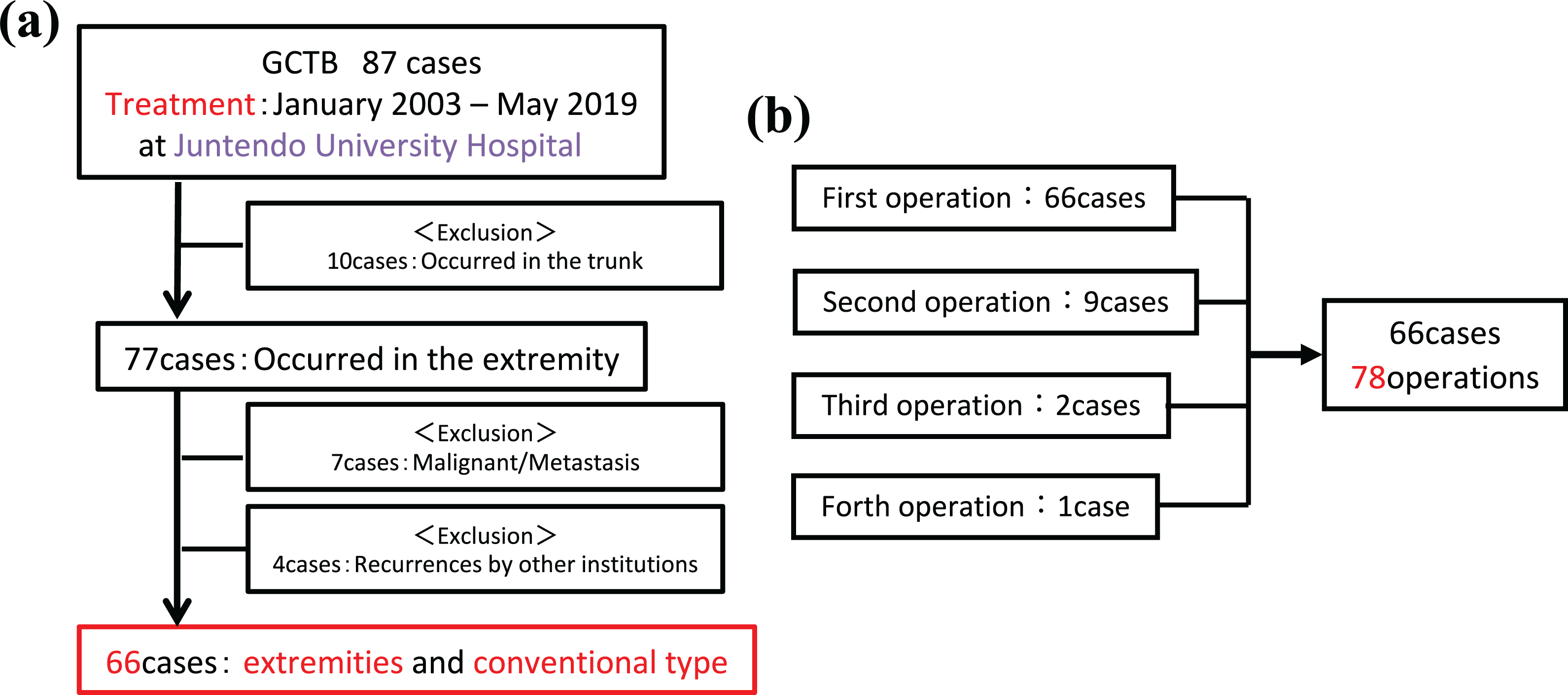
Analysis charts for this study: The cases considered in this study were analyzed retrospectively using a protocol approved by the Institutional Review Board at Juntendo University, Tokyo. (a) From January 2003 to May 2019, 87 cases of GCTB were treated at Juntendo University Hospital. We excluded 21 cases with occurrence in the trunk, histological malignant transformation, metastases, or lack of follow-up, thus including 66 cases in this study. (b) Regarding cases treated multiple times, each operation was counted as one treatment, and a total of 78 treatments were subsequently analyzed.
Immunohistochemical analysis
In 22 GCTB preoperative samples, immunohistochemistry was performed using anti-Ki-67 (MIB-1) antibody. As the clinical results of treatments regarding these 22 cases that were treated with curettages, 10 cases were developed local recurrence cases and 12 cases did not have local recurrence. Of those 10 cases that were developed local recurrences, 3 cases were treated with curettages with preoperative denosumab and 7 cases were treated with curettages without preoperative denosumab (either no denosumab or postoperative denosumab).
Immunohistochemical analysis was performed using the streptavidin–biotin method with monoclonal antibody against Ki-67 (clone: MIB-1, Dako, CA, USA) and 4-µm formalin-fixed paraffin-embedded tissues (FFPE) from the available GCTB samples. Immunostaining was assessed independently by two authors without prior knowledge of clinical pathology data. Any discrepancy was resolved by reevaluation until consensus was reached. Ki-67 labeling indices were evaluated in the most representative areas with the highest immunoreactivity, by counting the number of positive cells out of 1000 tumor cells.
Statistics
Associations between recurrences and clinicopathological factors were analyzed with Student’s t-test and χ 2 test, while differences of local survivals were analyzed with Kaplan–Meier and log-rank tests. Statistical analyses were performed using IBM SPSS Statistics 25.0 software (IBM, Armonk, NY, USA) with significance defined as p < 0.05.
Results
The characteristics of 66 GCTB cases were the following: gender, 34 males (52%) and 32 females (48%); average age, 35.3 years (14–73 years); and average follow-up, 58.8 months (7–183 months). Tumor locations of the 66 GCTB lesions consisted of distal femur in 28 (42%), proximal tibia in 12 (18%), proximal fibula in 7 (10%), distal radius in 5 (7%), distal ulna in 4 (6%), proximal humerus in 3 (5%), proximal femur in 2 (3%), foot in 2 (3%), proximal radius in 1 (2%), hands in 1 (2%), and distal tibia in 1 (2%). Of the 66 cases, 20 (30%) were in Campanacci grade I, 25 (38%) in grade II, 17 (26%) in grade III, and 4 (6%) were not evaluated (Table 1).
Characteristics of GCTB of 66 primary tumor patients who participated in this study.

Surgical treatments were performed on 78 GCTB lesions consisting of 66 primary tumors, 9 first recurrence tumors, 2 second recurrence tumors, and one-third recurrence tumor. In the primary tumors, 46 cases were treated with curettage and 20 with resection. Of the recurring tumors, six cases underwent curettage and six were resected. Surgical treatment was administered without tourniquets due to proximal humerus and proximal femur locations in 7 (9%) of the 78 lesions. Fifteen lesions were preoperatively treated with denosumab, with 3 lesions undergoing curettage and 12 resections. Nine lesions were postoperatively treated with denosumab, with six undergoing curettage and three resections. A total of 54 lesions were treated without denosumab, of which 43 underwent curettage and 11 resections (Supplemental Table 1).
The 3- and 5-year recurrence-free survival rate of all 78 GCTB lesions was observed to be 78.7% and 71.9%, respectively (Figure 2). Additionally, the 3- and 5-year recurrence-free survival rate of the 66 primary GCTB lesions was found to be 80.9% and 73.1%, respectively. Regarding associations between recurrences and clinicopathological factors in all 78 GCTB lesions, the timing of denosumab usage indicated statistical significance (p = 0.024), while other factors including age, gender, surgical site, tourniquet use, Campanacci grade, adjuvant use, history of pathological fractures, and reconstruction methods had no statistically significant association with recurrences (Supplemental Table 2). This analysis also identified resected cases to not have recurrences, which were, therefore, excluded from further analysis.

The 3- and 5-year recurrence-free survival rates for all 78 GCTB lesions.: The 3- and 5-year recurrence-free survival rates for all 78 GCTB lesions, including initial and recurrent cases, were 78.7% and 71.9%, respectively.
The 3- and 5-year recurrence-free survival rate of 52 curettage lesions, including primary and recurrent ones, were 69.9% and 60.1%, respectively. With respect to associations between recurrences and clinicopathological factors, 5-year recurrence-free survival rate was significantly lower in cases with pre-curettage denosumab (Pre-Deno) treatment than in those with post-curettage denosumab (Post-Deno) treatment or no denosumab treatment (0% vs. 66.7%; p = 0.07, 0% vs. 59.9%; p < 0.01; Figure 3(a)), and the recurrence rates also indicated statistically significant differences across the three groups (Pre-Deno: 100%, Post-Deno: 17%, and No-Deno: 28%, p = 0.01 and p = 0.02, respectively, Figure 3(b)). Interestingly, although several previous articles and our current curettage cases without pre-Deno had demonstrated the recurrence rates and time of GCTB after curettage to be approximately 16–50% and within 2 years after curettage. 4 –12 respectively, this study revealed all three GCTB cases with pre-curettage denosumab treatment (Pre-Deno) to develop recurrences, and cases with post-curettage denosumab treatment (Post-Deno) showing recurrences over 2 years. In clinicopathological status between the pre-curettage denosumab treatment group and the curettage alone one (Table 2), tourniquet use (p = 0.05), Campanacci grade (p = 0.005), and recurrence (p = 0.01) had significant differences between the two groups. Regarding statistical analyses of recurrences in the curettage group, although the timing of denosumab usage had a statistical significant difference (p = 0.02), there were no statistical significant differences of recurrences in the other clinicopathological factors (Table 3). Our results suggested the cases with pre-curettage denosumab to have severe risk for local recurrences, although our series included only three cases.

Postoperative results for each operation considering the presence or absence of denosumab. (a)The recurrence-free survival rate of the group using denosumab before curettage was compared to that of the group using denosumab after curettage and the group not using denosumab at all, and results of the log-rank test indicated p = 0.07 and p = 0.01. The 5-year relapse-free survival rates were 0%, 66.7%, and 61.3%, respectively. (b) There was no recurrence in the resected group, only in the curettage group. Recurrence rates also showed statistically significant differences between the three groups (denosumab before curettage: 100%, denosumab after curettage: 17%, and curettage only: 28%, p = 0.01, p = 0.02, respectively).
The characteristics of patients in the group who used denosumab before curettage and who did not use denosumab during the perioperative period.

The two groups of the curettage group according to the presence or absence of recurrence and items related to recurrence.

Our analysis revealed the ratio of MIB-1 > 10% to be 33.3% (4 of 12 lesions) in non-recurrent GCTB cases and 80.0% (8 of 10 lesions) in recurrent ones; there was significant difference between the two groups (p = 0.03). In GCTB cases with pre-curettage denosumab treatment that developed recurrences, the ratio of MIB-1 > 10% was 33.3% (1 of 3 lesions), similar to that of non-recurrent GCTB cohort (Figure 4); however, all three cases had local recurrences. Based on our analyses including clinicopathological, surgical, and immunostaining data, we supposed preoperative treatment using denosumab with curettage to possibly be a risk factor for recurrence in GCTB.

Comparison of the ratio of preoperative MIB-1 index > 10% in the curettage cohort. (a) Group A: recurrence cases that were treated with curettages with preoperative denosumab, group B: recurrence cases that were treated with curettages without preoperative denosumab (either no denosumab or postoperative denosumab), group C: no recurrence cases that were treated with curettages without preoperative denosumab (either no denosumab or postoperative denosumab). (b) The ratio of high (MIB-1> 10%) was 33.3% (4 of 12 lesions) in non-recurrence GCTB cases (group C) and 80.0% (8 of 10 lesions) in recurrence ones (group B). There was significant differences between the two groups (p = 0.03). In GCTB cases with pre-curettage denosumab treatment (group A) that developed all case recurrences, the ratio of high (MIB-1 > 10%) was 33.3% (1 of 3 lesions). Status of the ratio in group A was similar with non-recurrence GCTB cohort (group C).
Discussion
The local recurrence rate in patients with GCTB at our institution was similar to that reported previously. 4,10,11,26 These studies revealed curettage treatment with chemical adjuvants and/or bone cement as factors that suppress local recurrences statistically 4,5,7,9,12,27 ; however, these factors did not significantly influence the recurrence rates in our cohort. We suppose these differences could be due to the limited number of cases and facility where there was no strict strategy regarding the use of cement and auto-/artificial bone. Finally, in the curettage group, only the timing (Pre-Deno, Post-Deno, or No-Deno) of denosumab was identified as a risk factor for recurrence (p < 0.05) (Table 3).
Recently, some reports have described that the use of denosumab before curettage leads to local recurrence rate. 20,21 Although a report had warned against the use of denosumab before surgery to possibly increase the rate of recurrence after surgery; however, owing to the small number of cases (n = 21), it was not possible to statistically show denosumab use as a risk factor for recurrence. 21 Another study (n = 247) that had shown that the use of preoperative denosumab is the only independent factor for recurrence demonstrated using univariate and multivariate analyses to investigate relapse-free survival and joint preservation. 12 Our results complied with those of previous studies that revealed the use of preoperative denosumab treatment in curettage as a risk factor for local recurrences of GCTB.
In the recurrent patients treated with preoperative denosumab, the cortical bones close to the cartilage of joints that were destroyed by the activity of tumor cells were improved by denosumab use. However, at the same time, the suppressed tumor cells were surrounded by hard ossified and cortical bones. Therefore, we believe the local changes led to the difficulty in performing curettage easily, and hence the advantage of local treatment could not be achieved. Regarding immunostaining, previous articles had described high MIB-1 index to be associated with high frequency of local recurrences in conventional GCTB. 23,24 Additionally, several studies have revealed that cases with the MIB-1 index of > 10% to tend to develop recurrences. 23,24 We also performed immunostaining in our GCTB cohort and evaluated the association between recurrence and MIB-1 index. Results revealed significant differences in the MIB-1 index between the recurrences and the non-recurrences of GCTB. In the curettage group without preoperative denosumab treatment, the frequency rates of GCTB cases that had more than 10% MIB-1 index was 33% in the non-recurrence group and 80.0% in the recurrence group. Interestingly, our GCTB cases with preoperative denosumab treatment, all of which developed local recurrences, had one of three (33%) cases with > 10% MIB-1 index; this was consistent with the immunostaining results of the non-recurrence group.
Of the 12 cases that were treated resection with preoperative denosumab, 4 cases were in proximal fibula, 3 cases in distal femur, 2 cases in distal ulnar, 2 cases in distal radius, and 1 case in proximal tibia. With respect to tumors in fibular and ulnar, we decided that these parts could be resected without major functional losses. In the other lesion including distal radius, distal femur, and proximal tibia, we mainly decided these tumors were unable to treat with curettages because of severe bone destructions and soft tissue invasions.
A limitation of the present study is that our GCTB cohort included only three patients with GCTB who were treated with preoperative denosumab before curettage. Furthermore, this was not a clinical trial with blinded study, and there may be some clinical bias in the choosing the cases treated with preoperative denosumab before curettage (the three cases are shown in Supplemental Figure 1 and Supplemental Table 3A). Additionally, clinical protocols demonstrate that denosumab should be administered 120 mg/day on days 1, 8, 15, and 29, and monthly thereafter. 28,29 In this study, the three patients with GCTB were treated with slack schedules (treating with enough intervals of drug withdrawal) of denosumab to avoid adverse events; ossification was evaluated by image studies (Supplemental Table 3AB). Therefore, based on our present limited data, we found curettage with preoperative denosumab treatment is potentially associated with a risk of recurrence in GCTB. Studies with high cohort of patients, in future, might help elucidate the recurrence risk associated with preoperative treatment with denosumab before curettage in further detail.
Conclusion
In this study, we revealed the between recurrence risk and preoperative denosumab treatment before curettage in local GCTB control. Based on this investigation, it would be critical to consider the use of denosumab in surgical setting, especially when curettage is planned.
Supplemental material
Supplemental_Figure_4thR - Preoperative denosumab treatment with curettage may be a risk factor for recurrence of giant cell tumor of bone
Supplemental_Figure_4thR for Preoperative denosumab treatment with curettage may be a risk factor for recurrence of giant cell tumor of bone by Kei Sano, Yoshiyuki Suehara, Taketo Okubo, Keita Sasa, Taisei Kurihara, Keisuke Akaike, Daisuke Kubota, Tomoaki Torigoe, Nobuhiko Hasegawa, Midori Ishii, Yasuhiro Nakamura, Youngji Kim, Tatsuya Takagi, Kazuo Kaneko, Takuo Hayashi and Tsuyoshi Saito in Journal of Orthopaedic Surgery
Supplemental material
Supplemental_table_4thR - Preoperative denosumab treatment with curettage may be a risk factor for recurrence of giant cell tumor of bone
Supplemental_table_4thR for Preoperative denosumab treatment with curettage may be a risk factor for recurrence of giant cell tumor of bone by Kei Sano, Yoshiyuki Suehara, Taketo Okubo, Keita Sasa, Taisei Kurihara, Keisuke Akaike, Daisuke Kubota, Tomoaki Torigoe, Nobuhiko Hasegawa, Midori Ishii, Yasuhiro Nakamura, Youngji Kim, Tatsuya Takagi, Kazuo Kaneko, Takuo Hayashi and Tsuyoshi Saito in Journal of Orthopaedic Surgery
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grants-in-Aid from the Japan Society for the Promotion of Science [Grant Number # 19H03789, # 19K22694, and #15KK0353 to YS, 17K08730 to TS, #17K10987 to KK, #19K16753 to KA, #18K16634 to YK, and #18K15329 to TO].
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
