Abstract
Purpose:
To determine the expression and distribution of bone morphogenetic protein (BMP)-7 and related molecules during peripheral nerve regeneration and to assess whether administration of parathyroid hormone (PTH) drug (1-34) potentiates the intrinsic upregulation of BMP-7/Smad signaling.
Methods:
The rat sciatic nerves were crushed with an aneurysm clip resulting in axonal degeneration. In the normal nerve, and at 1, 2, 4, and 8 weeks after injury, BMP-7, BMP receptors, p-Smad 1/5/8, and Noggin, the endogenous BMP antagonist, were evaluated. Additionally, the distribution of BMP-7 was assessed by fluorescent double immunostaining. In vitro studies were also performed to examine the effect of BMP-7 and PTH (1-34) administration on rat Schwann cells (SCs).
Results:
Aneurysm clip made reliable animal model of the nerve injury with recovery at 8 weeks after the injury. BMP-7/Smad protein and mRNA were significantly upregulated on axon-SCs units at 1 week after injury, and this upregulated expression was maintained for 4 weeks. Besides, significant upregulation of Noggin’s expression was observed on axon-SCs units at 2 weeks after injury. Moreover, fluorescent double immunostaining showed co-localization between expression of BMP-7 and p75NTR during axonal regeneration. In the in vitro study, administration of BMP-7 induced significant proliferation of SCs. Application of PTH (1-34) upregulated BMP-7 on SCs.
Discussion/conclusion:
BMPs were reported to be involved in protection and recovery after injury as well as in neurogenesis. Our current study showed that BMP/Smad signaling molecules were upregulated on dedifferentiated SCs after peripheral nerve injury and that administration of BMP-7 increased SC viability in vitro. These results suggested that axonal regeneration could be induced via upregulation of endogenous BMP-7 on SCs by PTH (1-34) administration.
Keywords
Introduction
In contrast to the central nervous system, peripheral nerves can regenerate after injury because of a permissive environment and activation of the intrinsic growth capacity of neurons. 1 In particular, Schwann cells (SCs) are well-known key players in repair of peripheral nerves. After nerve injury, SCs undergo dedifferentiation and proliferation, and Bungner’s bands that form at injury sites result in a permissive environment for axon regeneration and remyelination, followed by release of nerve growth factors, cell adhesion molecules, and extracellular matrix proteins. 2 –4 In fact, cell therapy with SCs is an effective method for improving nerve recovery. 5
Application of neurotrophic factors is important for the induction and effective formation of a scaffold during axonal regeneration as reported by many researchers using various neurotrophic factors such as nerve growth factor and glial cell-derived neurotrophic factor. 6 –8 Bone morphogenetic proteins (BMPs), which are members of the transforming growth factor-β subfamily, were initially identified by their ability to promote bone formation. 9 Recent cumulative evidence suggests that BMPs play important roles in induction of regeneration of various tissues including bone, tendon, and cartilage. 10,11 Development of the central nervous system is also controlled by BMPs. 12,13 Thus, BMPs are now considered multifunctional cytokines that possess neurotrophic functions. 14,15
In the central nervous system, BMP ligands and their receptor subunits are expressed throughout development, and among other functions, they help regulate cellular proliferation, survival, and lineage commitment. 16 Moreover, several authors reported that application of BMP-7 reduces the brain infarction volume and mortality in rats. 17,18 BMP-7 also enhances recovery of sensorimotor function in impaired limbs, decreases body asymmetry, and increases locomotor activity. 19,20 Based on these findings, we hypothesized that BMP-7 may be an inductive factor for axonal regeneration after peripheral nerve injury.
However, it is difficult to locally apply BMP-7 in clinical setting, since corrective site of nerve injury could be frequently undiagnosed, especially in axonotmesis, and we have not established the effective system for drug delivery in clinical use. In addition, application of high dose of BMP-7 for effective stimulation to peripheral nerve would be worried about the bone formation at applied site owing to their ability of osteoinduction. Therefore, we focused on recombinant human parathyroid hormone (PTH) drug, which are clinically used for the patients with osteoporosis, because one of the mechanisms underlying the effects of the PTH drug was shown to be mediated through transduction of the BMP/pSmad1/5/8 signaling pathway, although the precise mechanisms have remained obscure on signaling pathway. 21 –24 The PTH receptor is also expressed on SCs, 18,19,25 indicated that the PTH drug could induce intracellular signaling by binding to PTH receptors.
Few reports have investigated the expression, localization, and role of BMPs during peripheral nerve injury. Therefore, the purposes of this study are (1) to evaluate the expression of BMP-7 and related molecules in normal and injured nerves, (2) to identify where BMP-7 was expressed in peripheral nerves and whether application of BMP-7 potentiates the inductive ability of SCs in vitro, and (3) if so, to determine if administration of PTH (1-34) can upregulate intrinsic BMP-7 in SCs.
Materials and methods
Subjects
Forty 8-week-old female Sprague-Dawley rats weighing about 200 g (SLC, Hamamatsu, Japan) were used. All experimental procedures were carried out according to a protocol approved by the animal research committee of Mie University. The animals were maintained under a 12-h light/dark cycle with food and water available ad libitum in the animal center at Mie University.
Animal models
All rats were deeply anesthetized with an intramuscular injection of a combination of ketamine 100 mg/kg and xylazine 10 mg/kg. Under deep anesthesia, a longitudinal cutaneous incision was made on right hind limb, and the sciatic nerve was exposed by separation from the gluteal muscle. The sciatic nerve was then crushed for 5 min using an aneurysm clip (150N; Aesculap, Melsungen, Germany) at the level of the sciatic notch. 26 The wound was closed in anatomical layers using 5-0 nylon. The nerves were reexposed and harvested at 1, 2, 4, and 8 weeks after surgery under deep anesthesia with ketamine and xylazine. Furthermore, normal sciatic nerves were harvested according to the surgical method as abovementioned.
Functional assessment
Before the harvesting of the sciatic nerves for immunohistochemical analysis, functional evaluation of the sciatic nerve regeneration was expressed by the sciatic functional index (SFI) in both injured and uninjured sites. Hind foot prints ware obtained on paper strips. The parameters measured in the footprints were print length, toe spread (TS), and intermediate TS, both in the normal and the experimental mice at 1, 2, 4, and 8 weeks at the nerve injury. The SFI was calculated as follows: SFI = −38.3 × (EPL − NPL)/NPL + 109.5 × (ETS − NTS)/NTS + 13.3 × (EIT − NIT)/NIT − 8.8 as follows: where: EPL, experimental print length from heel to the longest toe; NPL, normal print length from heel to the longest toe; ETS, experimental total spread, or the transverse distance between the 1st and the 5th toes; NTS, normal total spread, or the transverse distance between the 1st and the 5th toes; EIT, experimental intermediate toes, or the transverse distance between the 2nd and 4th toes; NTT, normal IT: intermediate toes, or the transverse distance between the 2nd and 4th toes. 27
Immunohistochemical analysis
The specimens were embedded in paraffin, and longitudinal, 4-µm thick sections were cut on a microtome. Specimens were dewaxed in xylene and rehydrated in graded ethanol (99% to 70% (v/v)) in distilled water. Endogenous peroxidase activity was quenched by 30-min incubation in 0.3% (v/v) hydrogen peroxide in 99% methanol. Heat-induced antigen retrieval was performed in 10-mM citrate buffer pH 6.0 using a pressure cooker (Delicio 6L; T-FAL, Rumily, France). The sections were then left to cool at room temperature in the citrate buffer. Nonspecific staining was blocked by incubating the sections in a solution of 1% bovine serum albumin for 20 min at room temperature. The bovine serum albumin was then drained off, and specimens were incubated at room temperature overnight in each antibody (1: 50). The primary antibodies used were rabbit anti-neurofilament (Nippon Chemicon, Osaki, Japan), goat anti-BMP-7, BMP receptor-1A, BMP receptor-1B, BMP receptor-2, Noggin (Santa Cruz Biotechnology, Santa Cruz, CA, USA), and rabbit anti-p-Smad1/5/8 (Nippon Chemicon) which is downstream of BMP signaling (n = 3 animals for each test). Bound primary antibodies were detected by applying secondary anti-goat Ig antibodies and anti-rabbit Ig antibodies conjugated to horseradish peroxidase (1:100; Dako Japan, Kyoto, Japan) for 1 h at room temperature. Bound antibodies were visualized by reacting with 3-3′ diaminobenzidine. Between incubation steps, sections were immersion washed by dipping (3 × 5 min) in phosphate bufferd saline to eliminate excess nonbound antibody or reagent. Sections were counterstained with hematoxylin. Control sections were not incubated with primary antibodies. Images were obtained with a microscope (BX50 OLYMPUS, Tokyo, Japan) equipped with a digital camera. In all cases, control sections showed no evidence of staining or color reaction under microscopic examination. The percent of the positively stained area was calculated by image analysis software (Lumina Vision, version 1.11; Minami Shoji Co., Fukui, Japan).
Fluorescent double immunostaining
The specimens were embedded in paraffin, and longitudinal, 4-µm thick sections were cut on a microtome as abovementioned. The secondary antibody for visualization of BMP-7 was conjugated to Alexa Fluor® 488 (green; Abcam, Tokyo, Japan). The SCs markers, rabbit anti-S100 (Dako Japan) and rabbit anti-p75NTR (Nippon Chemicon), were visualized with secondary antibodies conjugated to Alexa Fluor® 546 (red; Abcam).
Western blot analysis
Frozen nerves were homogenized in Sodium dodecyl sulfate sample buffer. Equal amounts of protein were loaded onto 15% gradient precast gels, and separated proteins were transferred onto Poly Vinylidee DiFluoride membranes by electro-transfer. The membranes were then incubated with primary antibodies against BMP-7, p-Smad1/5/8, Noggin, and β-actin (Abcam). We detected the target proteins with the Enhanced chemiluminescence system. Each band was measured and semiquantitatively analyzed using Image J 1.47 (National Institutes of Health, Bethesda, MD, USA).
Real-time polymerase chain reaction
Total RNA was isolated from homogenized frozen nerve using the mirVANA Isolation kit (Ambion, Naugatuck, CT, USA). The RNA was treated with DNase and converted to cDNA using the 1st Strand cDNA Synthesis Kit for real-time polymerase chain reaction (RT-PCR) (AMV; Roche, Basel, Switzerland). To quantify the expression level of BMP-7, Smad-1, and GAPDH, RT-PCR was performed using ABI PRISM 7000 Sequence Detection System (Applied Biosystems, Foster City, CA, USA). TaqMan PCR was performed using TaqMan Universal PCR Master Mix (Applied Biosystems). TaqMan gene expression assay primers were used to detect rat BMP-7 (Assay ID. Rn01528889_m1; Applied Biosystems) and rat GAPDH (Assay ID. Rn99999916_s1; Applied Biosystems; n = 5 for each test). The thermal cycling conditions were 50°C for 2 min, 95°C for 10 min, 40 cycles of 95°C for 15 s, and 60°C for 1 min. GAPDH was used as an endogenous reference gene for normalization. Expression levels of each gene were divided by GAPDH expression levels.
SC culture
Rat SCs were purchased from ScienCell Research Laboratories (Carlsbad, CA, USA; catalog #R1700), cultured in SC medium (ScienCell Research Laboratories), and maintained in a 37°C incubator with 5% carbon dioxide.
MTS cell proliferation assay
Cells were seeded at a density of 5000/ml in 100-μl medium, and recombinant BMP-7 (R&D Systems, Minneapolis, MN, USA) was added at concentrations of 0, 10, 50, or 100 ng/ml (n = 5 for each test) as previously reported. 28,29 Cells were incubated at 37°C for 24 h, and proliferation was analyzed according to the manufacturer’s instructions (Promega Corporation, Madison, WI, USA). Twenty microliters of MTS reagent was added directly to the wells and incubated for 3 h. Absorbance was measured at 492 nm on a Multiscan JX microplate reader (Thermo Labsystems, Waltham, MA, USA). The results are represented as the percentage of relative fluorescence units compared with the control.
Application of PTH (1-34)
The drug was provided by Asahikasei Pharmacy Corporation (Tokyo, Japan). As mentioned earlier, prepared SCs were treated with 0, 10−8, or 10−7 M PTH (1-34), of which concentrations were determined in reference to a previous report. 30 Cell proliferation was evaluated with the MTS assay (n = 5 for each test). Additionally, expression of BMP-7 was analyzed by fluorescent double immunostaining and Western blotting.
Statistical analysis
Data were analyzed using the Kruskal–Wallis or Mann–Whitney U test among groups. Values of p < 0.05 were considered statistically significant.
Results
Aneurism clip was successfully applied in the animal model of the nerve injury with functional recovery and axonal extension at 8 weeks after the injury.
In normal rats, SFI levels were almost same between the injured and the uninjured sites. At 1 week, the mean SFI levels was −99.6 in the injured site, which was significantly lower compared to −3.35 in uninjured site (Figure 1(b)). Immunohistochemical analysis concurrently showed that immunolabeling of neurofilament was drastically reduced at the injured site (Figure 1(a)). At 2 weeks after the nerve injury, the mean level of SFI was still low, while expression of neurofilament was upregulated and surrounded by a large number of cells at the injured site. The significant reduction of SFI levels was maintained for 4 weeks after the nerve injury, although the improvement of SFI levels and axonal extension was observed at that time. In addition, recovery of SFI levels and axonal extension were ascertained at 8 weeks after the nerve injury using aneurysm clip (Figure 1(a) and (b)).
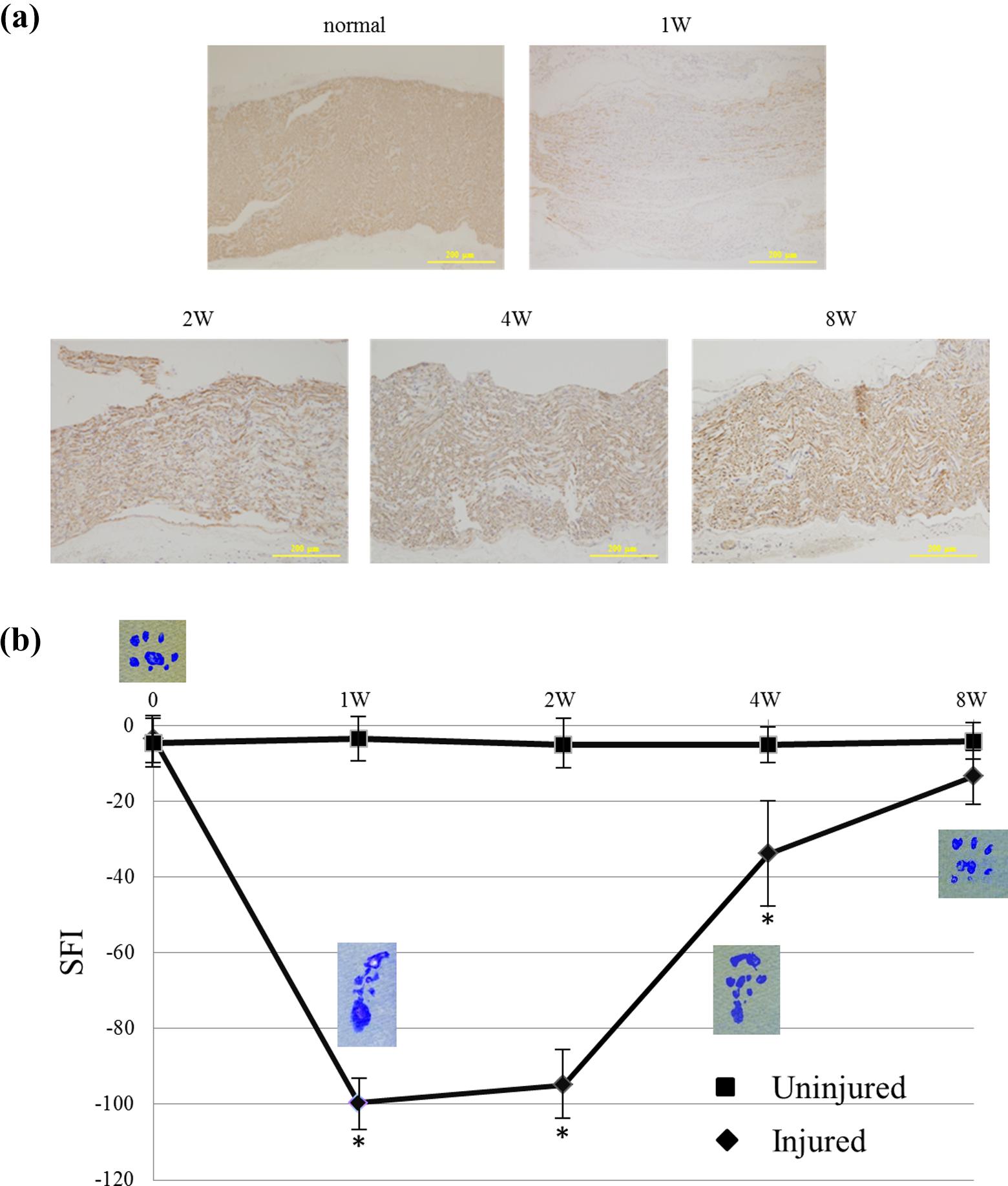
(a) Immunolabeling for neurofilament in the injured site of rat sciatic nerve. Expression of neurofilament was predominantly reduced in the injured site at 1 week. At 2 weeks later, the expression was recovered in the injured site. The axonal regeneration with proper cellularity of Schwann cells was likely to be achieved around the axons at 8 weeks. (b) Levels of SFI in the hind limbs with and without nerve injury using aneurysm clip. SFI levels were significantly lower in the injured hind limbs than the uninjured hind limbs at 1 week. The recovery of the SFI levels was observed at 4 weeks in the injured hind limbs, although the levels were still significantly low compared to that in the uninjured hind limbs. The levels of SFI were almost recovered at 8 weeks after the injury of sciatic nerves using aneurysm clip. SFI: sciatic functional index.
BMP-7 and related molecules were significantly upregulated at the site of injury at both the mRNA and protein levels, whereas these molecules were expressed at low or undetectable levels in normal nerve.
BMP-7, BMP receptors, p-Smad 1/5/8, and Noggin were weakly expressed in the axon-SC units of normal sciatic nerve (Figure 2(a)). The mRNA of these BMP-7-related factors was also present in the extract from normal sciatic nerves (Figure 3(b)). Following nerve injury, expression of BMP-7, BMP receptors, and p-Smad 1/5/8 was clearly upregulated at axon-SC units near the injury (Figure 2(a) and (b)). Western blotting and PCR demonstrated that the expression of these molecules except Noggin was significantly upregulated from the distal site to the injury at 1 week after nerve injury (Figure 3(a) and (b)). Four weeks after injury, the expression of these molecules was downregulated in an inverse proportion to the axonal extension and functional recovery. On the other hand, expression of Noggin, which is an endogenous antagonist of BMPs, was significantly upregulated at 2 weeks after the injury and was maintained at high expression until 8 weeks after injury.

Immunolabeling for BMP-7, BMP receptors, and p-Smad 1/5/8 in the injured site of rat sciatic nerve. BMP-7, BMP receptors, and p-Smad 1/5/8 were weakly expressed in the axon-Schwann cell units of the normal sciatic nerve. Nerve injury induced significant upregulation of expression of these molecules from the distal site to the injured site. Upregulation of these molecules was maintained for 2 weeks. On the other hand, Noggin, which is a BMP antagonist, was highly expressed at 2 weeks after injury, and this expression was maintained up to 8 weeks. (a) Immunolabeling for BMP-7, BMP receptors, p-Smad1/5/8, and Noggin in the normal and injured rat sciatic nerve (n = 3). (b) The percentage of the area showing expression was calculated as the sum total of stained areas divided by the total area using image analysis software (Lumina Vision, version 1.11). Error bars represent the mean ± SE. *p < 0.01 (vs. normal). BMP: bone morphogenetic protein.

(a) Western blot analysis of BMP-7, p-Smad1/5/8, and Noggin in the normal and injured rat sciatic nerve. Quantification graphs reflected the intensity of staining of BMP-7, p-Smad1/5/8, and Noggin normalized to β-actin at each time point using Image J. Error bars represent the mean ± SE. *p < 0.01 (vs. normal). (b) Quantitative RT-PCR analysis of BMP-7 and Smad1 expression in the normal and injured sciatic nerve (n = 5). BMP-7 mRNA increased significantly 1 week after nerve injury. Error bars represent the mean ± SE. *p < 0.01 (vs. normal). BMP: bone morphogenetic protein; RT-PCR: real-time polymerase chain reaction.
BMP-7 was expressed on dedifferentiated SCs and promoted proliferation of SCs.
Expression of BMP-7 was shown on the cells around the axon by immunohistochemical analysis. In addition, fluorescent double immunostaining showed that most BMP-7 expression appeared to be distributed in dedifferentiated SCs because expression of BMP-7 overlapped that of p75NTR, a marker of dedifferentiated SCs. Figure 4(a) clearly shows that although only a portion of S100-positive cells expressed BMP-7, p75NTR was extensively co-localized with BMP-7 following nerve injury, especially at 1 week after the injury, when dedifferentiated SCs were longitudinally arranged for axonal regeneration. Additionally, an in vitro study demonstrated that cell viability of SCs was significantly increased by BMP-7 administration in a dose-dependent manner at 24 h (Figure 4(b)).
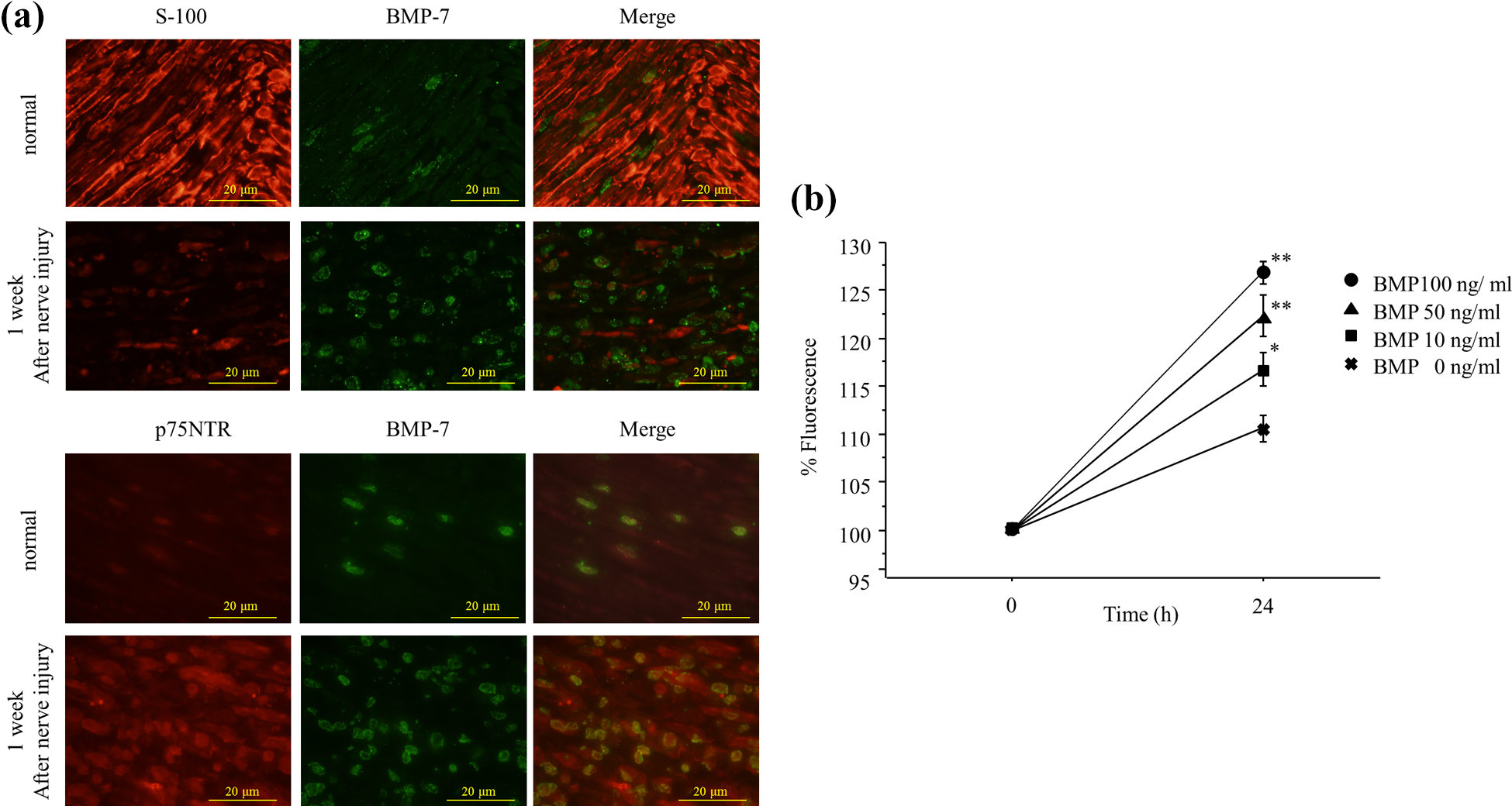
(a) Double immunofluorescence analysis of localization of BMP-7 1-week after nerve injury. Double-label immunohistochemistry was performed on normal and injured nerves using an antibody for BMP-7 (green), the Schwann cell marker S-100 (red), and the dedifferentiated Schwann cell marker p75NTR (red). Areas of BMP-7 expression overlapped substantially with p75NTR, suggesting that BMP-7/Smad signaling may be involved in the proliferation of Schwann cell. (b) Viability and proliferation of Schwann cells after a 24-h exposure to BMP-7 as determined with the MTS assay. Cell viability of Schwann cell was significantly increased following administration of BMP-7 in a dose-dependent manner at 24 h. Error bars represent the mean ± SE (n = 5). *p < 0.05; **p < 0.01 (vs. normal). BMP: bone morphogenetic protein.
Administration of PTH (1-34) induced significant upregulation of expression of BMP-7 on SCs as well as significant proliferation of SCs.
We treated SCs with PTH (1-34) to determine whether this drug can upregulate endogenous BMP-7 expression on SCs. We observed significant proliferation of SCs following treatment with 10−8 M and 10−7 M PTH (1-34) compared to saline (Figure 5(c)). Additionally, administration of PTH (1-34) clearly upregulated BMP-7 in the cytoplasm of SCs, whereas BMP-7 was not expressed on SCs in the absence of PTH (1-34) treatment, as shown with fluorescent immunostaining (Figure 5(a)). The upregulation of BMP-7 expression was also shown with Western blotting. Semiquantitative analysis of the expression showed significant upregulation (Figure 5(b)).

(a) Immunofluorescence staining for S100 (red) and BMP-7 (green) in normal and PTH (1-34) treated Schwann cells. The expression of BMP-7 was increased in the cytoplasm of Schwann cells after PTH (1-34) treatment. (b) Western blot analysis of BMP-7 in Schwann cells after PTH (1-34) treatment for 24 h. Quantification graphs reflect the intensity of staining of BMP-7 normalized to β-actin using Image J. The expression of BMP-7 was increased after PTH (1-34) treatment. **p < 0.01 (vs. normal). (c) Viability and proliferation of Schwann cells after exposure to PTH (1-34) for 24 h as determined with the MTS assay. Cell viability of Schwann cell was significantly increased by administration of PTH (1-34) in a concentration-dependent manner. Error bars represent the mean ± SE (n = 5). *p < 0.05; **p < 0.01 (vs. normal). BMP: bone morphogenetic protein; PTH: parathyroid hormone.
Discussion
BMPs bind to BMP type I and type II serine/threonine kinase receptors on the cytoplasmic membrane. 31 Formation of the complex subsequently activates intracellular Smad proteins, which are key mediators of BMP signaling. 32 The present study demonstrated minimal expression of BMP-7 as well as the receptors in normal nerve and strong upregulation at 1 week after nerve injury. At this time, various conditions become favorable for induction of axonal regeneration such as secretion of neurotrophic factors and formation of the basement membrane. 33,34 At the same time, we observed significant upregulation of p-Smad 1/5/8 expression, which is downstream of BMP receptor binding, in the injured nerve. By contrast, expression of Noggin remained low, similar to the normal condition, at 1 week after nerve injury, and was then upregulated to a peak 4 weeks after nerve injury. These findings suggested that BMP signals play important roles in axonal regeneration at a relatively early phase.
Reexpression is a phenomenon in which proteins expressed in the embryo are again expressed and function in tissue regeneration following inflammation, tissue repair/regeneration, or tumor invasion. BMP ligands and their receptor units are also abundantly expressed in the embryonic central nervous system during neurogenesis and help regulate cellular proliferation, survival, differentiation, apoptosis, and lineage commitment. 16,35 Furthermore, BMPs including BMP-7 can be reexpressed after injury and are involved in functional recovery of the central nervous system. 36 This potential may be useful for treatment by gene introduction. 14 However, where and how BMPs were reexpressed and the time course of changes in expression were unknown in the peripheral nervous system after nerve injury. Our previous study demonstrated that BMP-7 and the receptors were reexpressed in the axon-SC unit around the injured site after injury of the rat sciatic nerve, although these proteins were expressed at low or undetectable levels in normal nerves. 37 Semiquantitative analysis in our current study also showed significant upregulation of expression of BMP-7, BMP receptors, and p-Smad 1/5/8 at the protein and mRNA levels after nerve injury of rats.
Furthermore, the distribution of BMP-7 and related factors has not been previously demonstrated in vivo in the peripheral nervous system. We speculated that these molecules could be expressed and function on SCs, because the expression of BMP-7 was predominantly upregulated and longitudinally arranged at an early phase after nerve injury in this immunohistochemical analysis. The SCs make an important contribution to axonal regeneration and remyelination after nerve injury in which the glial cells undergo dedifferentiation and proliferation, followed by formation of Bungner’s bands at injury sites, resulting in a permissive environment for axon regeneration and remyelination. However, the anti-S-100 antibody did not immunolabel the cells expressing BMP-7 in either normal or injured nerves. Further analysis was performed using the p75NTR antibody as a marker of dedifferentiated SCs to detect which cells expressed BMP-7. 2,38 BMP-7 was clearly expressed on p75NTR-positive cells, especially after nerve injury. In fact, dedifferentiated SCs reexpress many genes that are expressed during embryonic development and secrete growth factors such as nerve growth factor, hepatocyte growth factor, vascular endothelial growth factor, and brain-derived neurotrophic factor. 19 In addition, SCs in vitro showed dose-dependent, significant proliferation following administration of BMP-7 at 24 h. Thus, BMP-7 is expected to function for proliferation of SCs as either an autocrine or paracrine neurotrophic factor.
We, therefore, hypothesized that administration of BMP-7 could be useful for induction of axonal regeneration as a new treatment strategy for nerve injury. Several previous studies also showed the possibility of BMP administration for nerve recovery. In the central nervous system, treatment with BMP-7 before generalized hypoxia or ischemia reduces the brain infarction volume and mortality in rats. 17,18 Additionally, administration of BMP-7 after focal cerebral stroke enhances recovery of sensorimotor function in impaired limbs, decreases body asymmetry, and increases locomotor activity. 19,20 Likewise, in the peripheral nervous system, administration of BMP-2 induces thicker and denser morphology of the facial nerve during recovery after injury. 9 However, we were concerned about local application of BMPs to an injured peripheral nerve in the clinical setting, because BMPs have the capability of bone conduction. Therefore, PTH (1-34), which is clinically used to increase bone mass and promotes differentiation of osteoblasts through BMP signal activation, 39,40 was tested as a candidate to upregulate endogenous expression of BMP-7 on SCs as well as osteoblasts. Additionally, the PTH receptor is expressed on SCs, 25 and teriparatide could induce intracellular signaling by binding to PTH receptors. The present study demonstrated a significant increase in proliferation of SCs and significant upregulation of BMP-7 expression on SCs with administration of PTH (1-34). The results provided a possibility of PTH (1-34) to be a realistic drug for the peripheral nerve injury via endogenous induction of reexpression of BMP-7 in SCs, since PTH (1-34) has been commonly used in the clinical manner for the patients with osteoporosis.
However, we performed only in vitro study on SCs for evaluation of effectiveness of PTH (1-34), in which the drug could be assessed in the local treatment. However, PTH (1-34) was systemically administered in clinical use. This is a major limitation in our study. Therefore, the further investigation is required for assessment whether PTH (1-34) is usefulness for the injury of the peripheral nerve in in vivo study for evaluation of axonal regeneration using animal model, including immunohistochemical analysis, functional analysis, and electromyogram analysis.
Conclusion
This study showed that BMP-7, their receptors, and p-Smad1/5/8 were minimally expressed on axon-SCs unit of normal sciatic nerves of rat. After the nerve injury, the expressions of the BMP signaling molecules were significantly upregulated in both the protein and mRNA levels at 1 week later. By contrast, significant upregulation of Noggin’s expression could not be detected unit until 2 weeks after the nerve injury. Furthermore, fluorescent double immunostaining showed that most of BMP-7 were observed on the cells with expression of p75NTR, suggested that BMP-7 was distributed on the dedifferentiated SCs after the nerve injury. Besides, in vitro study showed a significant proliferation of SCs with application of BMP-7 in a dose-dependent manner. In addition, administration of PTH (1-34) induced significant upregulation of BMP-7’s expression on SCs as well as significant proliferation of SCs. These results provided a possibility of the therapeutic agent of PTH (1-34) through BMP/Smad signaling pathway for the peripheral nerve injury; however, this requires further investigation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
