Abstract
Background:
Cobalt (Co) toxicity-related cardiomyopathy (CMP) in hip arthroplasty has recently been reported in the literature. The purpose of this review was to identify and assess available published evidence of CMP in hip arthroplasty patients and to derive recommendations for management.
Methods:
We evaluated 23 cases reported till October 2018 and stratified into three categories, based on pre-existing risk factors for CMP, histological confirmation and evidence of systemic signs of Co toxicity.
Results:
Co toxicity was considered to be the definite cause of CMP in 8 cases and probably contributory in 13 cases. Two cases were considered to have developed CMP secondary to pre-existing risk factors. Majority of the patients had good recovery of cardiac function after hip revision and cardiac management, but five cases deteriorated and died.
Conclusion:
Although Co-related CMP has been reported in a small number of cases of hip arthroplasty, a delay or missed diagnosis may lead to significant morbidity and mortality. Timely diagnosis, removal of causative implant and avoidance of metal articulations in revision for fractured ceramic implants may help in effective management.
Introduction
Cobalt (Co) toxicity has been reported in the literature as an occupational hazard in hard metal industry, diamond polishing and mineral assay industry. Hip arthroplasty-related Co toxicity with significant morbidity and mortality has been highlighted recently in the literature, which generated some media attention. Among other clinical manifestations of Co toxicity, cardiomyopathy (CMP) has been reported in some cases. Medical and Healthcare products Regulatory Agency (MHRA) has recently been recommended surveillance guidelines of patients with metal-on-metal hip arthroplasty to identify any local or systemic signs of metal toxicity. 1
Hip prosthesis bearing surfaces may be made of metals, such as Co, chromium (Cr), stainless steel or non-metal materials, such as ceramic or polyethylene. Metal ions, such as Co and Cr, are generated from corrosion of fixed and modular components of the prosthesis, abrasion between bearing surfaces of prosthesis and micromovements of failing components of the modular implant. The Co bivalent ions are predominantly responsible for systemic and local tissue reactions, whereas Cr trivalent ions are reduced rapidly in biological systems. 2
High systemic concentration of Co ions leads to a specific form of CMP, along with other systemic effects like neurological symptoms, hypothyroidism and polycythemia. 3 This particular type of CMP was first reported in individuals with a high intake of Co containing beer and its features were distinctive from previously known features of this condition. 4 Recently, this type of CMP has been reported in patients with hip arthroplasty.
The purpose of this review was to evaluate all published case reports of Co-related CMP in patients with hip arthroplasty. Our aim was to identify the scale and discuss salient features and management of this condition.
Methods
A search of PubMed and Embase databases was conducted to identify relevant studies using terms “CMP and Co or metal and hip or replacement or arthroplasty” till October 2018. Two reviewers (MU and MFK) independently screened articles, assessed for quality and extracted data from 21 articles, which described 23 cases of CMP in patients with metal hip arthroplasty implants (Table 3).
Data collection included patient demographics, potential risk factors for CMP (Table 1), type of hip implants, presentation of CMP, serum Co levels, systemic Co toxicity features (Table 1) and outcomes.
Risk factors for cardiomyopathy, features of cobalt-related cardiomyopathy and systemic features of cobalt toxicity.

The selected cases were categorised into three subgroups (Table 2), based on histopathology evidence, systemic Co toxicity features and presence/absence of risk factors of CMP.
Three categories of cases with reported cardiomyopathy based on cobalt toxicity as cause of presentation.

Co: cobalt; CMP: cardiomyopathy.
Summary of cases of cardiomyopathy in patients with hip arthroplasty.
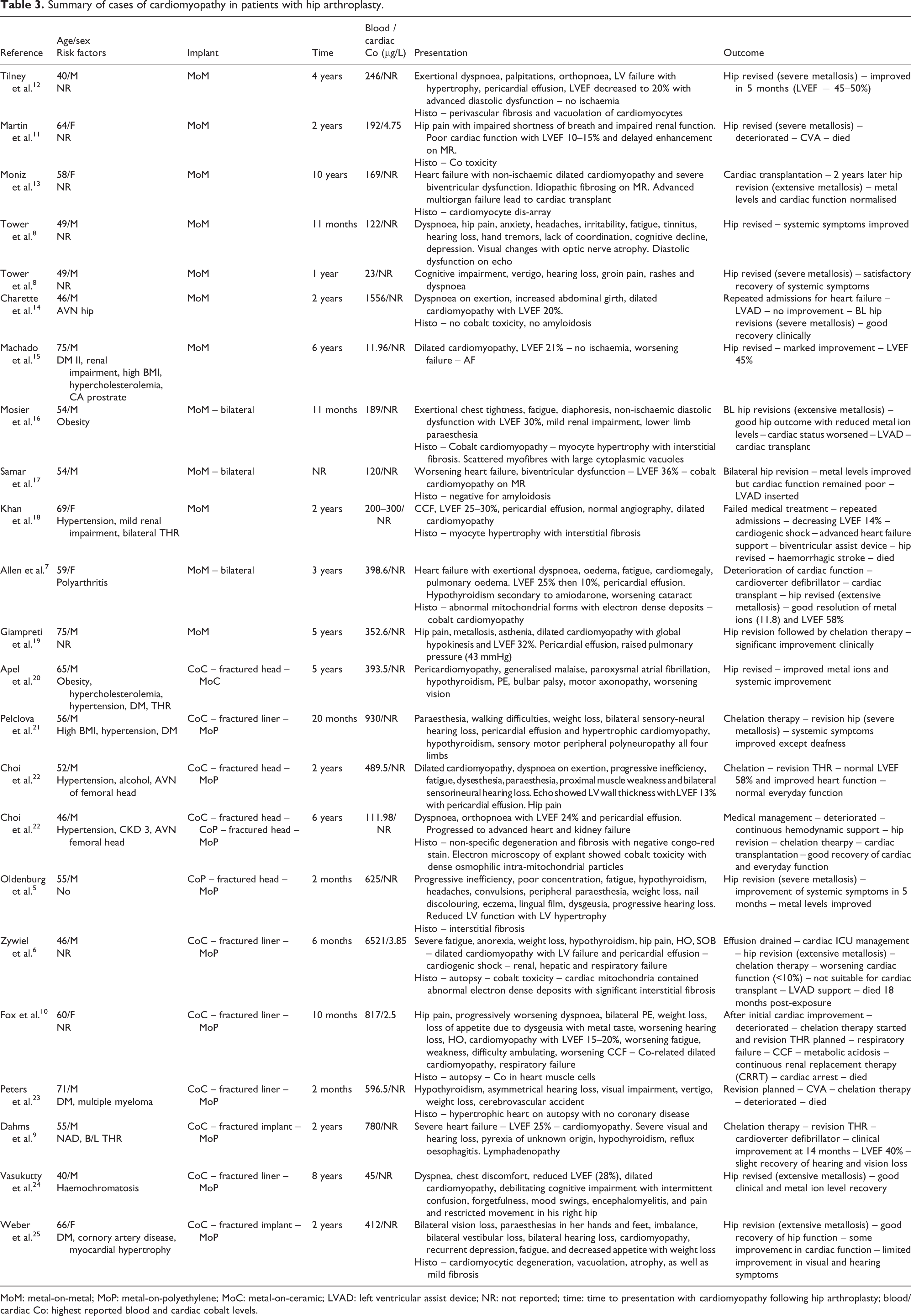
MoM: metal-on-metal; MoP: metal-on-polyethylene; MoC: metal-on-ceramic; LVAD: left ventricular assist device; NR: not reported; time: time to presentation with cardiomyopathy following hip arthroplasty; blood/cardiac Co: highest reported blood and cardiac cobalt levels.
Definite group (Co toxicity likely cause of CMP):
Probable group (Co toxicity may have contributed towards CMP):
Non-causal group (Co toxicity unlikely to be the cause of CMP): Have pre-existing risk factors for CMP (Table 1) before hip replacement. No evidence of systemic or cardiac Co toxicity.
Results
The cases were categorised into metal-on-metal (MoM, n = 12) and non-MoM group (n = 11). Non-MoM group included metal-on-ceramic (n = 1) and metal-on-polyethylene (n = 10). Interestingly, all cases in non-MoM group had implants as a revision procedure after fractured primary ceramic components.
The mean age was 58 years in the 12 cases of MOM compared to 56 years in 11 cases of non-MoM group (Table 4). The mean time to presentation with symptoms of CMP following hip procedure was comparable (MoM: 2.5 years vs. non-MoM: 2.6 years). In MoM group, the mean blood Co level was lower compared to non-MoM group (322 μg/L vs. 1041 μg/L; Table 5).
Mean age, gender distribution and time to presentation – categorized into MoM and non-MoM groups.

MoM: metal-on-metal; M: male; F: female.
Mean blood cobalt levels categorized by implant type and further stratified in three categories according to criteria in Table 2 (described in “Materials and methods” section).

MoM: metal-on-metal; MoP: metal-on-polyethylene; MoC: metal-on-ceramic.
Thirteen cases presented with systemic signs of Co toxicity of which four patients did not recover from hearing loss and visual loss. Most of non-MoM cases (8/11) showed systemic Co toxicity signs, whereas, in MoM group, only one-third (4/12) reported such features. Mean blood Co concentration was higher in patients with systemic signs (937 μg/L vs. 312 μg/L).
Twenty-one cases had their hips revised and a majority (n = 18) had reported extensive local tissue signs of metallosis, such as discolouration, degeneration, fluid collections and pseudotumour formation. Cardiac function recovered in 15 cases following hip revision. However, in nearly one-fourth cases (n = 6), cardiac function remained significantly impaired despite the improvement of Co levels. Three of these “poor responders” had further deterioration of cardiac function and developed fatal multisystem failures. Two patients had left ventricular assist device implanted and one patient had cardiac transplant.
Eight cases of Co toxicity-related CMP were confirmed in definite group 5 –11 (Table 3) based on confirmed histopathology features of Co toxicity of cardiac tissue and systemic signs of Co toxicity and no pre-existing risk factors of CMP. This group had the highest mean blood Co level of 1217 μg/L with a range of 122–6521 μg/L.
Thirteen cases were included in the probable group 11 –14,16 –20,21 –23 based on the presence of either cardiac histopathology or Co systemic toxicity features and evidence of pre-existing risk factors for CMP (Table 4). Mean blood Co level was 425 μg/L with a range of 112–1556 μg/L.
The two cases, 15,24 in non-causal group, had pre-existing risk factors of CMP and no evidence of cardiac or systemic Co toxicity features (Table 5). Blood Co levels in these cases were 45 and 14 μg/L.
Chelation therapy was initiated in seven cases upon a diagnosis of Co toxicity, which led to a reduction in serum Co concentrations but required additional measures for clinical improvement including hip revision and cardiac function support. Different types of therapies used include N-acetyl-cysteine, 19 2,3-dimercaptopropane-1-sulfonate 21 and ethylenediaminetetraacetic acid. 22
Five reported cases died of progressive deterioration of clinical presentation. In all of these cases, cardiac Co toxicity was confirmed either on biopsy or autopsy. Cerebrovascular accident was cause of death in three cases and other two died of multisystem failure. Three patients had revision of hip replacements but other two were not fit enough for any surgical procedure. Three of these patients received chelation therapy for Co toxicity.
Discussion
Several million MoM implants have been used worldwide since the emergence of second-generation arthroplasty implants. However, 23 cases of metal-induced CMP are reported in the literature, currently, suggest either low incidence or lack of awareness in medical community. Recent MHRA guidelines have strongly suggested regular long-term surveillance of patients with MoM implants.
Co-related metallosis
Majority of the reported cases (18) described extensive tissue metallosis at the time of revision hip surgery. In MoM group, four articles reported a potential link between MoM hip implant malpositioning and metallosis. Charette et al. 14 and Martin et al. 11 reported excessive anteversion of acetabular components as a potential cause of excessive metal wear.
In majority of patients with revision non-MoM implants for fractured primary ceramic implants resulted in severe abrasions and destruction of metal head components secondary to retained ceramic components, leading to third body wear. This may support the reason for significantly high serum Co concentrations in comparison to cases with primary MoM. In a recent review article, Rambani et al. 26 have recommended against the use of metal articulations in revision procedures for fractured ceramic components, and we endorse this recommendation.
Histopathology of Co-related CMP
Histopathology of myocardial tissue demonstrated myocardial hypertrophy and interstitial fibrosis in all reported cases, where either biopsy or autopsy was performed. These were generic features for any CMP. In our review, some studies also reported Co toxicity specific features, including increased vacuolation and lipofuscin, 11,12 myofiber disarray 13,16 and abnormal mitochondrial forms with electron-dense deposits. 6,7,10,22 Myocardial biopsy may help in diagnosis of Co CMP in suspected cases.

Blood cobalt levels of all studies. Key: Colour shows categories with reference to cobalt toxicity as the cause of CMP – red: definite; yellow: probable; green: non-causal. Shape shows type of implant – solid circle: MoM; empty circle: Non-MoM (MoP or MoC). CMP: cardiomyopathy; MoM: metal-on-metal; MoP: metal-on-polyethylene; MoC: metal-on-ceramic.
Role of chelation therapy
The chelating agent is a chemical, which binds metal (Co) and aids its renal excretion, thus reducing metal ion load in the body. In our review, different substances were used as chelating agents in seven cases. Although all such cases reported good outcome in terms of blood Co levels, all the authors suggested chelation therapy as an adjunct in management. Removal of the causative implant remains the recommended treatment, although chelation therapy can help to normalise Co levels while waiting for surgery or in patients who are not fit for any surgical intervention. If chelation therapy is initiated, patients kidney function should be monitored as it relies on renal excretion and Co toxicity that can lead to renal impairment.
Managing Co-related CMP
Lack of awareness in medical community has led to delay in timely diagnosis of metal-induced CMP. In one reported case, diagnosis of Co toxicity-related CMP was not made upto 4 years after initial presentation. However, in majority of reported cases, patients recovered from CMP after the removal of causative implant and with appropriate medical treatment.
Although a recent observational study by Lodge et al. 27 has not shown a significant increase in cardiac dysfunction in patients with MoM hip arthroplasty, Lassalle et al. 28 following a review of the French national health insurance database (255,350 patients) have recommended regular monitoring of cardiac function in patients with metal head hip arthroplasties, particularly with MoM articulation in women and older patients.
Recommendations
We recommend, based on the analysis of the above literature and MHRA guidelines, establishing a local framework for robust and cost-effective surveillance programme for the selected group of patients with hip implants, including MoM implants and all patients revision hip implants for fractured ceramic components.
If patient gives a history of new onset features of Co toxicity or cardiac symptoms and serum cobalt levels are above MHRA acceptable threshold for metal hips (7 ppb), 1 a cardiology review is advised to exclude cobalt-related CMP.
Diagnosis of CMP may be aided by investigations, including cardiac tissue biopsy, cardiac tissue cobalt levels and contrast-enhanced cardiac magnetic resonance scan.
Once a diagnosis of Co toxicity is established, the patient will benefit from the removal of causative implant and debridement of affected tissues to lower systemic Co concentration. Management of cardiac and systemic symptoms will be according to the clinical presentation. Chelation therapy can play a role in certain cases, where surgery is either delayed or not possible.
Conclusion
Co-related CMP has been reported in relatively very low number of cases of metal hip arthroplasty, however, a delay in diagnosis may lead to significant morbidity and mortality. All patients with MoM hip replacement and patients with a history of fractured ceramic components should be offered long-term surveillance for clinical, biochemical and/or echocardiographic features of Co toxicity. Timely diagnosis, removal of causative implant and avoidance of metal articulations in revision for fractured ceramic implants may help in effective management.
Limitations
This review only includes case reports. However, to our knowledge, this is the largest review study on Co-related CMP in hip arthroplasty patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
