Abstract
Purpose:
There is no consensus on the optimum timing of administration of tranexamic acid (TA) in bilateral total knee arthroplasty (TKA). We aimed to determine whether the timing of administration of single-dose intravenous TA (either given preoperatively or intraoperatively) has a significant effect on blood loss reduction.
Methods:
We compared two cohorts of patients with end-stage arthritis of knees who underwent bilateral TKA and were given single-dose intravenous TA (1 g or 15 mg/kg) at different times during surgery. The retrospective cohort group consisting of 40 patients (preoperative (PO) group) received TA before the skin incision. The prospective cohort consisting of 40 patients (intraoperative (IO) group) received TA 10 min before deflating the tourniquet on the first knee. Primary outcome measures were mean hemoglobin difference, A (between PO and day 1 postoperative hemoglobin), mean hemoglobin difference, B (between PO and lowest postoperative hemoglobin), and rate of allogeneic blood transfusion. Secondary measure was drain blood loss.
Results:
Both cohorts were well matched with respect to age, gender, duration of surgery, and length of hospital stay. The hemoglobin drop in the IO group was significantly lesser than the PO group on the first postoperative day (2 vs. 2.9 g/dL, p < 0.001). Although statistically insignificant, the patients in the IO group received less allogenic transfusion of packed cell units than in the PO group (11/40, 27.5% vs. 14/40, 35% ). Mean hemoglobin difference, B, and secondary drain loss were comparable in both groups.
Conclusion:
Single-dose intravenous TA given before the start of surgery is as effective as a dose given during arthroplasty of the first knee in reducing blood loss in bilateral TKA.
Keywords
Introduction
Blood loss in total knee arthroplasty (TKA) can be minimized with controlled hypotension, meticulous surgical dissection, developing proper soft tissue planes, adequate hemostasis, the use of tourniquet, pharmacological agents, such as erythropoietin (EPO), iron, tranexamic acid (TA), and autologous transfusion. 1,2 Bilateral simultaneous TKA is associated with more blood loss than unilateral TKA, 3 rendering the management of blood loss more challenging in the former. TA is a synthetic derivative of amino acid lysine that blocks lysine-binding sites on plasminogen and prevents fibrin degradation. Recent literature with single-dose regimens in bilateral TKA has shown up to 80% reduction in transfusion rate as compared to controls. 4,5 In TKA performed under tourniquet control, TA can be given at the beginning of fibrinolysis (before skin incision) or at the peak of fibrinolysis (just before deflation of tourniquet). There are no comparative studies in literature addressing bilateral TKA and timing of single-dose intravenous regimen of TA. Our study was conducted to review the results of administering TA (same dose, same route) at different time periods during bilateral TKA and determine whether there is any difference in the efficacy.
Our null hypothesis was that a single-dose intravenous TA, whether administered intraoperatively or preoperatively, has a similar effect on blood loss reduction in bilateral TKA.
Materials and methods
The approval for the study was obtained from the institutional review board. We reviewed the records of 106 patients from January 2013 onward until May 2017. We included patients who underwent bilateral simultaneous TKA for advanced arthritis of knees and received TA during the surgeries. Between January 2013 and February 2015, the protocol for tranexamic administration was a single dose of 1 g (15 mg/kg) intravenous TA given before skin incision on the first knee (preoperative (PO) group), which formed the retrospective cohort. From March 2015 until April 2017, the protocol was a single dose of 1 g (15 mg/kg) intravenous TA given 10 min before the deflation of the tourniquet on first knee (intraoperative (IO) dose), which was the prospective cohort. Patients with allergy to TA, severe renal failure (those whose creatinine clearance was >250 µmol/l), and history of venous thromboembolism were not given TA and hence were excluded from the study. Patients who received IO blood transfusion were excluded from the study as well. From the electronic medical records, data were extracted and a masterchart was prepared to highlight the variables. Demographic information included were name, age, sex, weight, body mass index (BMI), diagnosis, and comorbidities. Duration of surgery, length of hospitalization, PO, and postoperative hemoglobin on days 1, 7, and lowest hemoglobin during the stay before allogeneic transfusion or discharge, hemoglobin difference, A (difference between PO hemoglobin and day 1 postoperative hemoglobin), hemoglobin difference, B (difference between PO hemoglobin and lowest postoperative hemoglobin), autotransfusion, allogeneic packed red blood cells transfusions, drain loss in each knee, and use of postoperative EPO/iron complex were documented.
The surgeries were performed by the same surgeon, using a tourniquet inflated to 350 mmHg at midthigh, under spinal anesthesia with or without sedation in all the cases. Antibiotic prophylaxis consisted of intravenous cefuroxime 1.5 g and gentamicin 160 mg. A fixed-bearing cemented posterior stabilized prosthesis with metal-backed tibial tray (Journey 2, Genesis 2 [Smith and Nephew] and Triathlon [Stryker]) was used in all the knees. An intra-articular autotransfusion drain (Suretrans™; Davol Inc. Warwick, Rhode Island) was inserted. The tourniquet was released after wound closure and compression dressings applied. The surgery on the second knee was begun subsequently. The drain was unclamped 30 min after wound closure. If more than 200 ml collected within 4 h postoperatively, the blood was retransfused to the patient. The drains were removed after 24 h and the collected volume was noted by nurses. Analgesia was maintained with morphine, nonsteroidal anti-inflammatory agents, and paracetamol. Deep vein thrombosis prophylaxis with thromboembolism deterrent stockings and low-molecular-weight heparin (enoxaparin 1 mg/kg subcutaneous) during in-patient stay and for another 4 weeks with aspirin 100 mg once daily was instituted. The patient was made to walk the next day by physiotherapists with the help of a support. Discharge was planned when the patient was confident enough to walk with a low frame walker. Postoperatively, EPO injection (Eprex 40,000 units/ml) was scheduled to be given subcutaneously on second and seventh day along with two to three intravenous infusions of iron sucrose complex (Feromax 400 mg, 20 ml) in 480 ml of normal saline at 125 ml/h over 3 days as directed by the physician. Allogeneic packed red blood cells (one unit containing 300 ml) were given when the postoperative hemoglobin was <8 g/dl or when symptoms of anemia such as fatigue, dizziness, or shortness of breath or hypovolemic symptoms such as tachycardia and hypotension were present.
Statistical analysis
The data were checked for duplicate entries and missing values. The continuous variables were tabulated with descriptive statistics. The categorical variables were tabulated with frequency tables. Normality of distribution was ascertained with histograms and q–q plots. Mean (with range) was used when data distribution was normal and median (with range) was used when the data were not normally distributed.
Student’s t-test (two sample, independent) was used to analyze normally distributed continuous variables. Mann–Whitney U-test was used to analyze data with non-normal distribution. Categorical variables were analyzed using χ 2 test or Fisher’s exact test when the expected count in any one cell was <5. The statistical analysis was performed using computer software (SPSS 19.0). A p value <0.05 was considered significant.
Results
Bilateral TKA was performed in 106 patients with advanced arthritis among whom 62 were in the PO group and 44 were in the IO group. Among the 62 patients between 2013 and 2014, 22 were excluded since there was no documentation of TA administration both in the electronic records as well as the physical anesthesia chart during the surgeries. Among the 44 in the IO group, four were excluded (one was not given TA, another received PO EPO, and the other two received IO transfusions). The final analysis was done between 40 patients each in PO and IO groups. Both the groups are similar with respect to age, BMI, operative duration, length of hospital stay, and PO hemoglobin level (Table 1). There was a female preponderance observed in overall sample which was maintained in both groups. The PO diagnosis was osteoarthritis in 79 patients and rheumatoid arthritis in 1 patient.
Distribution of demographic variables in PO and IO groups.
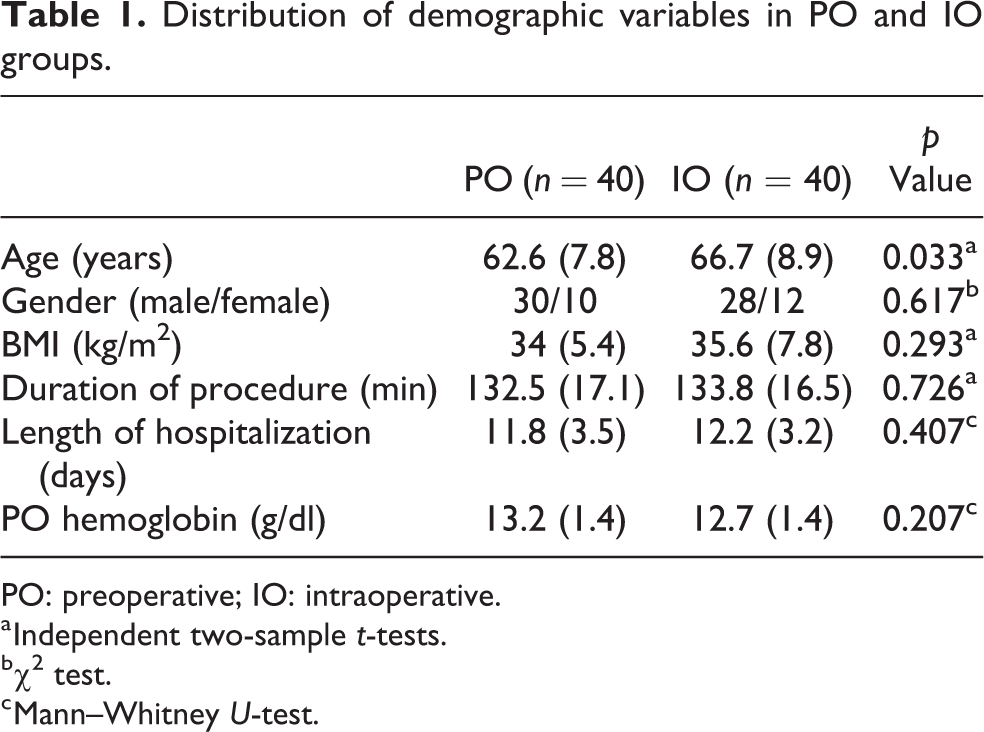
PO: preoperative; IO: intraoperative.
a Independent two-sample t-tests.
b χ 2 test.
c Mann–Whitney U-test.
The primary outcome measures were the difference in PO and the postoperative hemoglobin on day 1 (hemoglobin difference—A), the difference in PO and the lowest hemoglobin (hemoglobin—B) prior to receiving tranfusion or at discharge, and the number of allogeneic blood units transfused in each group. The secondary outcome measure was the drain loss in each knee in both groups (Table 2).
Distribution of outcome measures among both groups.

PO: preoperative; IO: intraoperative.
a Independent two-sample t-tests.
b Median values (minimum–maximum).
c Mann–Whitney U-test.
d Fisher exact test.
e χ 2 test.
All the patients were scheduled for follow-up at 6 weeks, 3–6 months, and 1 year. There were no incidences of venous thromboembolism. One patient developed anasarca with cirrhosis and renal failure at 3 weeks postoperatively; another developed posterior tibial arterial thrombosis on the right side (due to atrial fibrillation) at 12 days postoperatively and was treated with angioplasty and stent placement. Both were in the IO group. All the patients had a knee range of flexion up to or more than 90° at 6 weeks postoperatively.
Discussion
TA has been effective in blood loss reduction in both unilateral and bilateral TKA. Kim et al. 6 compared the use of intravenous two-dose and three-dose regimen of TA in unilateral and bilateral TKA, respectively. They analyzed 180 unilateral (90 each in unilateral TA and control groups) and 146 bilateral TKA (73 each in bilateral TA and control groups) in a randomized trial. The most important finding was that the use of TA had similar trends in reducing total blood loss and allogenic transfusion rate in both unilateral and bilateral TKA. Shinde et al. 7 conducted a similar study with three-dose regimen of intravenous TA in 28 patients in unilateral group (14 receiving TA and 14 controls) and 28 patients in bilateral TKA (14 receiving TA and 14 controls). They also concluded a similar trend in blood loss reduction in both unilateral and bilateral TKA.
The most common regimen of TA administration in TKA is the two-dose regimen, IO followed by postoperative dose TA. 8 Regarding bilateral TKR with multiple doses of TA, Maniar et al. 9 conducted a prospective randomized trial among 206 patients undergoing primary, unilateral TKA. They administered intravenous TA (10 mg/kg) in various regimens: intraoperatively, additional preoperative dose (POIO), additional postoperative dose (IOPO), all three doses (POIOPO), IO local application, and a control group. Each group had 40 patients. They concluded that the addition of another preoperative or postoperative dose in addition to single-dose TA was more effective, of which the POIO was better than IOPO. The best regimen was POIOPO. MacGillivray et al. 10 and Dhillon et al. 11 used a two-dose regimen of TA (IO, followed by postoperative) in their series of 40 bilateral TKA. They reasoned that most of the external blood loss occurs in the first few hours postoperatively after removal of tourniquet, and hence TA would be most effective in this period where fibrinolysis was at its peak.
Single-dose intravenous TA has been reported in three studies involving bilateral TKA. 4,5,12 Karam et al. 5 in their retrospective study compared 37 patients who underwent bilateral TKA were given a single-dose intravenous TA (20 mg/kg) before skin incision with 50 controls. Four among 37 (10.8%) received allogenic transfusion as compared to 25 patients (50%) in the control group. In another retrospective study by Bagsby et al., 4 46 patients undergoing bilateral TKA received single-dose intravenous TA (10 mg/kg) 10 min prior to tourniquet deflation compared to 57 controls. They reported a transfusion incidence of 17.4% (8/46) among those who received TA as compared to 57.9% (33/57) in the control group. Both studies demonstrated a reduction in the rate of transfusion by about 70–80% with the administration of TA. Comparing the two aforementioned studies, the PO group in the study by Karam et al. 5 received less allogenic transfusion compared to the IO group in the study by Bagsby et al. 4 This may be attributed to the shorter operating time and the higher dose of TA in the former study. The only randomized trial involving single-dose TA in bilateral TKA was conducted by Chen et al. 12 with a 1 g PO dose (before tourniquet inflation) in 60 patients against 60 controls. They reported transfusion rate in 36/60 patients (60%) in TA group as compared to 58/60 controls (96.7%). The transfusion trigger in all of the above studies was 8 g/dl. Our study showed a higher transfusion rate of 35% (14/40) in the PO group while 27.5% (11/40) in the IO group, albeit without statistical or clinical significance. The mean number of packed cells transfused per patient is almost similar, even after including the outlier who received six units in the PO group. The mean hemoglobin difference, A, was 0.9 but accompanied by a large standard deviation. Therefore, irrespective of a few statistically significant differences, there may not be a clinically significant difference between the two groups.
Sun et al. 13 performed the only randomized trial published in literature comparing different timings of TA (30 mg/kg) administration in unilateral primary TKA. The four groups were A and B (before and after tourniquet inflation), C (before tourniquet deflation) and D (no TA), with 45 patients in each group. They concluded that the optimal timing was 15 min before tourniquet inflation. Their reported transfusion rates were 4.4%, 17.8%, 20%, and 44.4% for A, B, C, and D, respectively, with transfusion trigger at 8 g/dl. There is about 80% blood loss reduction with a PO dose (A) as compared to an IO dose (C). Extrapolating the results and contrary to our study, we have reported a blood loss reduction of 20% with the IO dose, rather than PO dose. However, it is important to mention that their study was not adequately powered to detect a statistical significance between the subgroups.
Weng et al. 14 published the largest meta-analysis on bilateral TKA and the use of TA, which included eight randomized controlled trials and seven nonrandomized trials. They concluded that TA reduced the total blood loss, drain blood loss, and the need for blood transfusion. They, however, did not reach a consensus on the optimal regimen of administration of TA. Ker et al. 15 in their meta-analysis of 104 randomized trials across various surgical disciplines concluded that 1 g dose TA was sufficient for most surgeries while higher doses provided no added benefit. Wei et al. 8 in their meta-analysis of the use of TA in both knee and hip arthroplasties involving 39 studies concluded that there was no difference in the route of administration (intravenous, intra-articular, or oral) with regard to the efficacy. Interestingly, both the aforementioned studies suggested that the best time to administer the TA dose was after skin incision (5–20 min before deflation of tourniquet) rather than before incision. 8,15 This recommendation was based on the commonest mode of administration observed in the various studies involved in the meta-analyses.
There are several merits in the present study. The cohorts were well matched with respect to age, gender, BMI, and PO hemoglobin level. All surgeries were performed by the same surgeon, using a standard technique with posterior stabilized prostheses. The route and dose of TA were the same in all the patients. We did not attempt to establish the efficacy of TA in TKA because it is already known. Hence, we did not have a control group. Also, we did not exclude patients with cardiovascular diseases, respiratory diseases, super-elderly (>80 years of age), and mild–moderate renal disease as done by other authors. The chief exclusion criteria were severe renal failure, history of venous thromboembolism, and allergy to TA. This enabled us to evaluate the safety of TA, thereby generating data that may be of use in future studies investigating the role of TA in high-risk patients.
The limitations of the study necessitate mention. The sample size was small and the lack of statistical significance could represent a type II error. As this was a pilot study, we did not have previous data to rely on to conduct a power analysis. A post hoc analysis based on the 20% difference in blood loss reduction, significant at 0.05, and power set at 0.8 revealed a sample size of 600 patients in each cohort. It is a matter of debate whether it is justifiable to enroll 1200 patients to detect a difference that may not be clinically relevant.
The retrospective design of the first cohort (PO group) lends itself to inherent weakness in the quality of data. Although electronic medical records provided most of the information, many important elements were either missed (such as whether PO TA was given or not) or were prone to subjective errors (the recording of drain values by the ward nurse). The prospective arm of the study (IO group) eliminated these biases to an extent.
The major confounding factor in this study was the use of concurrent blood-saving strategy. We suspect that the observed lack of difference in the outcome measures other than hemoglobin difference, A, was due to the potent blood-saving action of EPO/iron therapy administered from day 2 postoperatively. Previously, in our institution, it was observed that the hemoglobin was very low on day 7 in patients undergoing knee arthroplasty and many of them required transfusions. EPO given postoperatively showed a marked improvement in the hemoglobin just before discharge and thus was adopted as an important part of postoperative care of the patient. Our intention was to provide EPO preoperatively but geographic constraints and the patients’ unwillingness to visit the hospital frequently made this impractical. Hence, all the patients were planned for EPO/iron therapy postoperatively. In our study, EPO was given as two doses postoperatively (40,000 units subcutaneously) on second and seventh postoperative days. Although all the patients were scheduled to receive them from second postoperative day, it was not mentioned in the records of 11 patients (7 in the PO group and 4 in the IO group).
In an effort to curb perioperative blood loss, postoperative blood salvage with the use of retransfusion drains has been advocated. We did not exclude the four patients in the PO group who had retransfusion of the drained blood (mean 600 ml) because most studies have reported that autotransfusion drains neither contribute to reduced blood loss nor increased hemoglobin levels postoperatively. 16 –18
Finally, we could not calculate the total blood loss and subsequently hidden blood loss. The hidden blood loss represents the difference between total blood loss (calculated from height, weight, PO, and postoperative hematocrit) and visible blood loss intraoperatively and in drains, which constitute 50% of true blood loss. 19,20 In our hospital, hemoglobin was calculated on day 1 and day 7 as protocol. Total blood loss could have been calculated if day 4 or 5 hemoglobin was available to us. This could have helped us address the effect of TA on hidden blood loss.
All the patients had their first follow-up at 6–8 weeks. We did not have any Venous thromboembolism (VTE) events in the patients during their first visit, which was within 2 months of surgery. It is known that the cumulative incidence of VTE after TKA within 3 months is 2.1%. 21 In particular, TA in TKA has been shown to reduce the blood loss and transfusion rate without an increased risk of VTE. 8,22,23 It is, however, difficult to elucidate the role of TA in causing VTE since most of the studies exclude high-risk patients in their trials such as history of cardiovascular disease, thromboembolic events, renal failure with creatinine level >250 µmol/l, allergy to TA, and those on warfarin or on therapeutic dose of low molecular weight heparin.
We believe that a randomized trial involving more patients without concurrent blood conserving strategies will reliably define the optimal timing of administration of TA in bilateral TKA.
Conclusion
TA given as single intravenous PO dose (15 mg/kg) is as effective as an IO dose in reducing blood loss in bilateral knee arthroplasty.
Footnotes
Authors’ note
A part of this study formed the dissertation of the first author (GB) who was admitted to the degree of Master of Surgery in Trauma and Orthopaedics (ChM) by The University of Edinburgh in 2017.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
