Abstract
Objective:
The aim of this study was to evaluate the mid- to long-term outcomes of revision surgery for Vancouver type B2 periprosthetic femoral fractures (PFFs) after total hip arthroplasty using an uncemented extensive porous titanium-coated long femoral stem prosthesis (solution prosthesis) with or without cortical strut allograft.
Methods:
A total of 34 patients with Vancouver type B2 PFFs who underwent revision hip arthroplasty using a posterolateral approach and received a solution prosthesis at our institution from December 2006 to January 2014 were retrospectively recruited. Patients were asked to assess their walking ability and pain status using a questionnaire. Limb function, pain, and physical and mental health were assessed using the Harris Hip score, University of California-Los Angeles score, Western Ontario and McMaster Universities osteoarthritis index, and Short Form-12 Health Survey score. Patients were also asked to rate their satisfaction with the surgery. Fracture union and stress shielding were assessed by radiography, and computed tomography was used to assess stem fixation. Single-photon emission computed tomography performed to assess radionuclide distribution in patients given cortical strut allografts. Patients were asked about their condition before PFF and evaluated at 6 weeks, 3 months, 6 months, 1 year, and annually after surgery. Any complications during follow-up were recorded.
Results:
Of the 34 patients, 29 completed follow-up. There was no significant difference in patient’s health before PFF or at the last follow-up. Fracture union was achieved in all patients. Mild-to-moderate stress shielding occurred in three patients, and no patients exhibited femoral stem loosening or obvious subsidence. Radionuclides were concentrated in the cortical strut transplantation area, and the cortical strut was integrated with the host femur. The incidence of postoperative complications was low.
Conclusions:
Revision surgery using the solution prosthesis with or without cortical strut allograft is effective in treating Vancouver type B2 PFFs, with satisfactory mid- and long-term clinical and radiological outcomes.
Introduction
Periprosthetic femoral fractures (PFFs) during or after total hip arthroplasty (THA) pose a severe challenge for orthopedic surgeons. 1 The most common cause of PFFs is bone fragility and a loose prostheses, which can be influenced by numerous factors, including gender, age, metabolic disease, inflammatory arthropathy, biomechanical problems, osteoporosis, osteolysis, and surgical technique. 2 PFFs are the third most common cause of revision hip arthroplasty after prosthesis loosening and recurrent dislocation, 3,4 occurring in approximately 1% of patients after primary THA and 4% after revision surgery. 5 –7 With the increasing number of primary and revision THAs performed each year, the number of PFFs is expected to rise. 8
The Vancouver classification categorizes PFFs based on location, implant stability, and residual bone mass, and it recommends surgical treatment algorithms. 9,10 Vancouver type B2 PFFs are defined as fractures occurring around the stem or just distal to it and with a loose stem. 11 This PFF type is particularly difficult to treat due to loosening of the prosthesis. Most patients with type B2 fractures are elderly, with poor bone quality and chronic diseases. Surgical treatment of these injuries is difficult and often associated with complications. Many surgical techniques and implants have been used to manage Vancouver type B2 PFFs. 12,13 The most common surgical method is open reduction and femoral stem revision. 14 –16 However, there is no clear consensus on the optimal surgical technique and stem design for revision surgery. 11
In this study, our aim was to evaluate the effectiveness of femoral stem revision surgery in patients with Vancouver type B2 PFFs using the solution prosthesis (DePuy Synthes, New Brunswick, New Jersey, USA). The solution prosthesis is an uncemented, nonmodular, distally fixed prosthesis with a porous titanium-coating on seven-eighths of the stem surface (Figure 1). We chose to study patients using this prosthesis because prostheses with a long-stem or uncemented extensive coating can give better patient outcomes than those with a short stem 17,18 or that are cemented. 19 Radians on the sagittal plane help accommodate the physiological anatomic radians of the femur. Bone can easily grow into the porous titanium-coating, which improves prosthesis stability. The prosthesis has a cylindrical distal end with a length of 4–6 cm in contact with the femur and a stem with a rectangular cross-section creating a larger contact area to reduce stress shielding. 20 Based on these considerations, we reasoned that the solution prosthesis should give optimal outcomes in the treatment of type B2 PFFs. Patients with transverse or short-oblique femoral stem fractures and reduced femoral cortical bone were also concurrently given a cortical strut allograft.

The solution prosthesis.
Methods
Patients
We retrospectively analyzed 34 patients with Vancouver type B2 PFFs who underwent revision hip arthroplasty using a posterolateral approach and a solution prosthesis (DePuy Synthes) at West China Hospital from December 2006 to January 2014. All patients who satisfied these inclusion criteria were eligible for enrollment in the study. This study was approved by the Clinical Trials and Biomedical Ethics Committee of our institution, and written informed consent was obtained from all participants.
Preoperative management
Patients were encouraged to sit up on their beds and train cough and sputum. Low-molecular-weight heparin was prescribed to prevent deep vein thrombosis.
Prosthesis implantation
All revision surgeries were performed using the posterolateral approach. The extent of the incision and exposure were determined by the fracture location. If exposure and extraction of the implant were difficult, the extension method was used. The surgery was performed by first dislocating the hip and exposing the fracture. Granulation and scar tissue at the fracture end and the cement and pulp plug at the distal end of the medullary cavity were removed. The fracture was then reset and fixed using cerclage wire. Next, the femoral stem was removed and the medullary cavity was cleaned. Dry gauze was packed into the medullary cavity to reduce bleeding. Finally, the medullary cavity was dilated and flushed to finish implantation of the prosthetic stem. To ensure prosthesis stability, the prosthetic stem was placed beyond the distal end of the fracture at a distance of least twice the diameter of the femoral stem (usually at least 4 cm).
Preparation of cortical strut allografts
Cortical struts were made by refrigerating the allogeneic tubular bone at −70°C for 4 weeks and then soaking it in povidone-iodine for 30 min. After rewarming, the bone was cut into cortical struts with an average length of 14.6 ± 3.2 cm using an electric swing saw. Allogenic cancellous particles were also prepared and treated using the same methods as cortical struts. The cortical struts and allogenic cancellous particles were washed with antibiotic saline repeatedly, degreased with 75% ethanol, and then washed again with antibiotic saline.
Cortical strut allografting
Fracture reduction was performed after removal of the prosthesis and bone cement. Two cortical struts were then implanted in the anteromedial and lateral sides of the femur, followed by wire cerclage fixation. Allogenic cancellous particles were implanted between the cortical struts, between the cortical struts and the host bone, and in the femoral medullary cavity.
Postoperative care
After revision surgery, all patients were given antibiotics and medication to prevent thromboembolism and pain. Patients were required to start knee and hip movements in bed as soon as possible and partial weight-bearing exercises within the first 6 weeks of surgery. After 6 to 12 weeks, patients progressed to walking with full weight. Clinical and radiological assessments were performed at 6 weeks, 3 months, 6 months, 1 year, and annually after surgery. All patients were advised to return to our institution for follow-up.
Clinical assessment
We evaluated patient bone condition before the PFF by asking patients to assess their walking ability (free walking or limited walking) and pain status (none, mild, moderate, or severe pain) prior to surgery. Patients were also asked to complete a standardized self-administered questionnaire to assess their Harris Hip score, University of California-Los Angeles (UCLA) score to assess limb function, 21 Western Ontario and McMaster Universities (WOMAC) osteoarthritis index, 22 and Short Form-12 Health Survey (SF-12) score to assess physical and mental health. 23
To assess clinical outcomes after surgery, patients were again asked to assess their walking ability and pain status and complete the same questionnaires as above. Patients were also scored for their satisfaction with the surgery. 24 Any complications during follow-up were recorded.
Radiographic assessment
Patient bone condition before surgery was evaluated using a preoperative radiograph to assess femoral cortical bone mass, bone density, and detect osteoporosis.
During postoperative follow-up, a full-length radiograph was taken of the femur from the anterior and lateral views. The femur was assessed for fracture union (defined as the presence of osseous trabeculae crossing the fracture line) and stress shielding using a modified Engh and Bobyn grading method. 25 Computed tomography (CT) was used to evaluate fixation of the stem. Stability of the stem is defined as bone ingrowth stable, fibrous ingrowth stable and unstable, respectively, according to different imaging manifestations. 26,27 Strut graft healing was classified as none, partial, or complete based on the review of radiographs for union and bridging. Allograft resorption was defined as partial resorption of one cortex, where <1 cm resorption was defined as mild, >1 cm as moderate, and full resorption as severe. 28 All cortical strut allograft patients underwent single-photon emission computed tomography to visualize the distribution of radionuclides in the cortical strut transplantation area.
Statistical analysis
Statistical analysis was performed using SPSS 25 (IBM, Chicago, Illinois, USA). All data are presented as mean ± standard deviation, unless otherwise indicated. Intergroup differences in continuous variables (e.g. Harris Hip score and UCLA score) were assessed using the Mann–Whitney U test. Differences in categorical variables (e.g. walking ability and pain status) were assessed using Pearson’s χ 2 test or Fisher’s exact test. Differences associated with a p-value of <0.05 were considered statistically significant.
Results
In total, we enrolled 16 males (47.1%) and 18 females (52.9%), comprising a total of 19 fractures on the right side (55.9%) and 15 on the left (44.1%) (Table 1). The mean age was 70.6 ± 13.6 years (range 33–92 years). Of the 34 PFF cases, 32 were caused by falling (94.1%), while the remaining 2 had no obvious cause (5.9%).
Basic information of the included patients.
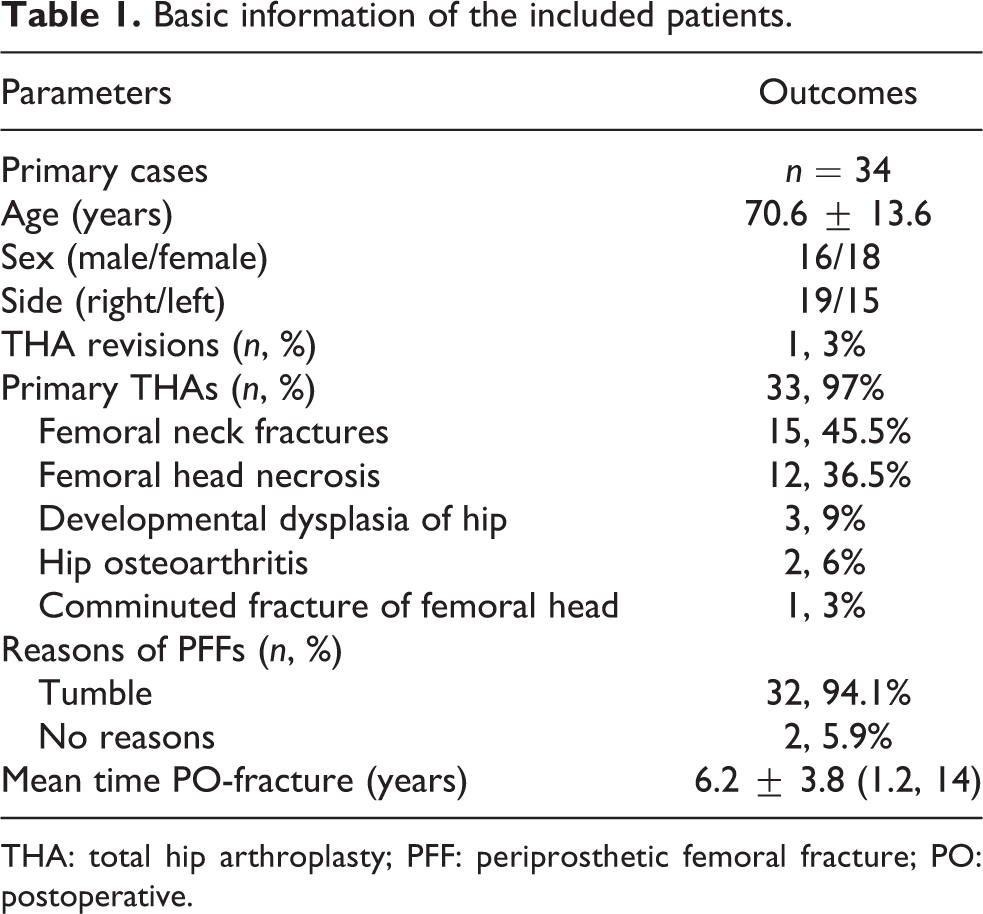
THA: total hip arthroplasty; PFF: periprosthetic femoral fracture; PO: postoperative.
PFF occurred after primary THA in 33 cases (97%) and after THA revision in 1 case (3%). Among the primary THAs, there were 15 femoral neck fractures (45.5%), 12 cases of osteonecrosis of the femoral head (36.5%), 3 cases of developmental hip dysplasia (9%), 2 cases of osteoarthritis (6%), and 1 comminuted fracture of the femoral head (3%). The average time from the last surgery to PFF was 6.2 ± 3.8 years (range 14 months–14 years), and the average length of the operation was 107 ± 31 min. Sixteen patients (47%) also underwent concurrent acetabular component revision, and six patients (17.6%) with transverse or short-oblique femoral stem fractures and less femoral cortical bone also received a cortical strut allograft comprised of two cortical struts. The average hospital stay length was 17 ± 6.9 days.
Patients were followed up for an average of 102 ± 24.5 months (range 55–140 months). Three patients (8.8%) died in the follow-up period, one due to cardiovascular and cerebrovascular disease, one due to pulmonary fibrosis, and one due to natural causes. The three deaths occurred at an average of 69 ± 26.1 months after revision surgery, and none was directly related to the operation. Two additional patients (5.9%) were lost to follow-up, leaving a total of 29 patients (85.3%) who completed follow-up. As of August 2018, 28 patients (96.6%) had not required additional surgery, while one (3.4%) had an additional surgery due to refracture.
Clinical outcomes
Patients started full-weight walking at an average of 4.6 ± 1.0 months after surgery. Prior to the PFF, 26 patients (89.7%) could walk freely and 3 walked with crutches (10.3%). By the final follow-up (55–140 months after surgery), 24 patients (82.8%) walked without crutches, 4 walked with crutches (13.8%), and 1 could not walk due to old age (3.4%).
There was no significant difference in self-reported pain before PFF and at last follow-up (Table 2). The average Harris Hip score decreased slightly from 87.2 ± 5.4 before PFF to 85.6 ± 8.4 at final follow-up, but this decrease was not significant. When excluding the one patient who could not walk due to old age, the mean Harris Hip score at final follow-up was 87.0 ± 4.1. Between the preoperative period and final follow-up, there was no significant change in limb function or pain as assessed by the UCLA score or WOMAC score for pain, stiffness, and function. Similarly, SF-12 survey scores did not change significantly over the same period for physical health and mental health. The patient satisfaction score was 88.6 ± 6.7 at the last follow-up.
Clinical assessment results before fracture and at last follow-up.a

WOMAC: Western Ontario and McMaster Universities; SF: Short Form-12 Health Survey.
aWOMAC and SF-12 scores are normalized to a range of 0–100 points, with 0 being worst and 100 being best. Satisfaction score: 0 is worst, 100 is best.
bPearson’s χ 2 test.
cMann–Whitney U test.
Complications occurring during follow-up included one patient who had a refracture and underwent an additional revision surgery at 7 months; one case of hip dislocation 16 days after surgery, which was treated by closed reduction; two cases of venous thrombosis in the lower extremities, which did not require special treatment; and one patient who returned to the hospital 6 days after surgery with severe hip pain. After 6 days of conservative treatment by functional exercise and medication, the patient was discharged.
Radiographic outcomes
Preoperative radiography showed that of the 29 patients, 6 had decreased cortical bone mass of femur and 9 had decreased bone density. A total of 9 patients showed signs of osteoporosis. (Table 3). Radiographic assessment after revision surgery showed that all patients had achieved fracture union by the final follow-up (Figures 2 to 7). The average time to fracture union was 4.3 ± 1.2 months (range 3–6 months; Table 4). The average length of femoral stem fixation beyond the distal end of the fracture was 5.2 ± 0.3 cm.
Bone condition before fracture.

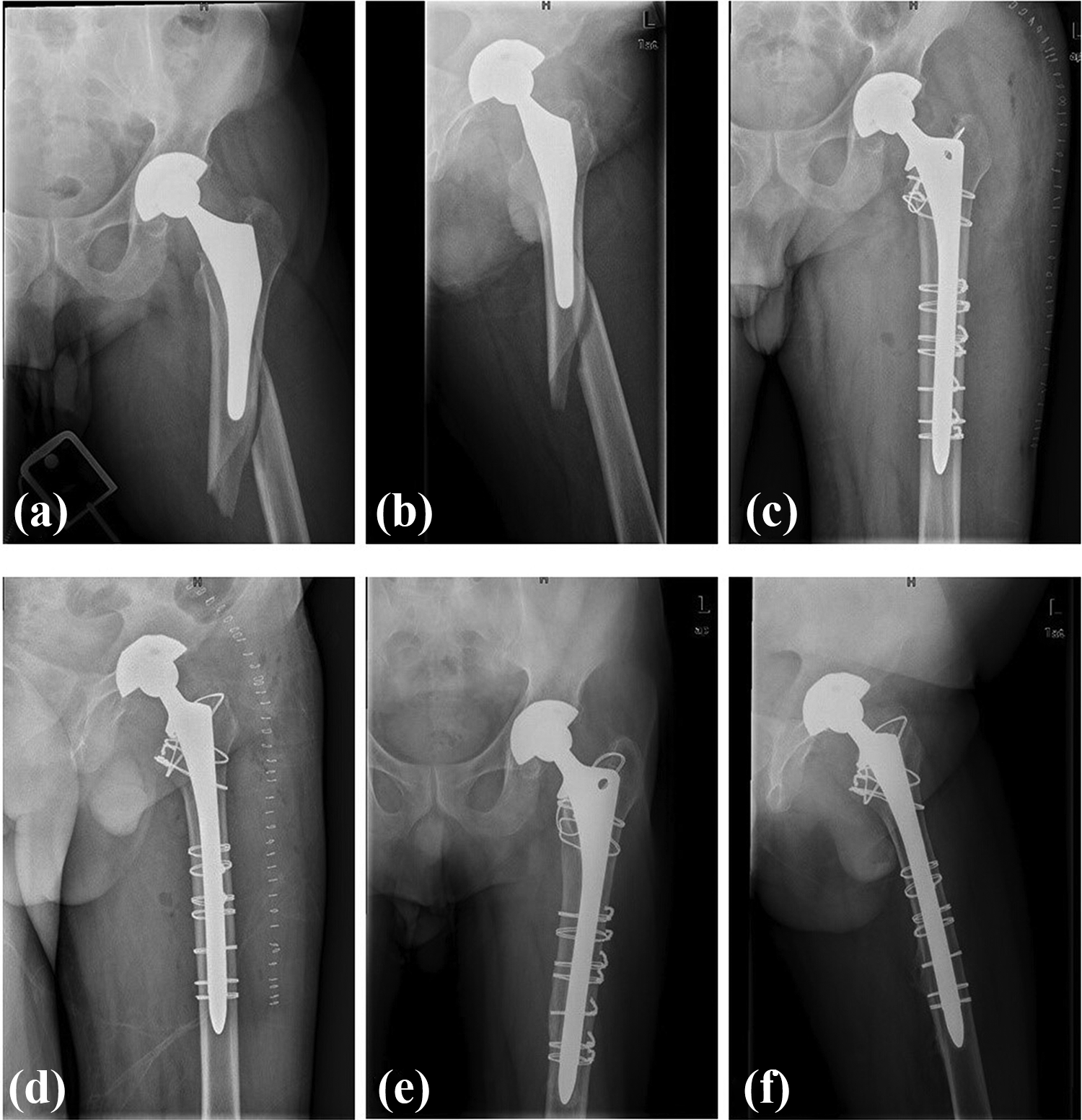
(a and b) Preoperative and (c and d) postoperative radiographs of a Vancouver type B2 periprosthetic femoral fracture that was treated with the solution prosthesis combined with wire-ring fixation. (e and f) The radiographs at 98 months of follow-up.

(a and b) Preoperative and (c and d) postoperative radiographs of a first periprosthetic femoral fracture that was treated with cortical strut allograft and wire-ring fixation.

The radiographs belong to the same patient of Figure 2. (a and b) The radiographs of a refracture that occurred 7 months after the first surgery. It was a Vancouver type B2 periprosthetic femoral fracture. (c and d) The postoperative radiographs of the Vancouver type B2 periprosthetic femoral fracture treated with the solution prosthesis combined with cortical strut allograft. (e and f) The radiographs at 1 month of follow-up.

The radiographs belong to the same patient of Figure 2. (a and b) The radiographs at 6 months of follow-up. (c and d) The radiographs at 22 months of follow-up. (e and f) The radiographs at 112 months of follow-up.
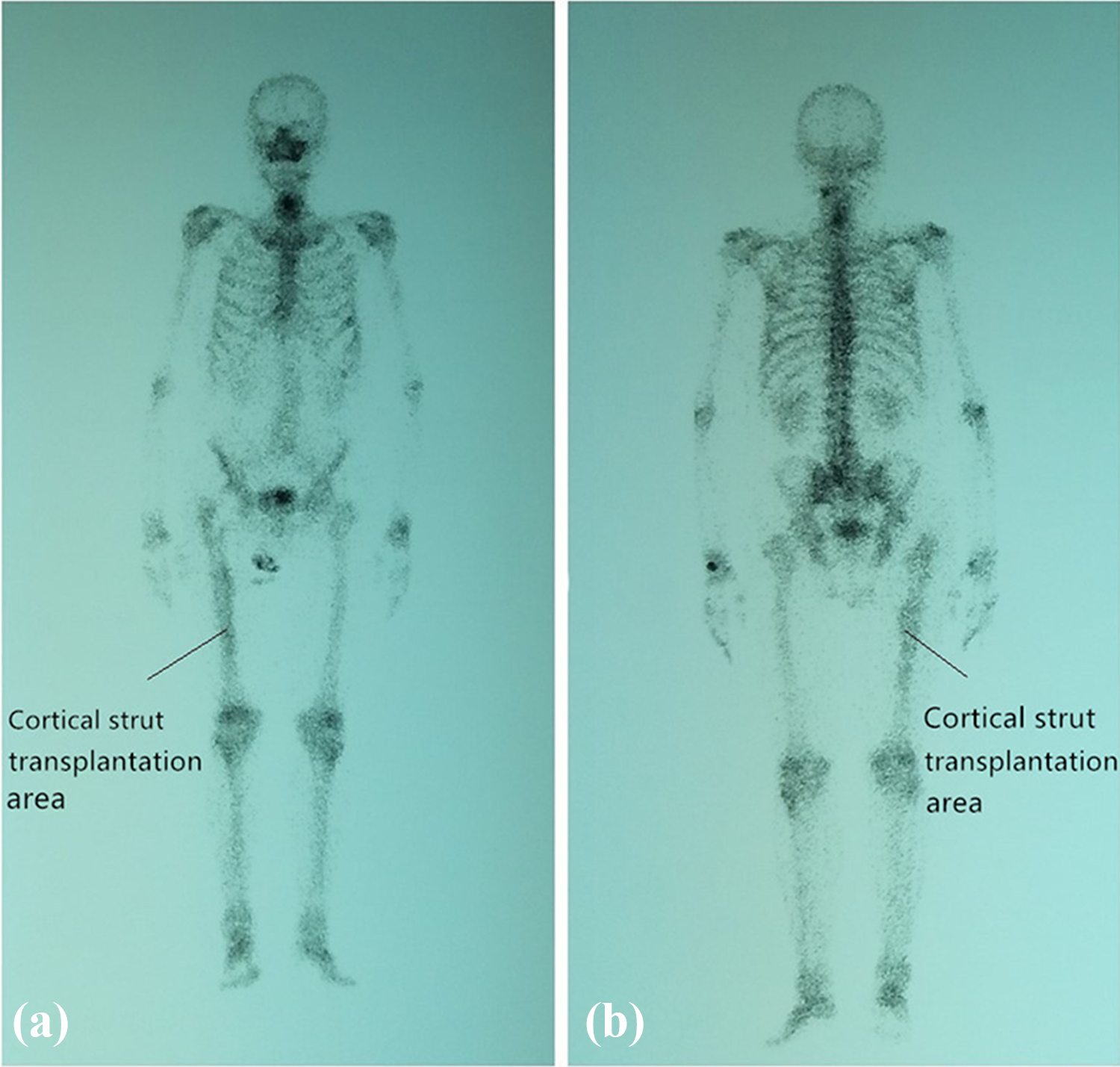
The radiographs belong to the same patient of Figure 2. (a and b) The radiographs of systemic radionuclide bone imaging with SPECT at 112 months of follow-up. The arrows in (a and b) pointed to the cortical strut transplantation area. SPECT: single-photon emission computed tomography.

The radiographs belong to the same patient of Figure 2. (a and b) The radiographs of systemic radionuclide bone imaging with SPECT at 112 months of follow-up. The arrows in (a and b) pointed to the cortical strut transplantation area. SPECT: single-photon emission computed tomography.
Radiographic assessment results at the last follow-up.
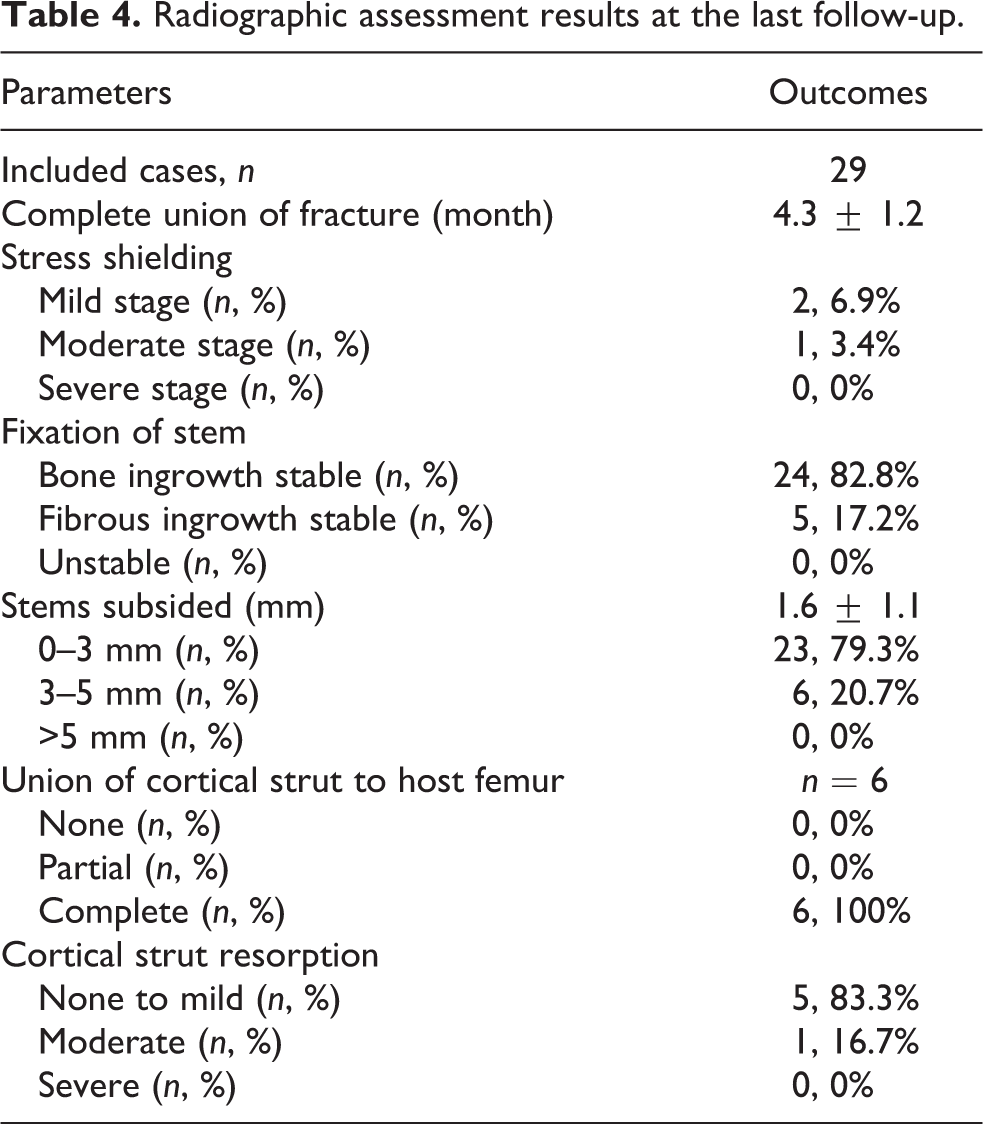
At the final follow-up, two patients had mild stress shielding at the proximal femur and one had moderate stress shielding. Twenty-four patients had achieved stable stem bone growth, while five showed fibrous ingrowth stable, and no patient had the stem not yet stable. There were 23 cases with a stem settlement of ≤3 mm and 6 cases with a stem settlement between 3 mm and 5 mm, with no cases showing > 5 mm settlement.
Of the six patients with cortical strut allografts, all achieved complete osseous union of the cortical strut to the host femur. One patient had moderate cortical strut resorption, while the remaining five did not exhibit any resorption or had mild resorption. Radionuclides were more concentrated in the cortical strut transplantation area than the contralateral side (Figures 6 and 7). Cortical struts had integrated with host bone by an average of 2 years after surgery, and resorption had ceased.
Discussion
In this study, we show that femoral revision surgery using the solution prosthesis is effective in treating Vancouver type B2 PFFs after THA, with the successful recovery of limb function and health in the mid- to long-term. Fracture union occurred in all patients with only a small number of complications.
PFFs are a major challenge to orthopedic surgeons and are often accompanied by bone loss, development of bone defects, and prosthesis loosening. Fixation is also frequently associated with complications such as fracture nonunion and prosthesis loosening. 15 Many surgical techniques and prostheses are available for treatment, but there is still no agreement on the optimal strategy. For type B2 fractures, proximal fixation prosthesis is unsuitable as it hinders bone growth due to the mutual fretting between the proximal fracture block and prosthesis. A modular distal cemented prosthesis is also disadvantageous as the cement may be exuded from the fracture end, creating a compressive effect that interferes with fracture healing and may lead to complications such as prosthesis loosening and delayed union. Cemented prostheses have also been reported to have a higher refracture rate of 15% compared to 7% in uncemented prostheses. 29 Uncemented, extensive-coated long-femoral-stem prostheses have been reported to perform better than partial-coated or cemented prostheses. 15,19 Therefore, we chose the solution prosthesis to treat Vancouver type B2 PFFs.
Our patients with transverse or short-oblique femoral stem fractures and less femoral cortical bone were concurrently given a cortical strut allograft. Large cortical struts are known to be effective in treating periprosthetic fractures 30 and have an elastic modulus and strength similar to that of bone, which minimizes stress concentration. Allogeneic bone contains growth factors that promote fracture healing, is nonimmunogenic, and helps increase bone mass and strength. 31,32 Cortical strut allografts can even be used for type B fractures in patients with healthy bone mass and without a loose femoral prosthesis to increase the fixed strength of the prosthesis and femoral cortical bone mass. 17
As shown in Tables 2 and 3, the living quality and bone condition of the patients before PFF were good. Only very few patients showed signs of severe osteoporosis. Therefore, there is often obvious trauma in the cause of these fractures, whether it is low-energy trauma such as indoor and outdoor fall, sprain, or high-energy trauma such as car accident injury and high-fall injury. 13,29 Of course, loose prostheses may also be a prerequisite for these fractures.
The majority of patients treated with the solution prosthesis recovered their prefracture functional level, including good limb function, quality of life, and radiographic outcomes, with the exception of one elderly patient. The incidence of complications was also low, with refracture occurring in only one patient (3.4%). All patients had achieved fracture union by the final follow-up, and the length of femoral stem fixation beyond the distal end of the fracture was greater than 4 cm, which matched well with the femur. The patients in our study achieved better clinical outcomes compared to those in previous studies, 11,33 –37 such as Harris Hip score (Table 5). Further studies with larger sample sizes will be required to determine whether this method will be effective for routine treatment of Vancouver type B2 PFFs.
Review the literature of the treatment of Vancouver type B2 periprosthetic femoral fractures.

PFF: periprosthetic femoral fractures; NR: not reported.
There are several potential disadvantages of using the solution prosthesis. First, the solution prosthesis is a nonmodular prosthesis, which makes regulate femoral offset decline. However, nonmodular stems can also increase the risk of shortening and dislocation. 38 Second, because the prosthesis is firmly fixed only at the distal end and patients with fractures have poor bone condition, only partial-weight functional exercises can be performed in the early stages of recovery. Third, the distally fixed prosthesis may cause stress shielding, which can reduce cortical bone at the proximal end of the femur and cause stress concentration at the distal end. However, we found a very low incidence of dislocation (3.4%) and stress shielding (10.3%). This is consistent with previous studies, which have also reported a low incidence of stress shielding in patients with the solution prosthesis. 39,40 A cortical strut allograft was also sufficient to enhance prosthesis fixation in patients with less femoral cortical bone. In summary, our analysis suggests that the solution prosthesis is an effective treatment for Vancouver type B2 PFFs, with relatively few complications occurring during mid- to long-term follow-up.
Our study involves longer follow-up (102 ± 24.5 months) than most previous studies, which followed up for 14.5–60 months. 11,33 –37 Nevertheless, our study still has several limitations. First, the sample is relatively small, so statistical power is low. Second, the retrospective design is open to biases that would be reduced with a prospective design. Third, the patients in our study are heterogeneous in bone condition before PFFs, so the results may not be applicable to all patients with type B2 fractures. Fourth, we did not include a control group as there were no suitable cases available. Despite these limitations, we found that the solution prosthesis consistently provided excellent fracture stabilization and healing in all patients. We, therefore, propose that uncemented extensive porous titanium-coated long-femoral-stem prostheses should be considered as a routine treatment for Vancouver type B2 PFFs.
Conclusions
Treatment of Vancouver type B2 PFFs after THA with uncemented, extensive porous titanium-coated, long-femoral-stem prostheses with or without cortical strut allograft is effective in promoting fracture healing and restoring patient’s quality of life. Revision surgery using these prostheses should be considered as a routine treatment for Vancouver type B2 PFFs.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Program of Sichuan Province [grant no. 2019YFS0123].
