Abstract
Purpose:
A recent 3-month randomized, open-label controlled trial found that the intra-articular hyaluronic acid injection (GO-ON®) given as a single dose of 5 mL is as effective and safe as three repeated doses of 2.5 mL in patients with knee osteoarthritis. However, the information on the long-term efficacy and economic implications of the single-dose regimen is still limited. Hence, this follow-up study was designed to compare the effectiveness and costs of the two regimens 12 months following the treatment.
Methods:
All the 127 patients, who received either three repeated doses (n = 64) or a single dose (n = 63) of GO-ON in the previous trial, were followed up in month 12 following the treatment. The effectiveness of both the regimens was assessed using the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), and the mean WOMAC scores were compared with those recorded at the baseline and in month 3. Additionally, the total treatment costs of the two regimens, taking account of both direct and indirect costs, were computed and compared.
Results:
A total of 125 patients (98.4%) completed the assessment. Despite the reduction of the overall mean WOMAC score from 39.24 to 19.93 (p < 0.001) in the first 3 months following the treatment with GO-ON, no further changes were observed up to month 12 (p > 0.95). In the meantime, the two regimens did not differ in the mean WOMAC scores (p = 0.749) and in the subscale scores for pain (p = 0.970), stiffness (p = 0.526), and physical functioning (p = 0.667) in month 12. The cost for single-dose injection was found to be approximately 30% lower compared to the repeated doses.
Conclusion:
These findings indicate that the single larger dose of GO-ON is as effective as the repeated doses over 12 months, and yet the total treatment cost is lowered.
Introduction
Osteoarthritis (OA) is the most frequently diagnosed condition in patients with chronic pain. 1 Currently, symptomatic OA is seen in approximately 10% and 18% of elderly men and women, 80% and 25% of whom, respectively, have movement restriction and difficulties in performing normal daily activities. 2 Knee OA, one of the common types of OA, is a chronic joint disease, which regularly causes severe disability at its advanced stages, resulting in poor quality of life. 3 –5 To date, approximately 250 million people are affected by knee OA globally. 6
Typically, in OA patients, the molecular weight (MW) and concentration of hyaluronic acid (HA), which is a glycosaminoglycan constituent of synovial fluid and cartilage matrix, is decreased. Therefore, one of the symptomatic treatment options for knee OA is exogenous HA, which is normally injected into the diarthrodial joints to reduce pain and delay the progression of the disease. 7 –9 Exogenous HA in an injectable form was first approved by the US Food and Drug Administration in 1997. It has since then been widely used, particularly in knee OA patients who do not respond well to conservative nonpharmacological and pharmacological treatment. It functions mainly by reducing the pain transmission and blunting the inflammatory cascade as well as by stimulating the synthesis and deposition of extracellular matrix molecules, which are suppressed in osteoarthritic joints. 10
In fact, viscosupplementation with an intra-articular HA injection in knee OA patients has been shown to be as effective as nonsteroidal anti-inflammatory drugs and other analgesics 11 and has therefore been highly recommended by therapeutic guidelines. 9,12 In addition, this approach is less invasive and less expensive in comparison with the total knee replacement (TKR) surgery. Studies demonstrate that the intra-articular HA injection is likely to delay the need for TKR surgery for 1–3 years. 13,14 The improvement of pain control and functional outcomes was also noted within 6 months following the injection. 15,16
Intra-articular HA injection is currently marketed as either low (0.5 × 106 to 1 × 106 Daltons (Da)), intermediate (2 × 106 Da), or high MW (6 × 106 Da) formulations. 17 The low and intermediate MW formulations are commonly recommended to be injected weekly for three to five doses, whereas the high MW formulation is to be administered as a single larger dose. Although it was suggested that the intermediate MW HA given as a single-dose regimen could yield a similar efficacy to a multiple-dose regimen, 18 the evidence remains limited to a specific product and a relatively short follow-up period. While the impaired mobility of patients and the increasing treatment cost of knee OA are of equal concern, further evidence to support the effectiveness of a single-dose regimen of a more cost-effective formulation is warranted.
GO-ON® (Meda Groups, Sollna, Sweden) is a preparation of the intermediate MW HA (0.8 × 106 to 1.5 × 106 Da) obtained from Streptococcus equi through a fermentation process. It has been marketed at a concentration of 10 mg/mL in a 2.5-mL prefilled syringe and has been approved to be given weekly for three to five doses for OA. Although a recent 3-month randomized, open-label controlled trial shows that GO-ON given as a single dose of 5 mL is as effective and safe as three repeated doses of 2.5 mL in knee OA patients, 19 the information on the long-term efficacy and economic implications of the single-dose regimen is still limited. This study was therefore designed to compare the effectiveness and cost of the two dosing regimens of GO-ON, 12 months following the treatment.
Methods
One-year efficacy assessment
In the previous trial, patients in the age range of 40–80 years, diagnosed with either unilateral or bilateral knee OA, were recruited. 19 They were randomized to receive GO-ON given either as three repeated doses of 2.5 mL (n = 64) or a single dose of 5 mL (n = 63). The efficacy of both the regimens was assessed using the Western Ontario and McMaster Universities Arthritis Index (WOMAC), which consisted of 24 items divided into three subscales, including pain (5 items), stiffness (2 items), and physical functioning (17 items). 20,21 Each item was scored using a four-point scale based on the patients’ responses (0 = none, 1 = mild, 2 = moderate, 3 = severe, and 4 = extreme), yielding a possible total score ranging from 0 to 96. The mean WOMAC scores at the baseline and in month 3 were subsequently compared.
In this study, the efficacy of both the regimens was reassessed using the same instrument in month 12 following the treatment. The overall mean total WOMAC scores, as well as the mean total WOMAC scores of each group, were then compared with those recorded at the baseline and in month 3. The differences in age, gender, and grade between the two groups were tested using the Pearson’s χ 2 and independent t-tests. Besides, the overall and intragroup (within-group) changes of the total and subscale scores over 12 months, along with the intergroup (between-group) differences in the score changes, were detected using the repeated measure analysis of covariance. The results presented were controlled for age, gender, and Kellgren–Lawrence (KL) grade. All the statistical analyses were performed using the IBM SPSS for Windows version 21.0 (IBM Corp., New York, USA) and were considered significant only if p < 0.05.
Cost comparison
The 1-year direct and indirect costs incurred by both the government and patients for both the regimens were calculated. The components contributing to the direct medical cost in the calculation were medications (acquisition cost in 2017), consumables used in the administration of the medications (cost as in 2017), professional services (monthly incomes of doctors and nurses divided by the time spent on each follow-up visit; assumed to be 10 min per visit), imaging services (nonsubsidized charge per procedure), and hospital facilities (nonsubsidized charge per visit). The transportation cost of each patient was estimated based on the average fare of public transportation in Kedah State (MYR 0.60 per km), while the indirect cost was estimated based on the income loss during each visit (national minimum wage of MYR 900 per month). Societal perspective sums up all the cost components.
Ethics approval
The current study forms part of a randomized controlled trial to compare two dosing regimens of GO-ON. It was registered with the National Medical Research Registration, Malaysia, under the protocol number NMRR-13-245-14932, and was approved by the Medical Research Ethics Committee (MREC), Malaysia.Results
Of 127 participants of the previous trial, only two (1.6%) were lost to follow-up (Figure 1). A total of 62 and 63 of the patients in the single-dose and weekly dose groups, respectively, completed the assessment. The two groups did not differ significantly in the distribution of age and KL grade; however, the single-dose group had a higher proportion of female patients (p = 0.008; Table 1).

Trial profile.
Age, gender, and KL grade of patients (n = 125).

KL: Kellgren–Lawrence; SD: standard deviation.
a Pearson’s χ 2 test.
b Independent t-test.
One-year efficacy assessment
Despite the dosing regimens, a significant reduction was found in the WOMAC total score, as well as in the WOMAC pain and physical function subscores, throughout the 1-year period (Table 2). Overall, the mean total WOMAC score was found to reduce by approximately 50% from 39.24 to 19.93 after 3 months (p < 0.001); however, no further changes in the score were found between month 3 and month 12 (p > 0.95; Table 3). A similar trend was also detected for the pain and physical function subscores. However, the subscore for stiffness showed significant improvement in month 3 but the improvement did not sustain up to month 12. On the other hand, the two regimens did not differ in the mean WOMAC scores (p = 0.749) and in the subscale scores for pain (p = 0.970), stiffness (p = 0.526), and physical functioning (p = 0.667) in month 12 (Tables 2 and 3).
Changes of WOMAC scores over 12 months following the treatment.
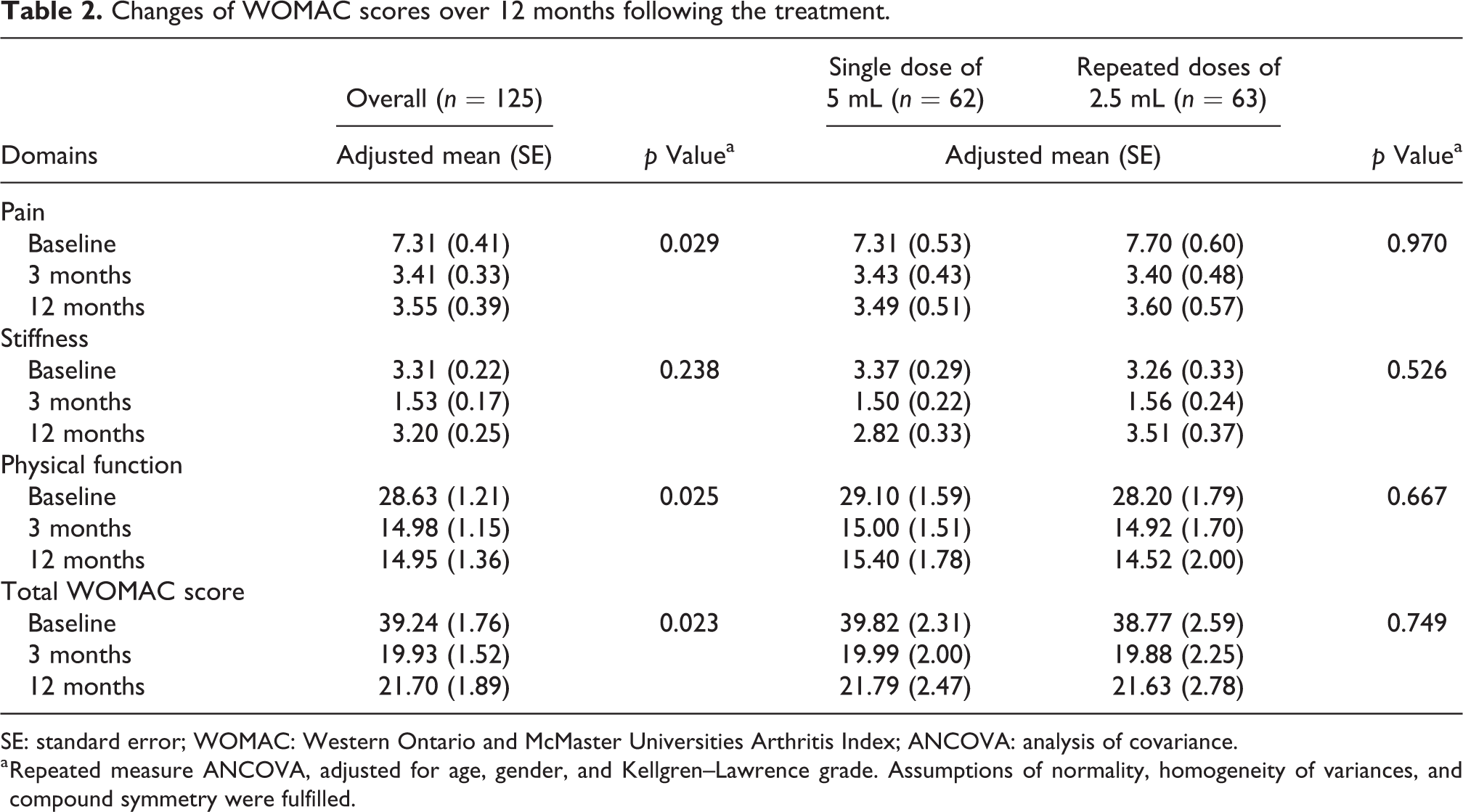
SE: standard error; WOMAC: Western Ontario and McMaster Universities Arthritis Index; ANCOVA: analysis of covariance.
a Repeated measure ANCOVA, adjusted for age, gender, and Kellgren–Lawrence grade. Assumptions of normality, homogeneity of variances, and compound symmetry were fulfilled.
Comparison of WOMAC scores following the treatment based on time.
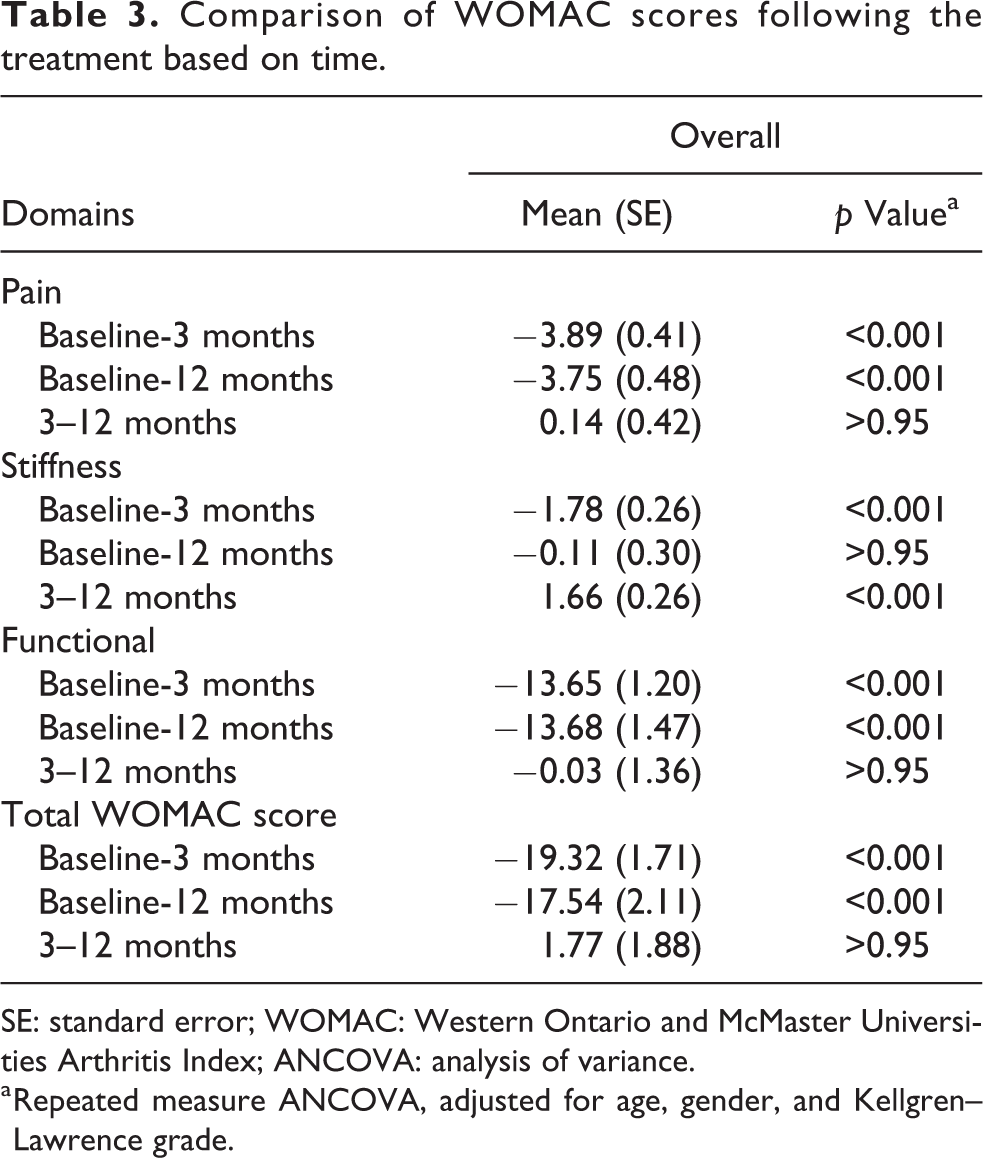
SE: standard error; WOMAC: Western Ontario and McMaster Universities Arthritis Index; ANCOVA: analysis of variance.
a Repeated measure ANCOVA, adjusted for age, gender, and Kellgren–Lawrence grade.
Cost comparison
The average cost per patient for the single-dose and weekly dose regimens was tabulated in Table 4. From the patient perspective, the total cost involved the cost of medication, the direct nonmedical cost, and the indirect cost. From the provider perspective, the total cost involved the direct medical cost, namely, the consumables, professional services, imaging services, and hospital facilities. The societal cost for single injection is MYR 1310.01, while the cost to the provider made up 62.9% (MYR 823.57) of it. The societal cost for triple injection is MYR 1852.17, while the cost to provider made up 60.6% (MYR 1122.51) of it. Single injection was found to save MYR 298.94 to provider, MYR 243.22 to patient, and MYR 542.16 to society.
Cost comparison of the two dosing regimens.

a Including syringe, needle, normal saline, and lignocaine injection.
bTen-minute services per patient during each visit by doctors and nurses.
c Standard nonsubsidized price for each specialist clinic visit.
Discussion
The current study, to our best knowledge, is the first that demonstrates the long-term efficacy of GO-ON, particularly in the improvement and sustainability of pain control and physical function in OA patients. To date, the evidence for intra-articular HA injection is limited to 3–6 months in general. 22,23 Furthermore, it was the first attempt to directly compare the conventional multiple-dose regimen with a single larger dose regimen of GO-ON. The findings could be used to guide the physicians in improving the current treatment strategy for OA, especially by minimizing the overall treatment cost and enhancing the quality of life of patients.
This study added to the previous study, which confirmed the efficacy of GO-ON intra-articular HA injection in alleviation of pain and improvement of physical function of OA patients after 3 months of administration despite the dosage used. 19 Recent systematic review suggested that the effects of intra-articular HA injection in reducing pain for knee OA were likely to take place in 4–8 weeks and could last up to 24 weeks. 16 Our findings suggest that the efficacy of GO-ON is sustainable for up to at least 12 months. However, consistent with the findings of several studies, such effects were found to be more sustainable on pain and physical function as compared with stiffness. 24,25
Besides, it is found that the two dosing regimens tested did not differ in the overall mean WOMAC scores, as well as in the subscale scores, over the 12-month period. Similar to a previous study, 26 this study also demonstrated that GO-ON intra-articular HA injection was safe regardless of the dosing regimens used. Within this context, the single larger dose is preferable to be used routinely in the future, as it is generally more convenient and acceptable for the patients. 27,28 Besides, it potentially reduces risks of complications at the injection site, the psychological fear of the procedure in the patients, and patient load of hospitals.
Apart from demonstrating the similarity of the two dosing regimens of GO-ON in efficacy, this study shows that the single-dose regimen is less expensive than the weekly dose regimen. Overall, the total cost of the single-dose regimen was shown to be approximately 30% less than the weekly dose regimen. As the feasibility of TKR surgery has always been limited by its high cost and patients’ anxiety, 29,30 intra-articular HA injection, especially in a single dose, could serve as an ideal treatment option. Such a modification in treatment regimen is expected to significantly reduce the financial burden among patients with knee OA. There are some limitations in the current study. Consistent with similar studies on HA, 31 only patients with grade 1–3 OA by the KL grading method were included. 32 Therefore, the results may not be generalizable to all OA patients, particularly those with a higher grade of severity. Secondly, due to the differences in the number of injections and patient visits, “blinding” was not possible in the study. As the effect of a larger volume used remains unclear, further investigation is also required to study the volume effect of single larger dose versus multiple smaller doses of GO-ON intra-articular HA injection in achieving maximum therapeutic effects in knee OA patients.
Conclusion
This follow-up study indicates that both the single-dose and weekly dose regimens of GO-ON intra-articular HA injections are equally effective over 12 months following the treatment, while the total cost of the single-dose regimen is approximately 30% lower. Such findings could be helpful in improving the current treatment strategy for OA, especially when TKR is not feasible or needs to be delayed. Additional studies to compare a single larger dose of GO-ON and other single-dose formulations are warranted.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
