Abstract
Background:
The purpose of this study was to evaluate clinical and radiological outcomes and to analyze the histological findings of repaired cartilage in patients more than 50 years old with underwent fibrin–matrix autologous chondrocyte implantation (ACI).
Methods:
From January 2013 to February 2014, a prospective study was conducted on 16 patients (16 knees) who underwent fibrin–matrix ACI for International Cartilage Repair Society grade 3 or 4 chondral lesions of the knee. The major lesion was in the medial femoral condyle in all patients. The mean age of the patients was 58.1 ± 6.2 (range 51–66) years, and the minimum follow-up period was 2 years. All patients had clinical and radiological evaluations at 3 months, 6 months, 1 year, and 2 years after surgery. Twelve patients had second-look arthroscopies at 1 year after surgery, and implanted chondral biopsies were performed in 11 of these 12 patients for histological analysis.
Results:
Functional disability assessment scales for the knee significantly improved after fibrin–matrix ACI (p < 0.05). The visual analog scale score significantly decreased from 6.7 ± 1.2 to 2.0 ± 1.8 (p < 0.001). The mean modified magnetic resonance observation of cartilage repair tissue score was 83.8 ± 17.3 at 1 year after surgery and 74.0 ± 19.2 at 2 years after surgery. Repair of the tissue with hyaline cartilage was confirmed histologically.
Conclusions:
Satisfactory clinical and radiological outcomes were obtained from gel-type fibrin–matrix ACI technique in older patients with a cartilage defect of the knee. Histological analysis confirmed that the new repaired tissue with hyaline cartilage filled the cartilage defect area. Therefore, fibrin–matrix ACI is believed to be an applicable treatment for older patients with chondral lesions of the knee.
Level of evidence:
Level IV.
Background
In patients with knee pain requiring arthroscopic intervention, the prevalence of damaged articular cartilage has been reported to be about 60%. 1 Injury to the articular cartilage is one of the most commonly encountered disorders in orthopedics. 2 Because articular cartilage has no blood supply and the mobility of chondrocytes is limited, articular cartilage has only a limited capacity to regenerate. 3 An untreated cartilage lesion may contribute to the development of osteoarthritis (OA). 4,5 The goals of surgical management of symptomatic cartilage damage are pain relief, improvement in joint function, and resumption of activities of daily living. 6 Numerous research and treatment methods have been developed in the management of damaged articular cartilage. 7 Unlike other treatments based on stimulating the bone marrow, autologous chondrocyte implantation (ACI) can repair damaged cartilage into hyaline cartilage. 8
Third-generation ACI has shown good results as a treatment modality for chondral defects in young patients. 9,10 Subsequently, a consensus has been formed on ACI as a treatment for young patients with cartilage defects. As age increases, metabolic activity of knee cartilage decreases, and the size of chondral defect lesions tends to be large and becomes chronic. Therefore, joint replacement tends to be preferred to cartilage regeneration procedures. As the average life expectancy has increased and older individuals want to maintain high activity levels and joint preserving procedure rather than joint sacrificing surgery such as total knee arthroplasty, the interest in ACI treatments for cartilage defects has increased.
However, there have been few reports on outcomes of ACI in older patients. Also, effectiveness of ACI in older patients has not been established. The aim of this study was to evaluate the clinical and radiological outcomes prospectively and to analyze the histological findings in older patients who underwent fibrin–matrix ACI for chondral lesions of the knee. It was hypothesized that fibrin–matrix ACI would be an effective treatment for damaged articular cartilage in older patients and that it would induce cartilage regeneration in chondral defects.
Methods
Participants were enrolled form November 2012, and a prospective study was conducted in 16 knees in 16 patients who underwent fibrin–matrix ACI for an International Cartilage Repair Society (ICRS) grade 3 or 4 cartilage lesion of the knee and over 50 years between January 2013 and February 2014. All patients were followed up for 2 years after surgery, and the last follow-up was in January 2016. The protocol for this study was approved by the Inha University Hospital Institutional Review Board (approval number: IUH-IRB 12-2521).
The indications for ACI were the presence of a single cartilage lesion with size not exceeding 12 cm2 or multiple lesions not exceeding 20 cm2 (Outerbridge grade 3–4). 11,12 The knee joint was stable in all cases; however, a high tibial osteotomy (HTO) was performed to correct malalignment when there was a varus or valgus deformity on the mechanical axis greater than 5°. 13
Exclusion criteria included OA that had progressed to the Kellgren–Lawrence grade >2, the presence of inflammatory arthritis, or a severe varus or valgus deformity greater than 15°. Patients under 50 years old and with patellofemoral instability, drug abuse, or a psychiatric history were also excluded (Table 1).
Inclusion and exclusion criteria.

ICRS: International Cartilage Repair Society; OA: osteoarthritis.
The demography date of our study is listed in Table 2. The surgical site in all of the patients was the femur, and the mean lesion size was 3.2 ± 2.3 (median (range) 2.6 (1.1–10.4)) cm2. One case was ICRS grade 3, and 15 cases were ICRS grade 4. The major lesion was in the medial femoral condyle in all patients. There was at least one additional lesion in all patients. There were six patients with two chondral defects, five patients with three chondral defects, three patients with four chondral defects, and two patients with five chondral defects. A corrective HTO was performed in nine patients with malalignment. The follow-up period was 2 years in all of the patients, except for two patients who were lost during the follow-up.
Patient demographics.

BMI: body mass index; MFC: medial femoral condyle; LCF: lateral femoral condyle.
Patient evaluation
Clinical evaluation
To assess pain and functional disability of the knee, the Knee Injury and Osteoarthritis Outcome Score (KOOS), the International Knee Documentation Committee (IKDC) subjective score, and the visual analog scale (VAS) score were measured in all patients at 3 months, 6 months, 1 year, and 2 years after surgery. Magnetic resonance imaging (MRI) was performed at 1 and 2 years after surgery, and the obtained images were analyzed using the modified magnetic resonance observation of cartilage repair tissue (MOCART) score. Second-look arthroscopy and histological examination were conducted 1 year after surgery.
Second-look arthroscopy and biopsy
It was designed that second-look arthroscopy and histological examination would be performed after ACI to evaluate the repaired cartilage status. Prior to the study, subjects were given informed consent including second look arthroscopy.
To assess graft condition, arthroscopy was performed by the same senior surgeon at a mean of 12 months postoperatively in 12 of the 16 patients who agreed to the procedure. The grafts were inspected and evaluated using the ICRS scoring system. The parameters that were assessed using this system included the degree of defect fill, the degree of graft integration to the adjacent normal articular surface, and the gross appearance of the graft surface. Biopsy was done in 11 of the 12 patients who agreed to have it performed. 14,15
Histological evaluation
The samples were immediately fixed in 4% paraformaldehyde (pH 7.4) for at least 2 days. Decalcification was performed with formic acid and nitric acid for 4 days. All of the samples were embedded in paraffin, and sections (5 μm) were cut using a standard microtome. The paraffin sections were stained with hematoxylin–eosin to evaluate general morphology, with safranin O to evaluate glycosaminoglycans (GAGs), and with Masson’s trichrome to evaluate general collagen distribution. The sections were also immunostained for type I and II collagen. Two specimens were examined from each procedure, and the representative histological sections were evaluated. All of the examinations were performed using a light microscope. For histological assessment, thickness of cartilage, the Oswestry (Os) score, and immunohistochemistry scoring for type I and type II collagen were evaluated (Figure 1). 16,17 The immunohistochemistry score was carried out to quantify the level of expression and to show the tendency for each type of collagen. Histological assessment was carried out by four independent observers, and the mean of four values was used as the final value.
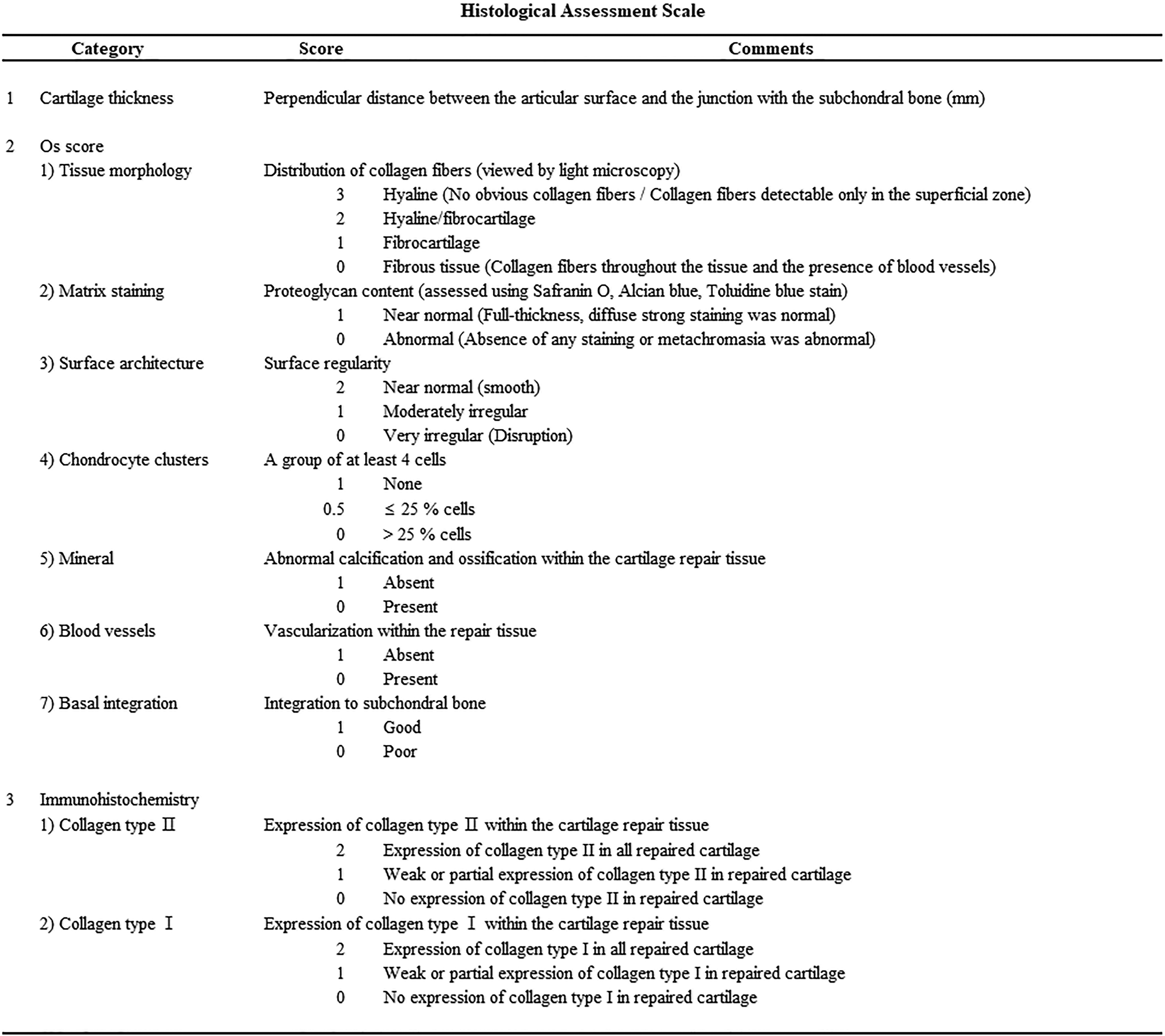
Histological assessment scales.
Surgical technique
Surgery was conducted in two steps. After the completion of the preoperative clinical evaluation and standing knee anteroposterior and lateral radiographs to evaluate for the presence of OA and deformity of the knee joint, arthroscopy was performed to measure the size and depth of the cartilage lesion. The presence of damage to the menisci, ligaments, and cartilage was assessed. Sample was collected 200–300 mg of cartilage from non-weight-bearing sites such as the upper ridges of the medial and lateral condyles of the knee or the intercondylar notch. The collected samples were sent to a Good manufacturing practice (GMP)-certified cell culture institution (Sewon Cellontech, South Korea). Corrective HTO was performed in patients with malalignment to create a neutral mechanical axis before the ACI procedure. Surgery of all cases was done by open-wedge method with TomoFix™ locking plate (Synthes, Oberdorf, Switzerland).
ACI was performed when 1.2 × 107 chondrocytes were cultured in one vial 4–6 weeks after the first surgery. The cell viability was more than 80% prior to the final packing. 18,19 During the second surgery, an arthrotomy was performed using the medial or lateral patellar approach depending on the cartilage defect site. Following the release of the tourniquet, bone bleeding was stopped with the application of direct pressure on the holes with a piece of gauze soaked in epinephrine. Two 1-mL syringes and a Y-connector were used for the injection. One syringe was filled with 1 mL of fibrinogen (75–115 mg/mL, Green Cross, Korea), and the other was filled with a 0.9-mL cell suspension and 0.1 mL of thrombin (400–600 UI/mL, Green Cross). The cultured autologous chondrocyte and fibrin were mixed at a 1:1 ratio and then slowly injected into the defect site. The number of vials that contained autologous chondrocyte was different depending on the size and depth of the lesion. Considering cell viability (more than 80%), the chondral defect was usually covered with two vials, but if the defect size was over 4 cm2, we used four vials for sufficient number of cells. The dependent position was maintained for 5 min to prevent overflow at the edge while waiting for the implant to harden. The graft stability was routinely tested by cyclic bending of the knee while visualizing the graft position after examining the status of gel hardening using a probe. A standard wound closure was performed. Continuous passive motion was recommended after surgery, and weight-bearing was prohibited for 6 weeks. Partial weight-bearing was allowed using a crutch at 6 weeks after surgery, and full weight-bearing was allowed at 12 weeks postoperatively.
Statistical analysis
Categorical variables were presented using frequency and percentage, while continuous variables were presented using the mean, standard deviation, and range. Since the cohort of patients is small (16 patients), medians and ranges are also provided. Clinical comparisons between preoperative and postoperative clinical scores were performed using the paired t-test. Unpaired t-tests and Pearson correlation coefficients were used to evaluate changes in the following variables: age, sex, defect size, defect location, number of implanted chondrocytes, and time interval between the cartilage transplant and the ACI. A p value for a two-sided test was presented, and a p value <0.05 was considered significant. Data were analyzed using SAS (version 9.3, SAS Institute, Cary, NC, USA).
Results
Among the 16 patients in this study, 14 followed up until 2 years and had an MRI. Corrective HTO was performed in nine patients with malalignment, and the malalignment was corrected on the mechanical axis from 7.59° (median 6.9°, range 5.1°–12.3°) varus preoperatively to 0.69°(median 0.8°, range −1.3° to 4.4°) valgus postoperatively. The preoperative and postoperative outcomes of VAS, KOOS, IKDC score, and the modified MOCART score are presented in Table 3 (Figure 2).
Functional and radiological outcomes of all patients treated with fibrin–matrix autologous chondrocyte implantation.

KOOS: Knee Injury and Osteoarthritis Outcome Score; IKDC: the International Knee Documentation Committee; VAS: visual analog scale; mMOCART: modified magnetic resonance observation of cartilage repair tissue; QoL: quality of life.

Sagittal T2-weighted MRI image of a patient who underwent autologous chondrocyte implantation for a cartilage defect. (a) On the medial condyle, a large, full-thickness chondral defect (white arrow) was visible on the preoperative image. (b) One year after fibrin–matrix ACI, the defect was completely filled with repair tissue, but mild hypertrophy was present (white arrow). (c) Two years after fibrin–matrix ACI, the regenerated cartilage was maintained well at the area of cartilage defect on the medial condyle. However, there was mild edema and sclerosis of the subchondral bone. MRI: magnetic resonance imaging; ACI: autologous chondrocyte implantation.
Second-look arthroscopy was performed in 12 cases. Subsequently, arthroscopic evaluation indicated that the cartilage repair was adequate (mild hypertrophy existed) when compared with the surrounding cartilage (Figure 3). Regarding ICRS classification scores to assess the overall repair, two cases were grade I (superficial lesions), eight were grade II (partial thickness <50%), one case was grade III (partial thickness, >50%), and one was grade IV (full thickness). The MOCART score of the grade IV patient at 2 years after surgery was low (55 points).

(a) Initial arthroscopic assessment showing an articular cartilage defect of the medial femoral condyle. (b) Second-look arthroscopy showed regenerated cartilage with visible borders at the level of the surrounding articular surfaces, complete integration with the surrounding cartilage, a fibrillated surface, and mild hypertrophy.
Histological analysis was performed in 11 patients. Nine patients (81.8%) recorded the highest matrix staining for Os score. This indicates a high expression of GAGs. For the expression of type II collagen, eight patients (72.7%) recorded the highest score. Nine patients (81.8%) expressed similar or greater levels of type II collagen than type I collagen. The mean of matrix staining on Os score was 0.80, while mean type II collagen expression by immunohistochemistry was 1.75. The histological analysis indicated that well-developed GAGs and type II collagen that were actively biosynthesized in the cell-implanted sites had more type II than type I collagen. In addition, Alcian blue staining results indicated that GAGs accumulated by the implanted cells (Figure 4). The average thickness of the regenerated cartilage was 3.6 ± 1.6 mm, the average Os score was 8.5 ± 1.2 (median 9, range 5.5–9.8) points (Table 4).

Histological analysis of second-look biopsy harvested from the patient treated with fibrin-mixed ACI at 12 months. (a) Hematoxylin and eosin staining, (b) Masson’s trichrome staining, and (e and f) collagen type I and type II immunohistochemical staining showed ideal regeneration of articular cartilage without disruption. (c) Safranin O staining and (d) Alcian blue staining showed even distribution of proteoglycan and GAG. These results indicate that fibrin-mixed ACI resulted in regeneration of hyaline cartilage. ACI: autologous chondrocyte implantation; GAG: glycosaminoglycan.
Histological outcomes of all patients treated with fibrin–matrix autologous chondrocyte implantation.

There were five cases of hypertrophy in the repaired cartilage, two of which proliferated more than the normal cartilage. However, there was no discomfort reported in patients with hypertrophic cartilage. Any other complications were not observed in the patients.
Correlation analysis between modified MOCART score with thickness, Os score, and immunochemistry score using Pearson–Spearman correlation method was conducted. The overall modified MOCART score did not significantly correlate with the histological finding. (Table 5)
Result of correlation analysis between mMOCART score and histological finding (Pearson–Spearman correlation).
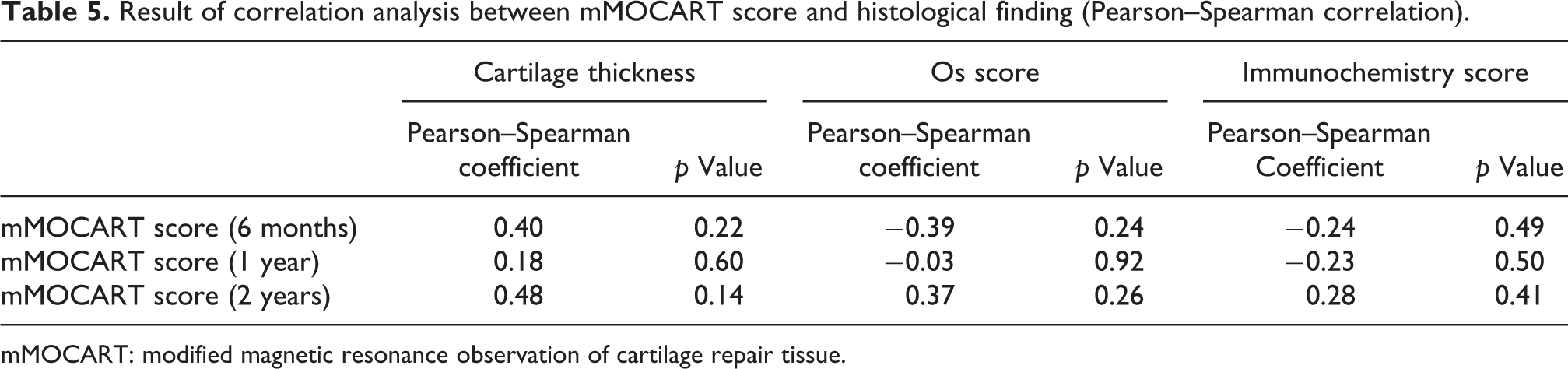
mMOCART: modified magnetic resonance observation of cartilage repair tissue.
When the outcomes 2 years after surgery were compared between the treatment groups, the mean KOOS scores of patients who had and had not undergone HTO were 76.2 ± 16.0 and 78.3 ± 12.6, respectively, for pain (p = 0.805); 67.5 ± 12.8 and 62.9 ± 14.9, respectively, for symptoms (p = 0.553); 73.4 ± 16.2 and 80.9 ± 12.2, respectively, for ADL (p = 0.385); 45.6 ± 27.1 and 53.0 ± 32.1, respectively, for sports (p = 0.652); and 51.4 ± 15.6 and 51.3 ± 6.9, respectively, for quality of life (p = 0.985). Additionally, after 2 years’ follow-up, the mean values for IKDC, VAS, and modified MOCART in those who had and had not undergone HTO were IKDC 50.0 ± 15.4 and 53.0 ± 14.2, respectively, (p = 0.726); VAS 2.4 ± 2.1 and 1.6 ± 1.3, respectively, (p = 0.452); and modified MOCART 72.2 ± 18.7 and 76.7 ± 21.4, respectively (p = 0.676). The average thickness of the regenerated cartilage was 3.9 ± 1.8 mm in patients who had undergone HTO and 2.8 ± 0.7 in those who had not (p = 0.361); and average Os score was 8.5 ± 1.4 points in patients who had undergone HTO and 8.5 ± 0.9 in those who had not (p = 0.990).
Discussion
We have performed the gel-type fibrin–matrix ACI in patients more than 50 years old with a chondral defect in the knee, which confirmed that the postoperative VAS, KOOS, IKDC score, and modified MOCART score had improved compared with preoperative score. Histological analysis through second-look arthroscopy and biopsy also showed well-developed GAGs and type II collagen.
Chondrocytes or chondrogenic cells have been used as implants in the treatment of physeal cartilage defects. 20 –24 Kim et al. reported that successful treatment outcomes were presented using fibrin–matrix ACI in patients with a mean age of 35 years. 8 Jones et al. suggested that ACI is commonly recommended on patients between fifteen and fifty-five years of age with excellent motivation and potential for compliance. 11 Although the efficacy of ACI for chondral defects in older patients is still controversial, there have been some reports of the good results of ACI in older patients. Rosenberger et al. performed ACI in 56 patients with cartilage defects older than 45 years. Of these, 28 patients with malalignment performed additional corrective osteotomy. As a result, at the final follow-up, they reported that 72% of the patients were considered to be in good or excellent condition, 78% had an alleviation of their symptoms, and 81% indicated that they would select ACI again in the future if they need it. 6 Furthermore, Niemeyer et al. also reported that performing ACI on older patients with chondral defects was an effective treatment. They reported that 74 patients who underwent ACI were divided into two groups: those older than 40 years or those younger than 40 years; clinical improvement was observed in both groups, and there was no statistically significant difference between the two groups. 25 Therefore, it was reported that ACI may be an effective treatment for chondral defects in older patients. In the present study, chondral defects were treated using gel-type fibrin–matrix ACI in patients whose mean age was 58.1 years. As a result, high patient satisfaction and good clinical results were obtained. The results of this study support the previous studies showing good results of ACI in older patients.
Saris et al. reported that when either ACI or microfracture was randomly assigned to young patients with a mean age of 33.9 years, the type II collagen generation rate was higher in the ACI group than in the microfracture group (p = 0.012). 26 Horas et al. performed ACI in patients with a cartilage defect who had a mean age of 31.5 years and confirmed the presence of type II collagen in the deep layer of the regenerated cartilage by histological analysis performed 24 months after ACI. 18 However, to our knowledge, there have been no studies to date that have reported on the histological analysis of the results of ACI performed in older patients. In the current study, second-look arthroscopy and histological analysis were performed 12 months after ACI. The second-look arthroscopy indicated satisfactory cartilage repair (nearly normal in the ICRS classification) in 83.3% of patients. Histological analysis confirmed the repair tissue with hyaline cartilage that contained well-developed GAGs and type II collagen. Therefore, ACI might be an applicable treatment option for a cartilage defect in older patients and induce the repair tissue with hyaline cartilage.
Takazawa et al. analyzed the follow-up MRI of 14 cartilage defect patients with a mean age of 33.1 years who underwent ACI. They reported that the MOCART score was 62.5 ± 24.7 at 1 year postoperatively and 70.7 ± 22.7 at 6 years; satisfactory cartilage regeneration was confirmed, and the implant was stable in the sixth year after surgery. 27 In a study conducted by Zak et al., ACI was performed in 23 patients who had a 2-year follow-up with clinical and MRI evaluations. The MOCART score increased from 49.1 ± 16.6 at 1 month after surgery to 73.2 ± 12.4 at 2 years after ACI. 28 Similar to the previously discussed studies, our study also showed satisfactory results with the MOCART score evaluating cartilage regeneration: 83.8 ± 17.3 at 1 year after surgery and 74.0 ± 19.2 at 2 years after surgery. However, the MOCART scores showed a decreasing trend at the 2-year follow-up compared to the 1-year follow-up. The degenerative changes appearing in the normal aging process of the cartilage could be the reason for the decreased MOCART scores, since this study is focused on older patients. Even though the MOCART score slightly decreased from the 1-year at the 2-year follow-up, it still showed satisfactory results compared with the 2-year follow-up results of other studies.
McCarthy et al. analyzed a correlation between MOCART and histological finding, and they reported that neither the structure of the repair tissue (on MRI) nor the overall signal intensity had any correlation with the tissue morphology of the biopsies assessed histologically. 29 Also, Tins et al. evaluated magnetic resonance (MR) imaging features of ACI grafts and compared these with graft histologic features 1 year after ACI. They reported that MR imaging findings cannot predict ACI graft histologic features, and graft histological appearance. 30 In the present study, MR findings and histological findings did not significantly correlate, and the results of correlation analysis were consistent with previous studies.
Malalignment causes degenerative changes in the articular cartilage as well as cartilage defects and can accelerate their progression. Rosenberger et al. found that the failure to correct malalignment can result in the recurrence of the chondral defect or outright transplant failure after ACI. 6 Therefore, they suggested that ACI and normalizing the mechanical axis through osteotomy should be attempted in patients with a narrowed joint space or malalignment in order to restore the joint through cartilage restoration. In our study, HTO was performed with ACI for the correction of malalignment, not for treatment, in patients with malalignment of 5° or more. The HTO alignment target was placed for neutralization and was not aligned with the Fujisawa point. 31 After the HTO was performed, the alignment was close to neutralization with 0.69° valgus. However, as corrective osteotomy is performed together, there may be controversy as to whether the improvement in clinical outcome is due to ACI. Therefore, we compared the HTO performed group and unperformed group to determine whether HTO has an effect on functional, radiological, and histological outcome. These comparisons of outcomes between the treatment groups revealed no statistically significant differences between them in KOOS, IKDC, VAS, or modified MOCART preoperatively or 2 years after surgery. Additionally, there were no statistically significant differences between the groups in regenerated cartilage thickness, Os score. Thus, it seems that HTO had no influence on the assessed outcomes.
This study had several limitations. First, this study had a follow-up period of 2 years, so long-term follow-up studies are required to observe the degenerative changes that may occur. Second, this study had only a small number of patients, and larger scale studies must be conducted.
Finally, this study did not include a control group with a different treatment method.
Conclusion
Satisfactory clinical and radiological outcomes were obtained from gel-type fibrin–matrix ACI technique in older patients with a cartilage defect of the knee. Repair of tissue with hyaline cartilage was also confirmed through histological analysis. Therefore, gel-type fibrin–matrix ACI may also be an applicable treatment for older patients with chondral lesions of the knee.
Footnotes
Acknowledgements
This study was supported by Inha University research grant.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Sewon Cellontech, South Korea.
