Abstract
Purpose:
Fibrous dysplasia (FD) of the proximal femur can result in severe deformity and disability. The results of surgical management in such situations have been reported to be poor. We present a novel, five-step surgical treatment to correct proximal femoral deformity in FD.
Material and methods:
This study is a review of prospectively collected data of cases of proximal femur polyostotic FD managed at our institute from 2012 onward. We managed three patients with FD involving four femora (one patient had bilateral disease). Mean age at presentation was 16 years with an average follow-up of 46 months. All underwent five steps, that is, (1) curettage/excision of the lesion, (2) allograft in intramedullary region, (3) lateral closing wedge valgus osteotomy, (4) fixation with extramedullary implant, and (5) augmentation of osteotomy site with autograft. Functional outcome was assessed using Harris Hip Score preoperatively and at the final follow-up.
Results:
The neck shaft angle was corrected from an average of 91.7° to 152.1°, while the Harris Hip Score improved from an average of 59 to 95. There was no clinical or radiological evidence of recurrence of disease or deformity in any patient till the last follow-up.
Conclusion:
This five-step technique ensures good functional and radiological outcomes in the management of proximal femur FD.
Introduction
Fibrous dysplasia (FD) is a benign skeletal lesion and constitutes approximately 5% to 7% of all benign tumors. 1 It is a spectrum where the presentation ranges from asymptomatic monostotic disease to a polyostotic disease with significant deformities and disabilities. In addition, it may have syndromic presentations like McCune-Albright syndrome or Mazabraud syndrome. 2 The upper end of femur is most frequently affected, as fatigue fractures can occur in the pathologically weakened bone. This results in coxa vara, which can be severe in some patients resulting in “shepherd’s crook deformity.” 3,4
Many methods of treatment have been described in the literature with highly variable outcomes. These include curettage with the use of cancellous autografts, fibular strut autografts, and morcellized allografts. 5,6 Severity of the deformity along with weakened bone are the critical factors posing a challenge to the surgical management of this condition. 3,4 The attachment of bulky muscles to the proximal femur leads to strong muscular forces and this makes achieving anatomical correction a daunting task. Achieving a stable fixation in the weakened bone is also no easy feat. In severe lesions, particularly those associated with polyostotic FD, the medullary canal is wide and the lateral cortex is often lost due to the lesion and extensive valgus osteotomy is needed to achieve correction. 7,8 Usage of an intramedullary implant would not only be technically difficult but also may not provide adequate stability. 8 We hypothesized that this situation demanded both intramedullary and extramedullary support to achieve adequate stability. We report the results of a novel five-step technique adopted by us to treat severe shepherd’s crook deformities involving (1) thorough curettage/excision of the involved segment, (2) intramedullary strut allograft, (3) valgus osteotomy, (4) stabilization with extramedullary implant, and (5) augmentation with cancellous autograft (Table 1).
A five-step guide to correct shepherd’s crook deformity.

Material and methods
This study is a report of a series of three cases of proximal femoral FD managed in our institution from 2012 onward. Institutional Review Board approval was obtained for the study. Three patients with four proximal femoral lesions with mean age of 16 (range 12–20 years) at presentation were included in this study. All of them were patients with polyostotic FD
Preoperative planning
An anteroposterior (AP) radiograph of the pelvis was taken with lower limbs in internal rotation to make the trochanter prominent. The anatomical medial proximal femoral angle (MPFA) was drawn as shown in Figure 1(b) (In case of greater trochanter involvement, neck shaft angle can be used.) In unilateral cases, opposite hip measurements were used as a template to assess the deformity, whereas in bilateral cases, the normal anatomical MPFA (84° ± 5°) and neck shaft angles (130° ± 6°) were used. Proximal cut of the osteotomy was planned parallel to the line from the center of the femoral head to the tip of the greater trochanter and distal cut was planned perpendicular to the anatomical axis of the femoral shaft at a level just below the lesser trochanter (Figure 1(b)). In addition, full-length alignment radiographs were taken to calculate the shortening as well as associated deformities.
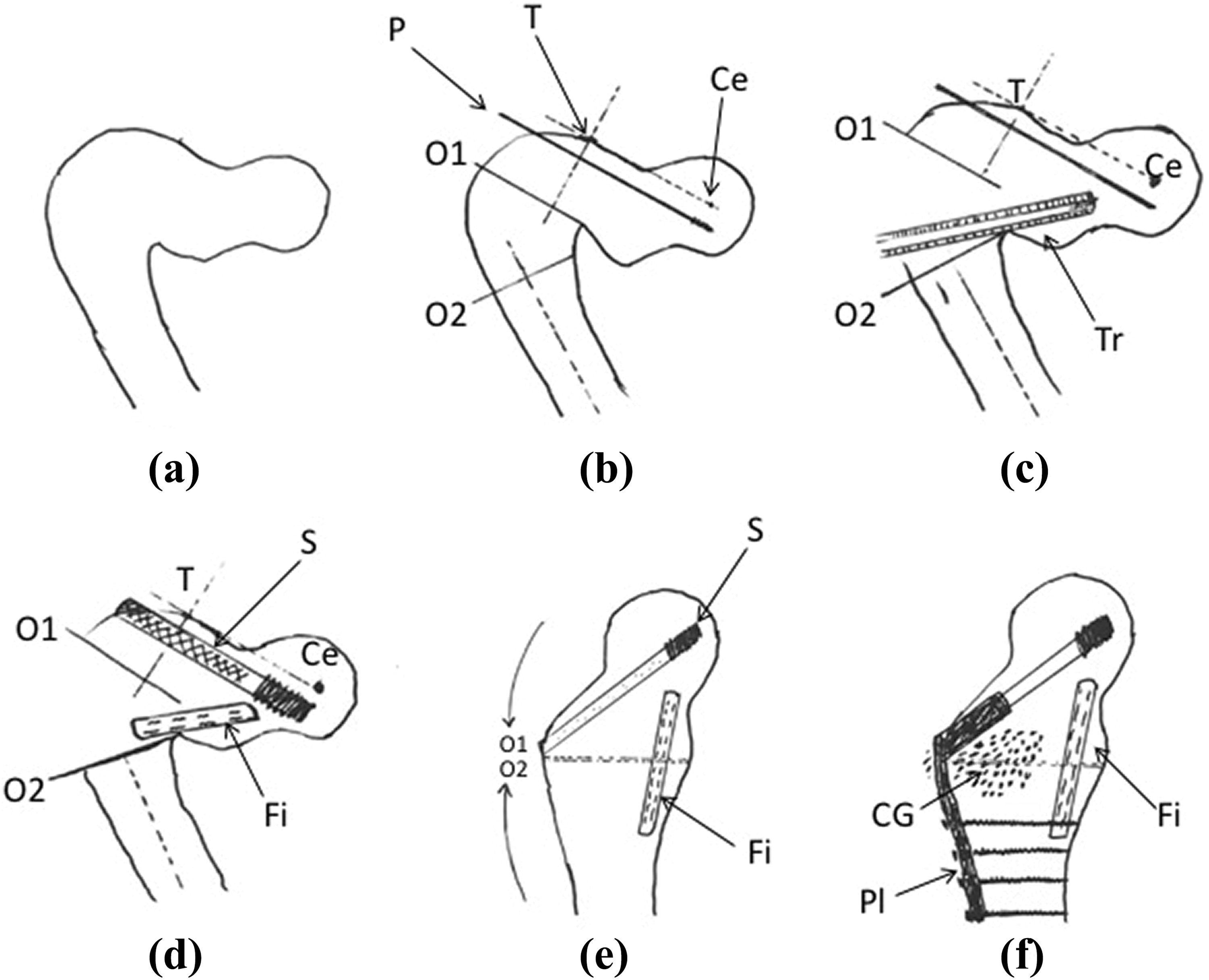
(a) Shepherd’s crook deformity of proximal femur; (b) preoperative planning and guide pin P introduced in inferior quadrant parallel to line TCe. Angle between line TCe and the long axis (dashed line) of femur is the anatomical MPFA; (c) osteotomy made along O1 and O2, Tr created for fibular strut graft; (d) S inserted along guide wire after triple reaming and tapping, Fi pegged into Tr; (e) osteotomy closed (solid arrows); (f) CG packed into osteotomy and curettage site, Pl applied. Ce: center of femoral head; T: tip of greater trochanter; O1, O2: site of osteotomy; P: guide pin; Tr: trough; Fi: fibular strut graft; S: lag screw; Pl: side plate; CG: cancellous bone graft; MPFA: Medial Proximal femoral angle.
Surgical technique
All patients were positioned supine with boot traction on a fracture table. The proximal femur was exposed using standard lateral approach. The key surgical steps are outlined in the line diagrams in Figure 1. The first step was to visualize the greater trochanter which was aided by keeping the limb in adduction and internal rotation. This was followed by thorough curettage to clear the diseased area. Samples were collected for histopathological confirmation of disease. Purchase in the proximal fragment was achieved with dynamic hip screw (DHS) or proximal screws of the locking compression plate (LCP). Intramedullary cortical strut allograft (fibula) was pegged in, in such a way that it supported the medial column and was in the axis of weight-bearing. When LCP was used, purchase was gained in the fibular strut with the proximal screws. Lateral closing wedge valgus osteotomy was performed at subtrochanteric level to correct the deformity. To improve the vector of body weight transmission and reduce stress on dysplastic bone, valgus over correction of neck shaft angle was done in all the cases. Osteotomy was closed by abducting the limb and adducting the proximal fragment with the implant (DHS Lag screw/LCP). Extramedullary stabilization was done with either a proximal femur locking compression plate, flipped over distal femur LCP of the opposite limb (Figure 8 right limb) or DHS plate. To improve the stability of fixation, longer implants were preferred. After fixation of implant, cancellous autograft chips were also placed into the osteotomy site.
In patients with severe deformities and large lesions fibular strut graft inserted in a tibial shaft cortical graft was used (Figure 6 left limb). Once the osteotomy was closed (with intramedullary strut graft in situ), it was observed that there was overlapping of the distal fragment by the proximal fragment. The limb was kept in neutral rotation and the overlapping bony margins were completely resected out. The bone ends were appropriately curetted, approximated and distal screws of the implant were fixed.
Postoperative protocol
Nonweight-bearing mobilization was started on the second postoperative day and partial weight-bearing was started after 6 weeks. Patients were followed up 6 weekly for radiological signs of union and full weight-bearing was started only after radiological union. Six monthly follow-up was done to look for recurrence, delayed collapse, and implant loosening. Harris Hip Score was used for functional assessment.
Case series
Patient 1
A 21-year-old male patient with a past history of multiple pathological fractures in his right humerus (managed in our institute) came with complaints of right hip pain and painful limp since the last 1 month. Radiological findings were consistent with FD of proximal femur (Figure 2).

Preoperative X-ray (a) and MRI (b) of patient 1. MRI: magnetic resonance imaging.
Neck shaft angle was found to be 75.6°. During surgery, a ballooned and cystic lesion filled with serous fluid was noted with a breach in femoral cortex anteriorly. The cavity was opened and thoroughly curetted using a high-speed burr. DHS was inserted through the proximal uninvolved part using a guide pin and reamer. A valgus subtrochanteric osteotomy with a wedge of approximately 55° was removed from the lateral surface. Particular attention was given to maintain the exaggerated neck shaft angle. A fibular strut allograft was placed in the intramedullary cavity along the calcar to provide an additional support. The wedge was collapsed by abducting the leg on boot traction and stabilization was done using a seven-hole DHS plate. The cavity was then filled with cortico-cancellous autograft and allograft.
Standard postoperative protocol was followed. Radiological union was achieved at 3 months. The neck shaft angle was maintained at the last follow-up (Figure 3).

AP (a) and lateral (b) view of patient 1 at the end of follow-up showing persistence of corrected angulation and also persistence of the fibula strut allograft. AP: anteroposterior.
Patient 2
A 16-year-old male patient, with polyostotic FD since the age of 3 years, presented bedridden to our institution with progressive deformity in both lower limbs. Radiological investigations showed severe shepherd’s crook deformity in both proximal femurs (left > right) (Figures 4 and 5). Imaging showed varus angulation as well as anterior bowing of both proximal femora. The right side was operated first, followed by the left femur after a 5-day interval. The lesion on the left was severe enough to warrant an excision of the involved segment of proximal femur, whereas the lesion on the right required only curettage. Owing to the large lesion and thinned out cortices, the left side required a fibula strut graft inserted in a tibial shaft segment graft. Fibular strut graft alone was used in the right side. Extramedullary fixation was done using a flipped over left sided distal femur LCP on his right femur and a proximal femur LCP on the left side (Figure 6). This patient also had angular deformity in left tibia for which corrective osteotomy was performed a week after the left femoral intervention. Postoperatively patient was kept immobilized in bed for 3 months and was then readmitted for rehabilitation which included walker assisted mobilization and knee and hip mobilization exercises. The patient had an excellent radiological and functional outcome at final follow-up and was able to perform all his activities of daily living (Figure 7).

Patient 2: (a) clinical picture (patient held up with the help of attendants); (b) preoperative AP radiograph of right lower limb and (c) left lower Limb; (d) axial CT images; (e) sagittal section of right proximal femur; and (f) left proximal femur showing anterior bowing also. AP: anteroposterior; CT: computed tomography.
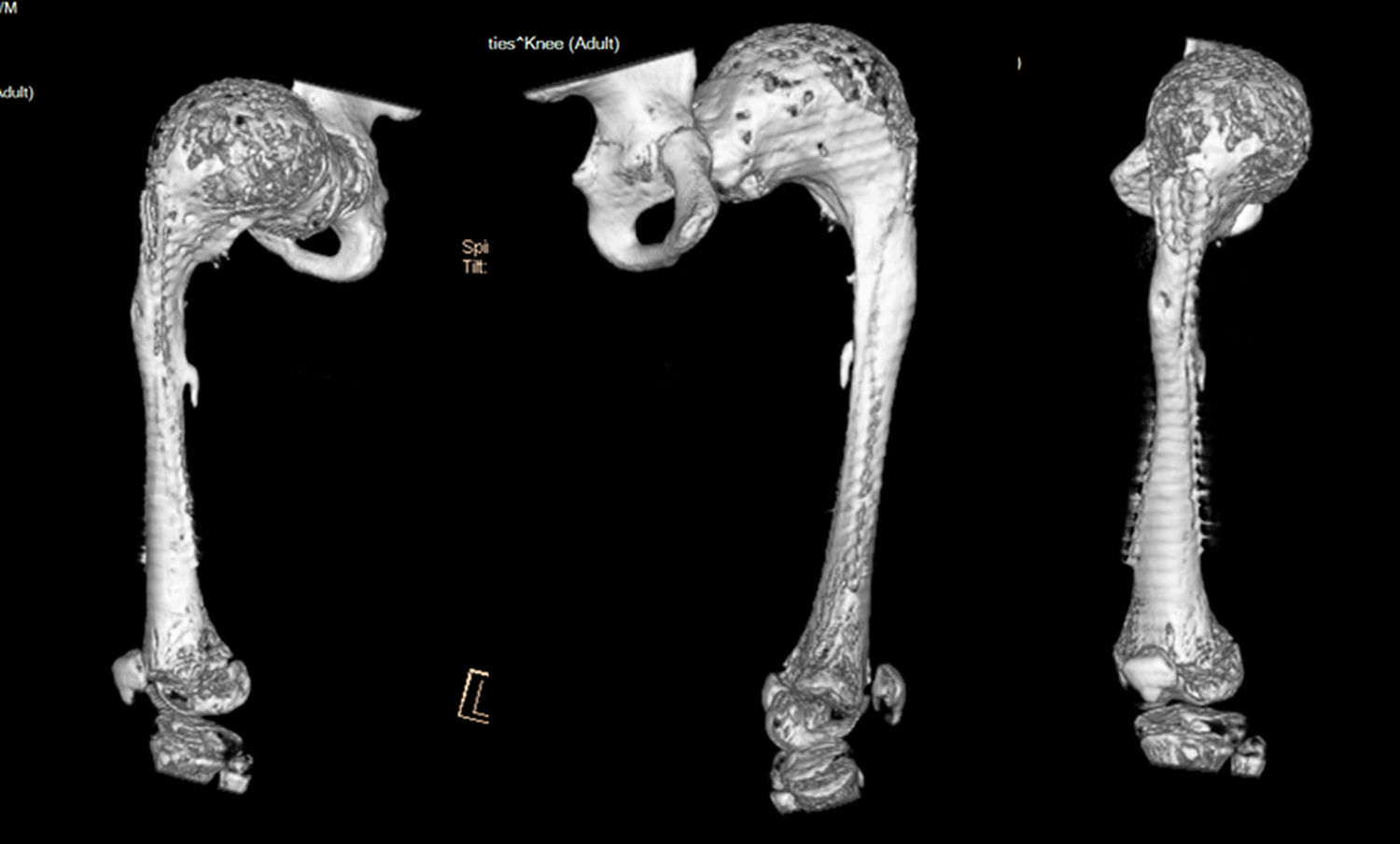
Preoperative 3D reconstructions films of left femur.

Postop AP (a) and lateral views (b) right and (c) left; note excised segment in left proximal femur and fibular strut allograft pegged into tibial segment allograft. AP: anteroposterior.

(a, b) Final follow-up radiograph showing gradual resorption of strut grafts and complete union.
Patient 3
A 12-year-old patient with polyostotic FD presented to us with a progressive deformity in his right lower limb since 7 years of age. Radiological investigations showed severe varus and significant procurvatum deformity in proximal femur (Figure 8). Surgical procedure was performed as above and stabilized with a proximal femur LCP (Figure 9). This patient was readmitted after a week with sterile serous ooze from the drain removal site which subsided with regular dressings. He had an excellent clinico-radiological outcome at final follow-up.

Preop AP (a) and lateral (b) radiographs and sagittal MRI (c) and CT (d) section showing anterior bowing of proximal femur. AP: anteroposterior; MRI: magnetic resonance imaging; CT: computed tomography.
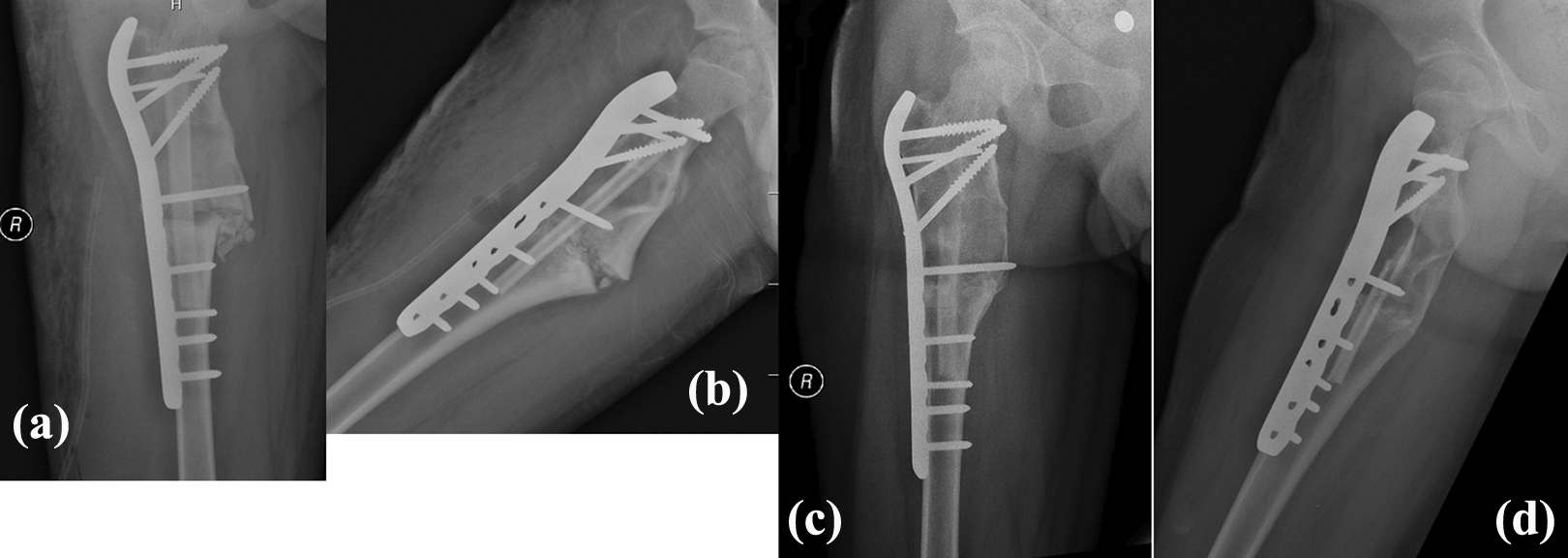
Immediate postop AP (a) and lateral (b) radiographs; final follow-up AP (c) and lateral (d) radiographs. Showing persistence of corrected angulation and also persistence of the allograft inserted. AP: anteroposterior.
Results
The neck shaft angle was corrected from an average of 91.7° to 152.1°, while the Harris Hip Score improved from an average of 59 to 95. Osteotomy site healed in all the patients uneventfully. All the patients were able to walk without any support and were able to do all routine activities without any assistance. None of the patients had a recurrence of disease or deformity or loosening/failure of the implant till the final follow-up. Readmission of patient 3 for serous ooze from the drain removal site was the only postoperative complication. Patient details, duration of follow-up, degree of deformity correction, and functional outcome have been summarized in Table 2.
Details of all the patients in the series: Demographics, angles, and outcomes.

Discussion
In FD, the indications for surgery are usually bone pains not relieved by medical management, recurrent fractures, correction of deformity and rarely for complications like malignant transformation. Correction of these severe proximal femur deformities compounded by poor bone quality is an orthopedic challenge 8
Surgical treatment with curettage and autogenous bone grafting gave very poor results and recurrence was very high in children. 7 Guille et al. have observed that the autogenous bone graft got resorbed and there was a high incidence of recurrence. 4 Recurrence of FD following curettage is more common in children than in adults. 7 Due to reports of resorption while using cancellous autograft, the trend shifted toward cortical allografts. Enneking and Gearen were the first to report use of allograft in the femoral neck lesion and vouched that it persisted without reabsorption for a long time. 3 Dicaprio and Enneking in 2005 said that cortical allogenic grafts had the least and slowest internal replacement by host bone and that allogenic grafts were biologically preferable to autogenous grafts. 7 The allografts used in our series persisted long enough till strong union was achieved and hence provided an additional support against failure of the extramedullary implant, which is a common occurrence in the subtrochanteric level. In situations where allograft facilities are not available, ipsilateral fibula can be harvested and used.
In our five-step technique, we proposed a combination of (1) clearance of the lesion with curettage or excision, (2) intramedullary stabilization with cortical strut allograft, (3) restoration of mechanical axis with valgus osteotomy, (4) extramedullary stabilization with implant, and (5) restoration of biology by augmenting the osteotomy site with cancellous autograft.
Wai et al. had a uniform treatment plan that consisted of biopsy, intralesional curettage, high-speed burring, and reconstruction with use of morcellized allograft, autograft, and a fixed-angle implant. This yielded excellent results and the authors advocated the use of a fixed-angled implant to provide a mechanical bypass for the structural lesions. 6 Li et al. treated 21 patients with shepherd’s crook deformity with valgus osteotomy and DHS fixation alone without any additional bone graft. All their patients had good clinical and radiological outcomes, although one patient had a recurrence of varus and eventually sustained a pathological fracture. 8 The addition of an intramedullary strut allograft in our patients minimized the risk of recurrence of deformity and pathological fractures.
We noted that while attempting to close the osteotomy site, particularly in large lesions with severe deformity, there was often collapse/comminution of the proximal fragment or pull-out of the implant owing to the impaired hold in the pathological bone. By securing purchase of the proximal screws of the implant in the intramedullary strut allograft, we ensured a strong hold in the proximal fragment, thereby enabling it to be adducted without collapse or implant failure. This was done in both the limbs of patient 2 (Figure 7) and in patient 3 (Figure 9) and was particularly helpful during collapse of the osteotomy site.
Several authors recommended overcorrection of the neck shaft angle to 160°–170° to overcome the varus vector and minimize the rate of recurrence of deformity. 4,5 All our patients underwent valgus overcorrection for their deformities. We feel that this along with the intramedullary strut reduced chances of recurrence.
Extramedullary implants like proximal femur LCP, flipped over distal femur LCP of the opposite side (Figure 6 right lower limb) or DHS system were used in our study. We preferred LCPs for large lesions and severe shepherd’s crook deformities, owing to their superior hold in weakened bone and additional purchase in the intramedullary strut which provided added stability. Many authors have also reported favorable outcomes following correction of proximal femur deformities and stabilization with intramedullary devices. 9,10,11 Several authors recommended stabilization with intramedullary nails and cross pinning. 12
The uniqueness of this technique can be attributed to the usage of autografts as well as a strut allograft and stabilization with an extramedullary implant. All our patients had excellent union in follow-up radiographs and excellent functional outcome as per the Harris Hip Score (barring one postoperative complication in patient 3). The major limitation of our study however is the limited number of patients. To the best of our knowledge, there has not been any prior use of a five-step technique for reconstruction of shepherd’s crook deformity. We recommend the use of this technique particularly in polyostotic and severe forms of the disease wherein there is impaired implant purchase in the weakened pathological bone, deficient lateral wall, and wide medullary canal.
Conclusion
Proximal femur FD is a difficult orthopedic problem. With this novel five-step technique, we achieved clearance of the lesion, restoration of mechanics, stability, and enhanced biology, thereby ensuring high success rates.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Ganga Orthopaedic Research and Education Foundation. The funding was to the institution and not directly paid to any author.
