Abstract
Purpose:
There is no definitive consensus on the optimal treatment of Achilles tendon rupture. We comparatively analyzed the clinical outcomes of two types of repair surgeries in treating Achilles tendon rupture.
Methods:
This retrospective study included 12 patients of Achilles tendon rupture (group A) treated with ultrasound-guided percutaneous repair and 18 patients (group B) treated with open repair. Clinical evaluation was performed using the Arner–Lindholm scale, American Orthopedic Foot and Ankle Society (AOFAS) ankle–hindfoot score, Achilles Tendon Total Rupture score (ATRS), visual analog scale, time to single heel raise, bilateral calf circumferences, recovery of athletic ability, and other complications.
Results:
While the Arner–Lindholm scale, AOFAS ankle–hindfoot score, ATRS, time point when single heel raise was possible, differences in bilateral calf circumference, and recovery of athletic ability compared to pre-rupture level were not significantly different between the two groups (p = 0.999, 0.235, 0.357, 0.645, 0.497, and 0.881, respectively), overall and aesthetic satisfaction levels were higher in the group treated with percutaneous repair under ultrasonography guidance (p = 0.035 and 0.001, respectively). Overall, there were no cases involving sural nerve injury in either group.
Conclusion:
Innovative percutaneous repair provides not only similar clinical outcomes but also greater overall and aesthetic satisfaction levels of operative outcomes and minimal complications (i.e. sural nerve injury) compared to open repair surgeries. Therefore, percutaneous repair may be a useful technique in the treatment of Achilles tendon rupture.
Introduction
There is no established consensus on the optimal treatment protocol for acute Achilles tendon rupture. Open repair surgeries allow for direct confirmation of ruptured Achilles tendon and accurate suturing with tension, thereby reducing the frequency of recurrent ruptures. However, large incisions leading to potential infection, adhesion, and wound-related complications have been highlighted as the drawbacks of open repair surgery. 1,2 Meanwhile, numerous studies reported relatively favorable outcomes from conservative treatment. Although the proportion of favorable outcomes is increasing, 1,3 the possibilities of recurrent rupture and tendon elongation remain. 4 To overcome these limitations, less invasive techniques have been developed. Nonetheless, there have been reported issues of recurrent rupture and sural nerve injuries from these innovative techniques. 2,5 –8
We performed ultrasound-guided percutaneous repair of Achilles tendon rupture, in an attempt to further reduce the complications from traditional open techniques. Additionally, we compared the clinical outcomes of ultrasound-guided percutaneous repair and open repair surgeries to identify which technique had the preferable results.
Material and methods
Subjects
This study was approved by the institutional review board of our center before study initiation. The need for informed consent was waived because of the retrospective nature of the study. Overall, 30 cases of Achilles tendon rupture diagnosed via physical examination and magnetic resonance imaging (MRI) in our center between July 2015 and February 2017 were included. Among these cases, 12 patients (group A) were treated using ultrasound-guided percutaneous repair and 18 patients (group B) were treated using open repair. In our center, open repair surgery has been the first-choice treatment of Achilles tendon rupture until April 2016. Since then, Achilles tendon ruptures were treated with percutaneous repair, which is an eclectic therapy of open repair and conservative treatment. The sex ratio in group A was 11 men to 1 woman, and in group B was 16 men to 2 women. Mean ages at the time of injury were 39.4 (range, 28–55) years in group A and 40.2 (range, 28–52) years in group B. The majority of cases—11 in group A and 16 in group B—involved sports activities as the cause of injury, and the remaining 3 cases were injuries caused by loss of footing. Under MRI, mean distances of Achilles tendon rupture from the calcaneal attachment were 58.0 (range, 40.5–85.0) mm in group A and 57.6 (range, 42.5–86.0) mm in group B, while mean gaps of rupture were 21.8 (range, 7.5–40.0) mm in group A and 23.8 (range, 10.2–40.0) mm in group B. Operations were performed after means of 3.5 (range, 0.5–6.5) days after the injury in group A and 3.0 (range, 0.5–6.0) days after the injury in group B (Table 1). Only acute ruptures were included in this study, and the following cases were excluded: chronic ruptures older than 3 weeks, open ruptures, rupture at the calcaneal attachment, mean gaps of rupture greater than 40.0 mm, chronic Achilles tendinopathy, diagnosis with neuromuscular disease or peripheral vascular disease, treatment with systemic steroids or immunosuppressants, and a follow-up period less than 2 years. All patients in both groups were operated on by a single surgeon, and mean follow-up periods were 29.5 (range, 24–36) months in group A and 34.3 (range, 26–42) months in group B.
The characteristics of two groups.a

M: male, F: female; group A: percutaneous repair under ultrasonography guidance; group B: open repair.
a There were no significant differences between groups A and B.
Operation and anesthesia of the lower limbs were performed by a single specialist in orthopedics, and all inductions of anesthesia were performed using ultrasound-guided peripheral nerve block (femoral nerve, sciatic nerve, and posterior femoral cutaneous nerve block). The patient was in the prone position before the induction of anesthesia, and the ankle angle of the unaffected side was measured with the knee joint bent at 90° for reference during surgery. Additionally, the site of rupture and pathway of the sural nerve were marked preoperatively. After operation, all patients in both groups were managed by same postoperative program, as below.
Open repair
An incision of approximately 6 cm along the medial border of the Achilles tendon was made, followed by longitudinal incisions of the paratenon and mesotenon, to expose the ruptured Achilles tendon. After trimming the ruptured muscle fibers, the proximal end was sutured using the Krackow stitch, and the distal end was sutured using the Kessler stitch, with 4 strands of 1-0 Vicryl. With the proximal and distal ruptured parts completely in contact with each other, the knee joints of the affected and unaffected sides were bent at 90°. Suture threads at the distal end were pulled to match the angle of the dorsal surface of the unaffected foot, and the suture knot was made within the ruptured region to complete the suture. Then, we performed a circumferential stitch around the rupture site by using 3-0 Vicryl. Lastly, the paratenon and mesotenon were also sutured using 3-0 Vicryl suture threads.
Percutaneous repair under ultrasonography guidance
Surgery was performed with the patient in the prone position without a tourniquet, and an ultrasound machine (MyLab25Gold, Esaote, Italy) was utilized in the operating room with the probe wrapped in a sterilized vinyl. A 1-0 Vicryl (Ethicon Inc., Somerville, New Jersey, USA) was used for sutures, and the modified Kessler stitch was used to perform sutures (twice with two strands; Figure 1). Real-time ultrasonography guidance was utilized to ensure that the needles penetrated the parenchyma without damaging sural nerves (Figure 2(a) to 2(d)). The suture thread was passed through the calcaneal attachment, from the distolateral end to the proximal end of the calcaneal-attached tendon. After passing the suture thread in the longitudinal direction 2 or 3 more times at the proximal tendon, the suture thread was crossed to the medial border of the tendon. The thread was passed down again from the proximal tendon to the distal, calcaneal-attached tendon. After passing the suture thread in the transverse direction from the calcaneal attachment to take the thread out through the first needle hole, the final suture was performed with plantar flexion of approximately 10° compared to the neutral ankle angle of the unaffected side, which was measured preoperatively (Figure 3(a) to 3(i)). The identical procedure was performed once more using the same needle hole, so that the sutures included four strands in total. Since the thread was taken out through the needle hole and the needle was reinserted through the same hole, the procedure was done cautiously so that the needle did not penetrate the suture thread (Figure 4). Specifically, the remaining thread was pulled in the opposite direction of the needle movement, and the needle was inserted through the same needle hole, slightly touching the bottom surface of the skin, to complete the suture.

Schematic diagram of percutaneous repair under ultrasonography guidance. The modified Kessler stitch was performed, starting at the distal lateral part of the Achilles tendon and rotating in a clockwise manner until reaching the starting point. Suturing was performed twice, using identical methods (dotted line: first suture; full line: second suture).

Intraoperative images (a) and axial ultrasonograms (b–d). (a) Real-time ultrasonography was utilized to avoid sural nerve and lesser saphenous vein injuries and to confirm that adequately deep sutures were being made. Axial scans of (b) the distal portion, (c) mid portion, and (d) proximal portion of the Achilles tendon. The sural nerve and lesser saphenous vein moved away from the Achilles tendon as they moved from the proximal to the distal end (arrow: sural nerve; asterisk: lesser saphenous vein; triangle: Achilles tendon).

Operative procedures of percutaneous repair under ultrasonography guidance on the left Achilles tendon. (a–h) The modified Kessler stitch was performed twice, starting from the distal lateral part and ending at the starting point. The region of the Achilles tendon rupture and pathways of sural nerve were marked. (i) The ruptured Achilles tendon was overcorrected by 10° compared to the ankle on the unaffected side, considering potential early elongation.

While performing suturing, the needle may penetrate through the thread (Vicryl), so it must be done with caution. To prevent this, the thread that has already passed through the Achilles tendon should be pulled down while inserting the needle through the same hole.
Postoperative treatment
The passive range of motion of the ankle started 2 to 3 weeks postoperatively, but partial weight-bearing started after 4 weeks because of the use of absorbable suture material. For 4 weeks postoperatively, a single-limb non-weight-bearing splint was put on the affected limb in the plantar flexion state. Between 4 weeks and 12 weeks postoperatively, the patient was equipped with a cam walker brace and performed dorsal and plantar flexor muscle exercises in sequential manner. Partial weight-bearing and full weight-bearing exercises were performed at 4 weeks and 8 weeks after the surgery, respectively. After 6 weeks, rubber bands were used to perform resistance exercise of dorsal and plantar flexor muscles. By week 8, the patients could perform indoor daily activities without walking aids, and they were allowed to perform squatting and double heel raise motions. After 12 weeks, the patients were allowed to perform jogging and single heel raise motions. Continuity of the tendon was confirmed at 3, 6, and 12 months postoperatively using ultrasonography. Additionally, bilateral ankle angles were compared in the neutral position with the patient in the prone position and knee joint bent at 90° to check for potential recurrent rupture or elongation of Achilles tendon.
Evaluation methods and statistical analyses
Clinical evaluation of postoperative outcomes was performed using the evaluation standards of Arner–Lindholm (excellent, good, or poor), 9 American Orthopedic Foot and Ankle Society (AOFAS) ankle–hindfoot score (0–100), 10 and Achilles Tendon Total Rupture score (ATRS, 0–100). 11 At the final follow-up, overall satisfaction level of the postoperative outcome was measured using the visual analog scale (VAS; 0 = extremely unsatisfied, 10 = extremely satisfied), 3 as well as the VAS score (0–10) for aesthetic outcome of scars and the time point when single heel raise was possible. Difference in bilateral calf circumference and recovery of athletic ability compared to the pre-rupture level were also assessed simultaneously. Calf circumference was measured at the thickest part of the lower limb, with the knees bent at 90°. Athletic ability was categorized into one of the following four categories, in comparison with the pre-rupture level: same level, diminished level, stopped, or never participated. Lastly, complications including infection, recurrent rupture, and sural nerve injuries were evaluated.
Statistical comparison of the clinical outcomes between the groups was performed using IBM SPSS Statistics version 24.0 (IBM Corp., Armonk, New York, USA), and p values <0.05 were considered statistically significant. Because of the non-normal distribution of some data, continuous variables were assessed using Mann Whitney’s U-test, and categorical variables were tested using Fisher’s exact test. To predict the limitations of the small sample size in advance, a priori power analysis was performed. With the effect size of 0.8 and statistical power of 0.8, the required sample size was 54. However, when the statistical power was calculated using the actual measured data, it exceeded 0.9, suggesting that there was no issue with reliability of the outcomes.
Results
There were no differences in sex, age, cause of injury, location and distance of rupture, and time between injury and surgery between the groups (p >.05 for all). Length of incision was negligible in group A (the size of needle hole), while the mean length of incision in group B was 5.7 (5.2–6.3) cm (Figure 5(a) and 5(b)). Postoperative Arner–Lindholm scale outcomes were as follows. In group A, there were 9 “excellent” and 3 “good”; and in group B, there were 12 “excellent,” 5 “good,” and 1 “poor.” There was no significant difference between the groups (p = .999). Similarly, there were no significant differences between the groups in both AOFAS scores (group A: 93.7 (87–99), group B: 92.6 (70–98)) and ATRS scores (group A: 90.7 (80–94), group B: 89.3 (70–96)) (p = 0.235 and p = 0.357, respectively). With the exception of one case in group B where the patient required additional surgery because of recurrent rupture and deep infection, all other patients in both groups were able to perform single heel raises, and the timing was not significantly different between the groups (group A: 3.75 (3–6) months, group B: 3.65 (3–6) months) (p = 0.645). Meanwhile, the overall satisfaction level of operative outcome was significantly different between group A (9.1 (7–10)) and group B (8.2 (2–10)) (p = 0.035), as was the aesthetic satisfaction level of scars (9.9 (9–10) vs. 7.1 (3–9)) (p = 0.001). There was no significant difference in calf circumference of the two legs between group A (15.0 (8–24) mm) and group B (14.4 (6–35) mm) (p = 0.497). Recovery of athletic ability compared to pre-rupture level was as follows: in group A, there were 3 cases of “same level,” 4 cases of “diminished level,” 5 cases of “stopped,” and 0 cases of “never participated;” in group B, there were 5 cases of “same level,” 7 cases of “diminished level,” 5 cases of “stopped,” and 1 case of “never participated.” There was no significant difference between the groups (p = 0.881). In other words, while the Arner–Lindholm scale, AOFAS hindfoot score, ATRS, time point when single heel raise was possible, differences in bilateral calf circumference, and recovery of athletic ability compared to pre-rupture level were similar between the two groups, objective satisfaction levels were higher in the group treated with percutaneous repair under ultrasonography guidance (Table 2).
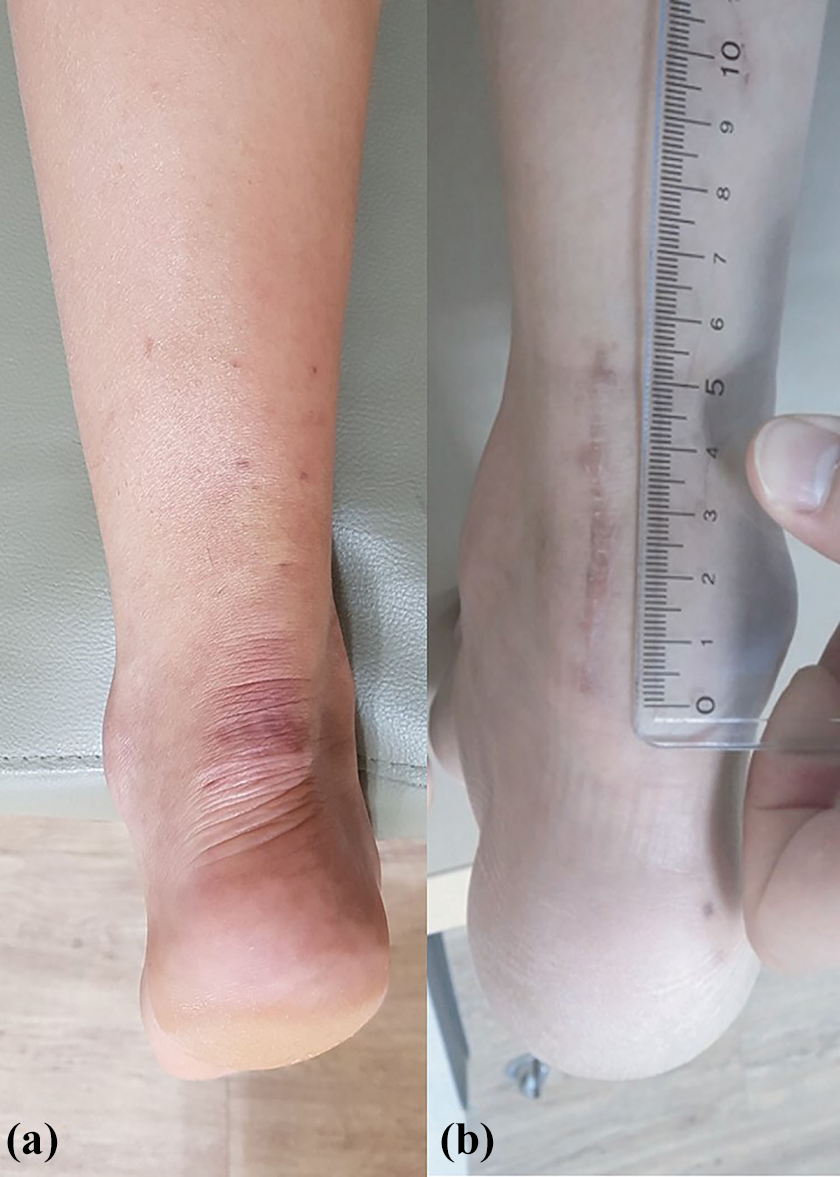
Images of the operative site scars in both groups, taken at the last follow-up. Scars from (a) percutaneous repair under ultrasonography guidance and (b) open repair group. The mean length of incision in the open repair group is 5.7 cm.
The clinical results of the two groups.a

AOFAS: American Orthopedic Foot and Ankle Society ankle–hindfootscore; ATRS: Achilles tendon Total Rupture Score.
a There were significant differences in overall patient’s satisfaction and aesthetic satisfaction of scar between groups A and B. We considered bold p-value < 0.05 was statistically significant.
In group B, there was a case of deep infection and recurrent rupture. For the patient with deep infection and recurrent rupture, debridement and washing were performed to control infection, followed by the rotation flap and sural flap turndown procedure. Overall, there were no cases of sural nerve injuries.
Discussion
There are three major findings in this study. First, unlike the traditional studies describing percutaneous repair under ultrasonography guidance, 12 –15 we have developed an innovative, unique method for performing percutaneous repair under ultrasonography guidance and compared its outcome with open repair. Second, in this study, two rounds of two-stranded modified Kessler stitches were sufficient to produce good clinical outcomes without recurrent rupture. This finding suggests that our method is an easy-to-perform, convenient method that does not require special equipment (i.e. the percutaneous Achilles repair system (PARS), Achilles Midsubstance Speedbridge, Achillon, etc.), self-developed equipment, 6 or difficult suturing techniques. In a previous study of cadavers, McCoy and Haddad 16 demonstrated no significant differences in suture strengths when performing a four-strand suture of the Achilles tendon using the Krackow, Bunnell, and Kessler stitch. Third, our method did not involve exposing the Achilles tendon, which does not interfere with the circulatory system.
Although the prevalence of Achilles tendon rupture is increasing with greater frequencies of sports activities, no consensus has been reached regarding an ideal treatment protocol. 17 Several studies have reported complications associated with infection, adhesion, and wound-related complications in traditional open repair surgeries, 1,2 and more studies on conservative treatment have highlighted the use of nonoperative treatment as a first choice treatment. 1,18 Nonetheless, operative treatment is more favorable in healthy, active patients.
Since Ma and Griffith 19 have reported percutaneous repair as an eclectic treatment of open repair surgery and conservative treatment, multiple studies have compared this innovative technique with open repair surgery, and there are ongoing studies on different minimally invasive surgery (MIS) techniques, which are improved versions of these operative techniques. Several previous studies have mentioned that percutaneous repair decreases the prevalence of skin complications and provides a similarly exceptional level of clinical and functional outcomes, 6 while Hsu et al. 20 demonstrated that there is no difference in the prevalence of complications between PARS and open repair surgery. Yang et al. 2 suggested that percutaneous repair is better than open repair in operative time, infection, and AOFAS scores.
However, the sural nerve has varying anatomical pathways, and thus, percutaneous repair will always expose the patient to the risk of sural nerve injury. 21 The possibility of sural nerve injury during percutaneous repair was approximately 15%. 8,22 Moreover, Soubeyrand et al. 14 demonstrated that inserting the suture needle with poor visualization during percutaneous repair has resulted in the needle not going into the parenchyma of the Achilles tendon in approximately 55% of cases, which may contribute to reduced suture strength. To improve these drawbacks of percutaneous repair, we have attempted an innovative technique of percutaneous repair under ultrasonography guidance, which allowed for avoidance of sural nerve injury in 100% of the cases through real-time visualization of the sural nerve position. Furthermore, we visually confirmed that the suture needle went through the parenchyma of the Achilles tendon, minimizing the chance of suture thread passing through the subcutaneous layer or surface layer of the tendon and consequently ensuring stronger sutures.
Additionally, use of absorbable sutures allowed for minimal stimulatory response against the sutures, resulting in more biocompatible sutures. Nonabsorbable sutures will retain tensile strength for an adequate period but can result in chronic stimulation of skin. 23,24 Although it is difficult to retain tensile strength using absorbable sutures, there are several advantages, including easy-to-maintain knots, less stimulation, and assistance with recovery of ruptured tendon. 24,25 Vicryl (polygalactin), which is a type of absorbable suture, exhibits reduction of tensile strength to 75% after 2 weeks, 50% after 3 weeks, and 25% after 4 weeks, until it becomes completely absorbed after 56–70 days. 23,24,26 However, the natural healing process of the tendon—characterized by an inflammatory phase (0–72 h), proliferation phase (2 days to 6 weeks), and remodeling phase (4 weeks to 12 months)—will proceed along with the sutures, and we believed that absorbable sutures will provide sufficient suture strength in early stages when supported with firm fixation in the early stages and stepwise rehabilitation exercises. In fact, Kocaoglu et al. 27 and Baiq et al. 28 utilized absorbable sutures for Achilles tendon rupture, reported that absorbable sutures produced less stimulatory responses and complications, and provided exceptional clinical outcomes compared to nonabsorbable sutures. Similarly, in previous study 24 and the current study, most patients without recurrent rupture could be treated simply through a postoperative rehabilitation program. One exception was the case that involved recurrent rupture with deep infection.
One limitation of this study is the small sample size. Because of this, there was nonnormal distribution of data in both groups. Second, since different repair techniques were used in the groups, the operative method may have affected differences in the prognoses. Third, no clear indications for the two different operative methods were set. Finally, the study design was retrospective nature. So, some bias may have occurred. Future studies should use a prospective study design that includes a larger number of patients and longer follow-up period. And, a previous study by Clanton et al. 29 has shown that, although the final strengths of MIS and open repair are similar, MIS exhibits weaknesses in early-stage elongation of the suture site. Considering this, we performed sutures with 10° plantar flexion relative to the angle of the ankle on the unaffected side. Therefore, additional studies on percutaneous repair and MIS during early-stage fixation should be performed, and additional education should be provided to the patients.
Conclusions
Percutaneous repair under ultrasonography guidance, in comparison with open repair surgery, provides not only similar clinical outcomes but also greater overall and aesthetic satisfaction levels. Additionally, this innovative technique minimized complications of traditional percutaneous repair. As a consensus has not yet been reached regarding the first-choice treatment for Achilles tendon rupture, this innovative approach of percutaneous repair under ultrasonography guidance may be considered a useful technique for treating Achilles tendon rupture.
Footnotes
Acknowledgement
The authors would like to thank In-Sun Kwon, PhD, for her help with professional statistical analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Research Fund of Chungnam National University in Daejeon, Korea.
