Abstract
Purpose:
Oxidative stress is closely associated with the pathogenesis of nontraumatic osteonecrosis of the femoral head (ONFH). This study aimed to determine whether the serum levels of antioxidant nutrients were decreased in patients with ONFH.
Methods:
We analyzed the serum levels of antioxidant nutrients in 39 patients with ONFH (ONFH group) and 78 age- and gender-matched healthy people (control group) who voluntarily participated in the Yakumo study, which is a comprehensive health examination program. We measured and compared the serum levels of α-tocopherol (vitamin E) and total carotenoids, including zeaxanthin/lutein, β-cryptoxanthin, lycopene, α-carotene, and β-carotene, in the ONFH and control groups using high-performance liquid chromatography.
Results:
The mean serum levels of total carotenoids were significantly lower in the ONFH group than in the control group (2.36 ± 1.26 and 3.79 ± 2.36 µmol/l, respectively, p < 0.001). However, no significant difference was found in α-tocopherol between the two groups (26.37 ± 6.90 µmol/l in the ONFH group and 26.24 ± 6.28 µmol/l in the control group, p = 0.920). Among each carotenoid, the serum levels of zeaxanthin/lutein, lycopene, and β-carotene were significantly lower in the ONFH group than in the control group (p < 0.001).
Conclusions:
The serum levels of carotenoids were lower in patients with ONFH than in healthy, community-living people. This result suggests that carotenoids may be related to the pathogenesis of ONFH.
Introduction
Nontraumatic osteonecrosis of the femoral head (ONFH) typically occurs in young adults. 1 It often leads to progressive collapse of the femoral head and secondary osteoarthritis, resulting in pain and functional disorder of the hip. Previous studies suggested that ischemia is the cause of ONFH, but the mechanisms have not been clarified in detail. 2,3 Various mechanisms of ONFH, such as coagulation abnormalities, hyperlipidaemia, oxidative stress, vascular endothelial dysfunction, vasculitis, and vasospasm, have been proposed. 2 –5
ONFH frequently occurs in patients who receive high dose of corticosteroid or drink excessive amounts of alcohol. 5 –9 Regardless of related risk factors, patients with ONFH show similar clinical, radiographical, and pathological findings, suggesting a common pathogenic pathway or causative factor. 10 Previous studies showed that corticosteroid or alcohol administration induces oxidative stress and results in harmful effects on several organs, including bone tissue. 11 –14 Thus, ONFH patients with corticosteroid therapy or alcohol abuse are speculated to be exposed to excessive oxidative stress.
Several studies using experimental ONFH models have reported that oxidative stress is closely related to the pathogenesis of ONFH. Ichiseki et al. 4 demonstrated that oxidative stress is by itself sufficient to induce ONFH in rats. In addition, Mikami et al. 15 showed that vitamin E, known as a fat-soluble substance that has potent antioxidant properties, prevents the occurrence of steroid-induced ONFH by suppressing oxidative stress in rabbits. Therefore, inhibition of oxidative stress is one of the major therapeutic targets to prevent the occurrence of ONFH. 16 –20
Carotenoids, which are associated with decreased risk of cardiovascular diseases and cancers, are also attracting attention as antioxidant nutrients. 21,22 Carotenoids are lipid-soluble natural pigments synthesized by plants, bacteria, and some algae. They are responsible for the yellow, red, and orange colors in food. In 1968, Foote and Denny 23 reported the strong scavenging activity for singlet oxygen of β-carotene. Subsequently, many studies about the antioxidative effect of carotenoids have been reported. 24 Thus, carotenoids, as well as vitamin E, are expected to have a preventive effect on the occurrence of ONFH. However, the association between carotenoids and ONFH has not yet been surveyed.
An inverse correlation between the serum levels of antioxidant nutrients and markers of oxidative stress is found, and lower serum levels of antioxidant nutrients increase the risk of diseases related to oxidative stress. 24 –27 However, to our knowledge, no report has evaluated these serum levels in patients with ONFH. Therefore, we attempted to measure the serum levels of antioxidant nutrients to evaluate the oxidative status of patients with ONFH. We hypothesized that patients with ONFH were exposed to excessive oxidative stress and their serum levels of antioxidant nutrients were lower than those of healthy people. This study aimed to measure and compare the serum levels of total carotenoids and vitamin E in patients with ONFH and healthy, community-living people to identify antioxidant nutrients that may be related to the pathogenesis of ONFH and could potentially prevent the occurrence of ONFH.
Materials and methods
Patient selection
Thirty-nine patients with ONFH who underwent medical examinations in our hospital from August to December 2013 were enrolled in this study (ONFH group). Diagnosis of ONFH was based on clinical history, physical examination, and evaluations on radiography and magnetic resonance imaging. 28,29 There were 15 patients with alcohol-associated ONFH and 18 patients with steroid-associated ONFH. Two patients had history of both alcohol abuse and systemic steroid administration, and four patients had history of neither.
For the control group, we used the serums of 552 healthy, community-living people who participated in the Yakumo study in 2012. The Yakumo study is a comprehensive health examination program involving volunteers from Yakumo, Hokkaido, Japan. This program has been conducted every August since 1982. 30,31 The exclusion criteria of the control group were people with hip diseases and intake of supplements. Finally, 78 healthy, community-living people matched by age, gender, smoking history, habitual alcohol intake, and race (only Japanese) were selected as the control group.
The protocol of this study was approved by our institutional review board. Informed consent was obtained from each participant prior to participation.
Interview of subjects
Physical characteristics were investigated by experienced interviewers using a questionnaire and an interview in both groups. We collected their information on clinical characteristics, including age, gender, body mass index (BMI), smoking history, habitual alcohol intake, and systemic steroid administration. Various characteristics of both groups are listed in Table 1. The mean age was 52.6 years (range 37–78) in the ONFH group, and 54.7 years (range 40–78) in the control group (p = 0.288). The mean BMI was 22.3 kg/m2 (range 13.1–31.6) in the ONFH group, and 22.8 kg/m2 (range 14.0–31.5) in the control group (p = 0.455). In both groups, rates of smoking and habitual alcohol intake were 64% and 77%, respectively. There were no significant differences in duration of smoking and alcohol intake between the two groups. No one had received systemic steroid administration in the control group. Among the 20 ONFH patients with systemic steroid therapy, 12 patients (60%) received current oral corticosteroid therapy with an average of 5.4 mg (range 1–10) of prednisolone equivalent per day at the study, and 11 patients (55%) had previously received steroid pulse therapy.
Characteristics of the ONFH and control groups.a

ONFH: osteonecrosis of the femoral head; BMI: body mass index; SD: standard deviation.
aValues are shown as mean (SD) for age, BMI, duration of smoking and duration of alcohol drinking.
b p < 0.05 with χ 2 test.
Measurement of antioxidant nutrients
The serum levels of α-tocopherol (vitamin E) and carotenoids (including zeaxanthin/lutein, β-cryptoxanthin, lycopene, α-carotene, and β-carotene) were measured using high-performance liquid chromatography (HPLC) according to the method we previously reported. 30,31 Briefly, these antioxidant nutrients extracted from 200 μl of serum using n-hexane were dissolved in 100 μl of ethanol, and 20 μl of the solution was then injected into an HPLC system (Waters Corporation, Milford, Massachusetts, USA) equipped with a Nova Pak Cartridge C column (4 pm, 8 × 10 cm2; Waters Corporation). In this assay system, as the peaks of zeaxanthin and lutein overlap at the same sites, they cannot be measured separately.
Among approximately 600 carotenoids identified in nature, only six (zeaxanthin, lutein, β-cryptoxanthin, lycopene, α-carotene, and β-carotene) represent more than 95% of total carotenoids in human plasma. 32 The sum of these six carotenoids was then defined as total carotenoids in this study. Eight different molecules of vitamin E exist including α-, β-, γ-, and δ-tocopherols and α-, β-, γ-, and δ-tocotrienols in nature. Among the tocopherols and tocotrienols, α-tocopherol is mainly found in the serum 33 and has the highest biopotency. 34 Thus, the serum levels of α-tocopherol were measured in this study as the representative of vitamin E.
Statistical analysis
Student’s t-test was used to analyze numerical variables, and χ 2 test was used for categorical variables. The serum levels of carotenoids and α-tocopherol were compared between the ONFH and control groups. Subanalyses were performed in smokers or alcohol drinkers between the two groups. To assess the influences of steroid administration on the serum levels of antioxidant nutrients, patients with steroid-associated ONFH were divided according to current steroid use at the study and medical history of steroid pulse therapy. Using 19 ONFH patients without medical history of systemic steroid administration and 78 healthy controls, logistic regression analysis was carried out to determine the serum levels of antioxidants as independent factors for ONFH and estimate odds ratios (OR) after adjusting for age, gender, BMI, smoking, and alcohol habits. All analyses were performed using SPSS version 23 (SPSS Inc., Chicago, Illinois, USA). A p value <0.05 was considered to be statistically significant.
Results
The mean serum levels of total carotenoids were significantly lower in the ONFH group than in the control group (2.36 ± 1.26 µmol/l in the ONFH group and 3.79 ± 2.36 µmol/l in the control group, p < 0.001; Table 2). However, no significant difference was found in α-tocopherol between the two groups (26.37 ± 6.90 µmol/l in the ONFH group and 26.24 ± 6.28 µmol/l in the control group, p = 0.920). The serum levels of zeaxanthin/lutein, lycopene, and β-carotene were significantly lower in the ONFH group than in the control group (p < 0.001, respectively), and β-cryptoxanthin and α-carotene were almost the same levels in the two groups.
Serum levels of antioxidant nutrients in the ONFH and control groups.a
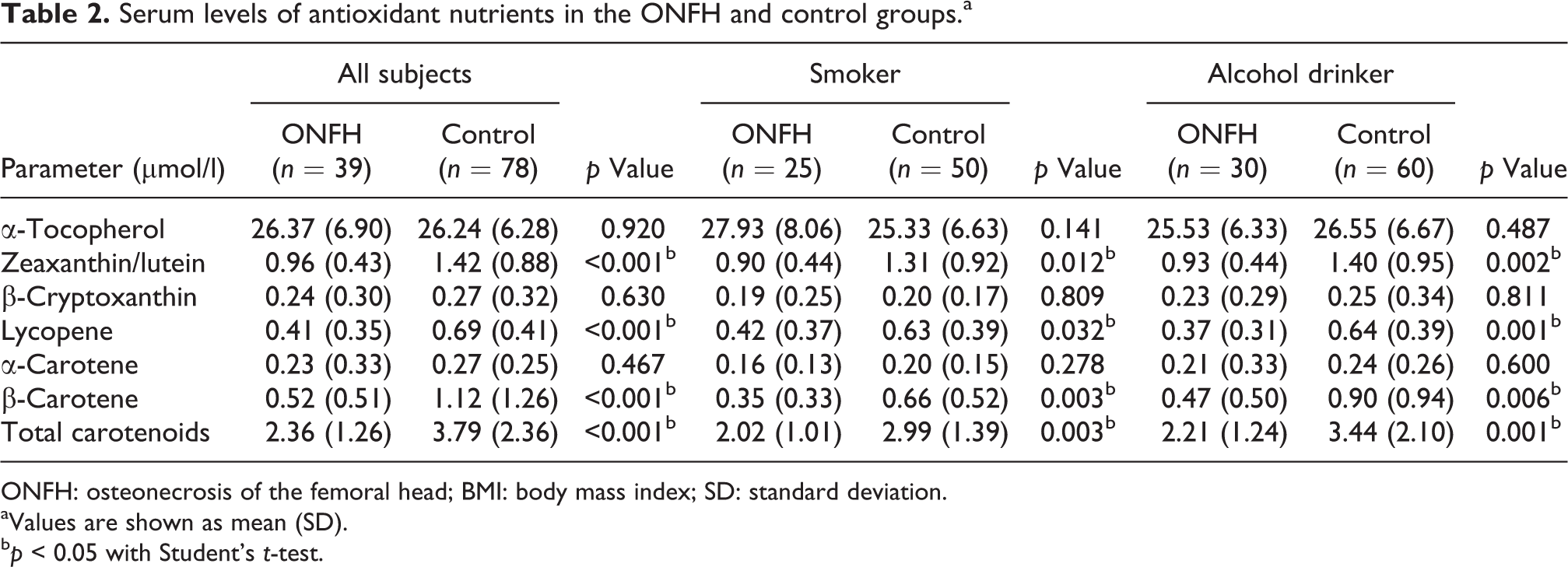
ONFH: osteonecrosis of the femoral head; BMI: body mass index; SD: standard deviation.
aValues are shown as mean (SD).
b p < 0.05 with Student’s t-test.
In the subanalyses of both smokers and alcohol drinkers, the mean serum levels of total carotenoids were significantly lower in the ONFH group than in the control group (Table 2). However, the serum levels of α-tocopherol did not decrease in the ONFH group compared with the control group in both comparisons. The serum levels of zeaxanthin/lutein, lycopene, and β-carotene were significantly lower in the ONFH group than in the control group in both smokers and alcohol drinkers.
The serum levels of both total carotenoids and α-tocopherol did not change regardless of the medical history of systemic steroid administration (Table 3). Furthermore, in steroid-associated ONFH, neither current use of steroid nor past steroid pulse therapy affected the serum levels of total carotenoids and α-tocopherol.
Serum levels of antioxidant nutrients according to medical history of steroid therapy in the ONFH group.a

ONFH: osteonecrosis of the femoral head; SD: standard deviation.
aValues are shown as mean (SD).
Logistic regression analysis revealed that a low serum level of total carotenoids was the independent risk factor for ONFH without medical history of systemic steroid administration after adjusting for age, gender, BMI, smoking, and alcohol habits (OR 2.28; 95% CI 1.32–3.92, p = 0.003; Table 4). When each carotenoid was evaluated separately, a low serum level of β-carotene was extracted as the independent risk factor (OR 13.64; 95% CI 2.34–79.65, p = 0.004). Any other factors included in logistic regression analysis were not calculated as independent risk factors for ONFH.
Adjusted ORs with 95% CI for risk of ONFH unrelated to steroid therapy by logistic regression analysis.

OR: odds ratio; CI: confidence interval; ONFH: osteonecrosis of the femoral head; BMI: body mass index.
aAdjusted for age, gender, BMI, smoking, alcohol drinking, and the serum levels of α-tocopherol and total carotenoids.
bAdjusted for age, gender, BMI, smoking, alcohol drinking, and the serum levels of α-tocopherol and each carotenoid including zeaxanthin/lutein, β-cryptoxanthin, lycopene, α-carotene, and β-carotene.
Discussion
We demonstrated that the serum levels of total carotenoids were lower in patients with ONFH than in healthy, community-living people. Endothelial dysfunction caused by oxidative stress leads to several vascular diseases. 35,36 Oxidative stress has also been proved to be closely related to the pathogenesis of ONFH in experimental animal models. 4,15 –20 Observational studies have reported that low serum levels of carotenoids are associated with increased risk of cardiovascular diseases. 25 –27 This study is the first to survey the serum levels of carotenoids in patients with ONFH. Our results have suggested that lower serum levels of carotenoids are also associated with the risk factors for ONFH.
The association between carotenoids and the pathogenesis of ONFH has not been clarified. Previous studies proposed that one of the pathogeneses of ONFH is vascular endothelial dysfunction due to inactivation of nitric oxide and induction of apoptosis of endothelial cells caused by oxidative stress. 5 In this study, the serum levels of zeaxanthin/lutein, lycopene, and β-carotene were significantly lower in patients with ONFH than in healthy people. Previous studies have proved that they have protective effects against endothelial dysfunction caused by oxidative stress. Zhu et al. 37 reported that lycopene attenuates endothelial dysfunction in rats with diabetes by reducing oxidative stress. Watarai et al. 38 showed that high serum levels of zeaxanthin/lutein and β-carotene may help to prevent elevated serum asymmetric dimethylarginine, which induces endothelial dysfunction by inhibiting endothelium nitric oxide synthase. Thus, we speculate that carotenoids are related to the prevention of ONFH via inhibiting endothelial dysfunction caused by oxidative stress.
Interestingly, significant differences were not found in the serum levels of α-tocopherol, which accounts for a large percentage of vitamin E in the human body, 27 between the ONFH and control groups in this study. This result is in contrast to those in previous studies that reported the preventive effect of vitamin E for ONFH in animal experiments. 15 –17 In the meta-analysis of randomized controlled trials, Ashor et al. 39 revealed that supplementation of vitamin E has beneficial effects on endothelial function in those with lower serum concentrations of vitamin E but has no effects in those with a serum vitamin E concentration of more than 20 µmol/l. Other studies also support the idea that individual serum levels of antioxidant nutrients may determine the extent of the influence of their supplementation on endothelial dysfunction. 40,41 Considering that the mean serum level of α-tocopherol in patients with ONFH was 26.37 µmol/l in this study, the preventive effects of vitamin E supplementation for the occurrence of ONFH via improving endothelial dysfunction may not be expected in humans.
Oxidative stress corresponds to an imbalance between the production of oxidants, such as reactive oxygen species and free radicals, and their depletion by protective systems of antioxidants. As previous studies showed that corticosteroid and alcohol administration induces excess oxidants, 5,13 we speculated that overproduction of oxidants depletes carotenoids and decreases antioxidative capacity in patients with ONFH. This theory is supported by several recent experimental studies demonstrating the preventive effects of antioxidants, such as edaravone, hydrogen-rich saline, and selenium, on the occurrence of ONFH in rabbits, 18 –20 as well as vitamin E. 15 –17
However, lower serum levels of carotenoids in the ONFH group might result from their lower oral intakes due to the differences in dietary habits between the two groups. If oral intakes of carotenoids were lower in ONFH patients, we should emphasize the importance of improvement in the dietary habits of patients with steroid therapy or alcohol abuse, because epidemiologic observational studies reported that lower dietary intake of vegetables and fruits rich in carotenoids is associated with increased risk of diseases caused by oxidative stress, such as cardiovascular diseases and some types of cancers. 42,43 In this study, zeaxanthin/lutein, lycopene, and β-carotene were identified as antioxidant nutrients that could potentially prevent ONFH. However, supplementation of a large amount of single carotenoid may not be recommended in patients with high risk for ONFH. This is because intervention studies in large randomized controlled trials have shown no beneficial or even adverse effects of long-term supplementation of single antioxidants, such as vitamin E and β-carotene, on malignant neoplasms and cardiovascular diseases. 44,45 We think that overall diet quality rather than supplementation of single nutrients is important, as balanced amounts of multiple carotenoids are required to show adequate antioxidative activity 46 and preventive effect for malignant neoplasms. 47 To examine the association between the pathogenesis of ONFH and carotenoids in detail, we plan to perform further studies with the survey of dietary habits with a questionnaire 48 and direct measurement of serum oxidative stress and antioxidant potential using dedicated test kits. 49
This study has several limitations. First, a relatively small sample size might interrupt exact statistical assessments. Second, different populations between the ONFH and control groups might introduce selection bias. Third, the differences in the amount of smoking and alcohol intake between the two groups were not examined, which prevented a detailed assessment of the influence of smoking and alcohol. Finally, we could not assess the influence of high-dose steroid administration on the serum levels of carotenoids and α-tocopherol in this cross-sectional study. Most steroid-associated ONFH occur very early within 3 months after high-dose steroid administration, but ONFH becomes symptomatic several months or years after the occurrence. 50 Therefore, to confirm the influence of high-dose steroid administration on the serum levels of carotenoids and the true causal relationship between them and the occurrence of ONFH, prospective studies with their sequential measurements in patients who are required to receive high-dose steroids, such as steroid pulse therapy, should be performed.
In conclusion, this study showed lower serum levels of carotenoids in patients with ONFH than in healthy, community-living people. We speculate that carotenoids are related to the pathogenesis of ONFH and attribute to the prevention of the occurrence of ONFH via inhibiting endothelial dysfunction caused by oxidative stress.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
