Abstract
Purpose:
Postoperative urinary retention (POUR) is a common complication after total joint arthroplasties (TJAs). The POUR is managed with urinary catheterization, which is associated with a risk of urinary tract infection and subsequent periprosthetic joint infection. The purpose of this review was to afford a comprehensive understanding of POUR and its management.
Methods:
We identified 15 original articles concerning POUR after TJA, which were published from January 2010 to February 2019. The diagnostic method, incidence, risk factors, and management of POUR of the 15 studies were reviewed.
Results:
The incidence of POUR was ranged from 4.1% to 46.3%. Ultrasound was used for the detection of POUR among the total of the 15 studies. The following factors of old age, male gender, benign prostatic hypertrophy, history of urinary retention, spinal/epidural anesthesia, excessive fluid administration, patient-controlled analgesia, the use of opiates, underlying comorbidities, and poor American Society of Anesthesiologists (ASA) grade were risk factors for POUR. Most of the studies did not use indwelling catheterization during surgery. The POUR patients were managed with intermittent catheterization. The most common volume criterion for bladder catheterization was 400 mL. In inevitable use of an indwelling catheter, it should be removed within 48 h.
Conclusions:
This review provided an up-to-date guide for the detection and management of POUR.
Level of Evidence:
Level III.
Introduction
Urinary retention is defined as the inability to void urine despite the full bladder. 1 Postoperative urinary retention (POUR) is a common complication following hip and knee arthroplasty. 1 Delayed diagnosis of POUR leads to atonic bladder and permanent impairment of detrusor function. 2 Urinary retention is managed with urinary catheterization, which is associated with a risk of urinary tract infection (UTI). 3,4 Furthermore, UTI can lead to hematogenous bacteremia 5 and subsequent periprosthetic joint infection (PJI) in the early postoperative period after total joint arthroplasty (TJA). 6,7
To avoid possible risk of UTI and PJI, arthroplasty surgeons should have a comprehensive understanding of POUR and its management. To our knowledge, only one review on the effect of anesthetic and analgesic techniques on POUR was published in 2010 in an anesthesiology journal. 1 Thereafter, no comprehensive review about POUR was published in the literature. The POUR is practically important to orthopedic surgeons especially after the introduction of clinical pathways and fast-track protocol of TJAs. To date, the indication and protocol of bladder catheterization in the POUR patients remain controversial.
In this review, we updated the published evidence on the incidence, diagnostic method, risk factors, and management of POUR after TJAs.
Materials and methods
We performed the current systematic review according to the preferred reporting items for systematic reviews and meta-analyses guidelines. 8
Study eligibility criteria
Studies were selected on the basis of the following criteria: (1) study population: patients who underwent total hip arthroplasty or total knee arthroplasty; (2) studies defining POUR; (3) studies reporting the incidence and/or risk factors of POUR; and (4) studies written in English.
Studies were excluded if (1) they failed to meet the criteria above; (2) patients underwent total ankle arthroplasty; and (3) studies confined to comparing the effects of anesthesia and drugs.
Search methods for identification of studies
We carried out a comprehensive search on PubMed, Cochrane Collaboration Library, and EMBASE to identify relevant studies from January 2010 to February 2019. We used the following search terms: “POUR arthroplasty.” Two researchers reviewed the titles and abstracts of all potentially relevant studies independently, as recommended by the Cochrane Collaboration. 9 Any disagreement was resolved by the third reviewer. After full-text review, articles were assessed according to the predefined inclusion and exclusion criteria, and then eligible articles were selected. The reviewers were not blinded to authors or institutions.
Data extraction
The following data were extracted from the articles, authors, date of publication, design of the study (retrospective or prospective), participant features (number, mean age, and gender), factors related with surgery (use of preoperative urinary catheter, hip or knee arthroplasty, type of anesthesia, and pain management methods), and factors related with urinary retention (diagnosis method, incidence, risk factors, and complications after urinary retention).
Ethics statement
This study protocol was exempted for review by the Seoul National University Bundang Hospital Institutional Review Board (X-1907-550-907) in accordance with the exemption criteria. The present study was exempted from institutional review board review because it did not involve human subjects.
Results
Search results
The initial search resulted in 174 references from the selected databases. After the screening of titles and abstracts, 152 were excluded because they were duplicates, unrelated articles, case reports, or review articles. The remaining 22 studies were thoroughly assessed for full-text review. After the review, seven studies, which were not eligible for this systemic review, were excluded. Finally, 15 studies (10 prospective studies and 5 retrospective studies), involving 6397 patients, were included in this review (Figure 1). The main features and results of the 15 studies are presented in Table 1. 2,10 –23
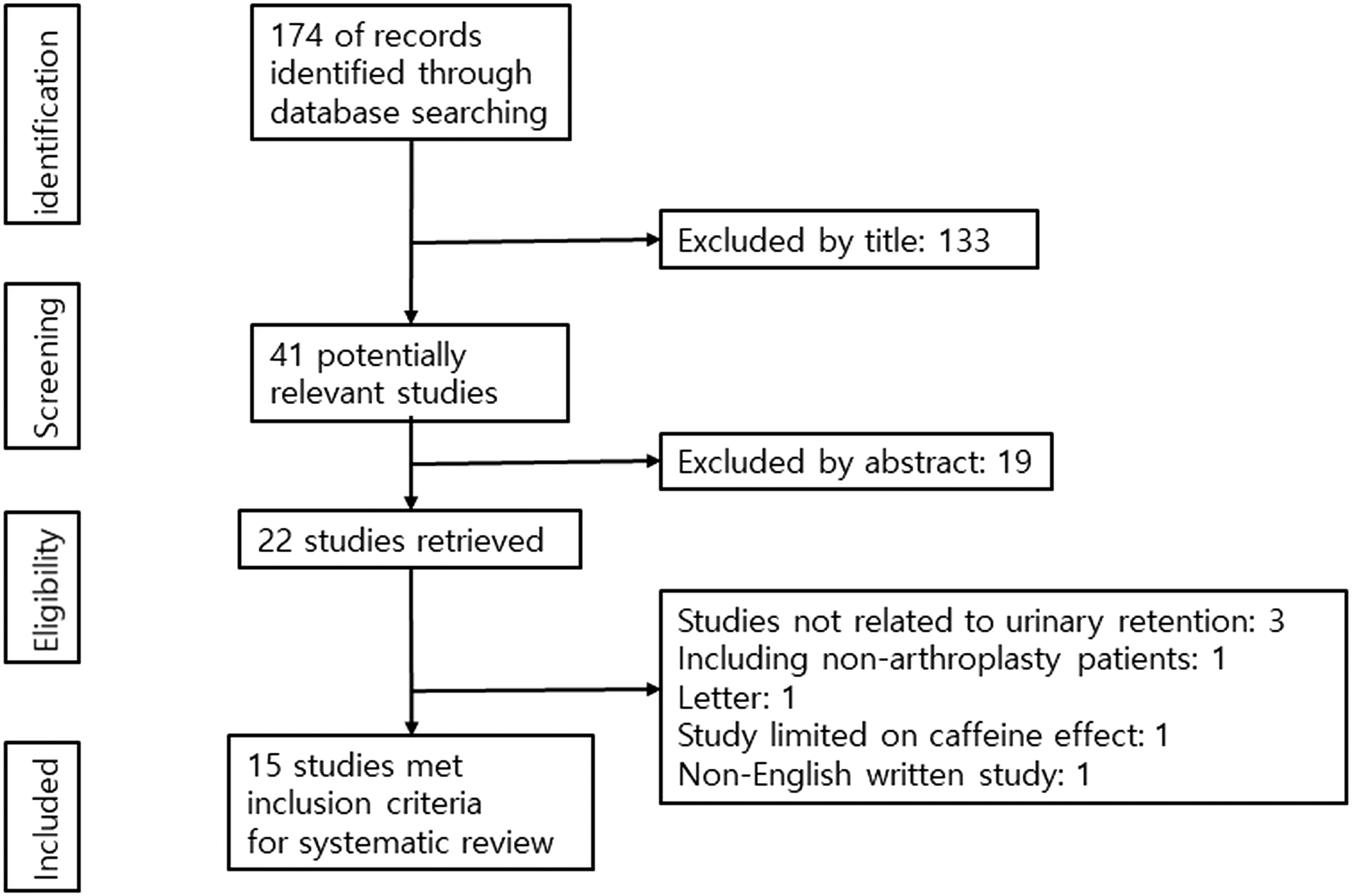
Systematic review flowchart following the PRISMA guideline. PRISMA: preferred reporting items for systematic reviews and meta-analyses.
Summary of 15 POUR studies.

M: male; F: female; G: general anesthesia; S: spinal anesthesia; E: epidural anesthesia; C: combined general and spinal anesthesia; PCA: patient controlled analgesia; US: ultrasound; N/D: no description; POUR: postoperative urinary retention; UIT: urinary track infection; NSAID: nonsteroidal anti-inflammatory drug; IV: intravenous; CCI: Charlson comorbidity score; min: minute.
Diagnostic criteria and incidence of POUR
In all of the 15 studies, the diagnosis of POUR was made by ultrasound. Each study adopted various volume criteria from 350 mL to 700 mL for the diagnosis of POUR. The most frequent volume criteria were 400 mL (five studies) followed by 500 mL (two studies) and 600 mL (two studies). The incidence of POUR varied widely from 4.1% to 46.3%.
Risk factors of POUR
In this review of 15 studies, male gender, benign prostatic hypertrophy, or history of urinary retention appeared as risk factors in eight studies. 10,12,14,16 –18,22,23 Five studies reported old age as a risk factor for POUR. 2,16,17,21,22 Age-related degenerative neuropathy seemed to be the reason for this causality. In three studies, the use of spinal/epidural anesthesia appeared as a risk factor compared with general anesthesia. 10,13,15 Administration of excessive fluid was a risk factor in three studies. 16,18,21 Other risk factors were patient-controlled analgesia, underlying comorbidities, 2 and poor ASA grade. 16
Indication of catheterization
Miller et al. reported that only 1% of POUR patients were treated with indwelling catheters and most of POURs were treated by one-time intermittent catheterization with no further urinary complications. 12 In the study by Markopoulos et al., all POUR patients recovered and none of them were discharged with an indwelling catheter. 22 In the study by Bjerregaard et al., 40% of patients had POUR and 8.2% required repeated intermittent catheterization, but only 0.9% necessitated an indwelling catheter. 13 However, in other studies, the rate of indwelling catheter application ranged from 16.8% to 36.2%. 2,15
There was a consensus that indwelling catheterization should not be used or at least should be limited in all of 15 studies. Each study adopted various threshold volumes ranging from 350 mL to 700 mL for the bladder catheterization. Five studies used 400 mL as the volume criteria.
Detection and management of POUR
Patients, who have a risk of developing POUR, should be screened before TJA and should be closely monitored during and after the operation. Ultrasound should be used for the diagnosis of POUR.
Once POUR has occurred, the patient should be treated with intermittent catheterization. Indwelling catheters and/or pharmacological treatments are used, only in cases of persistent POURs despite repeated intermittent catheterization. 2,12,15,16,18,20 If an indwelling catheter is inevitably used, it should be removed within 48 h. 24
Discussion
Patients undergoing TJAs have a greater incidence of POUR compared with other surgical procedures. In 2010, Balderi and Carli reviewed POUR after TJAs. In their review, the incidence of POUR ranged from 0% to 75%. At that time, clinical practice of bladder management in TJA patients included either preoperative insertion of an indwelling catheter or postoperative intermittent in-and-out catheterization. Both catheterization and ultrasound were used for the diagnosis of POUR at the time.
In our review, the POUR incidence ranged from 4.1% to 46.3%. Ultrasound replaced bladder catheterization for the diagnosis and management of POUR, and routine use of indwelling catheter was abandoned. Once POUR had occurred, the patients were treated with intermittent catheterization.
The reasons for the wide range in the POUR incidence were author-specific diagnostic criteria, different characteristics of TJA patients, the type of anesthesia, postoperative analgesia protocol, and the use of indwelling urinary catheterization during surgery.
Urinary catheterization is an invasive procedure, which is associated with complications including urethral trauma, infection, and patient’s discomfort. Ultrasound bladder scan has been introduced as a noninvasive diagnostic and monitoring tool of POUR. 25 In all of 15 studies published since 2010, ultrasound was used instead of catheterization.
Normal capacity of adult bladder ranges from 400 mL to 600 mL, 1 and bladder volume exceeding 600 mL has been considered to be pathological. 26 Overdistention of the bladder can cause urologic adverse events. 27 Because of this, an appropriate threshold volume for catheterization should be determined. Nevertheless, the threshold volume varied from 350 mL to 700 mL in the 15 studies, and there is no consensus about the threshold volume, yet. 13
According to the literature, myriad factors have been known to be associated with the development of POUR after TJAs. Well-known factors include old age, male gender, history of previous urological disease, the amount of intravenous fluid administration, type of anesthesia, anesthetic agents, and the use of opiates in the postoperative period. 1
Recently, ultrasound studies have reported that the occurrence of POUR can be predicted with the measurement of pre- and postoperative bladder volume. Scholten et al. reported that the preoperative residual volume was a risk factor for POUR. The POUR occurred in 15% when the residual urine was >150 mL. 20 Kort et al. showed that bladder volume >200 mL in the recovery room was a risk factor for POUR. 19 Keita et al. identified bladder volume >270 mL at the postanesthesia care unit as a predictive factor for POUR after orthopedic, abdominal, and urologic surgeries. 28
The International Prostate Symptom Score (IPSS) has been suggested as a predictor of POUR after TJAs. 29 Although IPSS was less costly than the ultrasound scan, it was not useful in predicting POUR in other studies. 30,31
Patients who have a risk of developing POUR should be screened before TJA and should be closely monitored during and after the operation. Recent studies reported that monitoring with the use of an ultrasound scan decreases the incidence of POUR and recommended the ultrasound scan at 6–8 h after the start of anesthesia. 15,19,25 In all studies, POUR patients were treated with intermittent catheterization. In case of inevitable use of an indwelling catheter, it should be removed within 48 h, because the duration of catheterization is closely related with the development of UTI. 24 Scholten et al. described the indwelling catheter as an inconvenience in the early mobilization of the patient and could cause delayed rehabilitation by hindering the patients’ mobility and increasing the length of hospitalization. 20 In addition, the use of α-blockers or 5a-reductase inhibitors may be used as a method of pharmacological treatment. 20
There are some limitations to this review. First, we could not assess differences according to patient’s constitution, ethnics, and surgical protocols. Second, we could not analyze the differences between anesthetic agents and postoperative pain control agents. Third, we could not differentiate urinary retention after revision arthroplasties from that after primary arthroplasties. POUR might differ according to the type of arthroplasty—primary arthroplasty versus revision. Among the 15 studies which were included in this review, 13 studies 10 –21,23 enrolled patients undergoing primary arthroplasty. The remaining two studies 2,22 seemed to include revision arthroplasties and primary arthroplasties. However, the authors did not document the number of revision patients. Nevertheless, this review provides an up-to-date guide for the detection and management of POUR.
Footnotes
Author contributions
Yong-Han Cha and Young-Kyun Lee contributed equally to this study and should be considered as co-first authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
