Abstract
Introduction:
Tuberosity healing is known to be one of the factors that increase functional outcome in Reverse Total Shoulder Arthroplasty (rTSA). In fragility fractures, tuberosity healing is difficult to be achieved. The fragment stability and bone-forming agent application become strategies to overcome this problem. The purpose of this study was to evaluate the benefit of teriparatide application to promote tuberosity union in rTSA on four-part proximal humeral fractures to achieve better functional outcome.
Methods:
A retrospective study of a prospectively collected shoulder arthroplasty medical record was performed. Eleven patients were included in this analysis and consisted of two groups, teriparatide and no-teriparatide groups. Patients were evaluated on the range of motions and clinical outcome using the American Shoulder Elbow Surgeon (ASES) Shoulder Score and X-ray to determine tuberosity union. The group differences were analyzed by t-test or Mann–Whitney U test with SPSS version 20.0 for Windows.
Results:
The statistical analysis revealed no significant differences in mean age in both the groups. There were significant differences between teriparatide and no-teriparatide groups in external rotation (29.5° vs. 7.5°; p = 0.004), external rotation in 90° abduction (55° vs. 12.5°; p < 0.020), forward flexion (120° vs. 105°; p = 0.002) range of motions, and ASES (84.5 vs. 74.6; p = 0.019). We found clinical difference between teriparatide and no-teriparatide groups in time to achieve the initial callus (33 ± 18.3 days vs. 150 ± 42.4 days) and tuberosity consolidation (165.8 ± 70.3 days vs. 315 ± 106.1 days).
Conclusions:
Teriparatide administration supports tuberosity repair results in a high tuberosity healing rate with restoration of external rotation after rTSA in acute complex proximal humeral fractures.
Introduction
Proximal humeral fracture is one of the most common fragility fractures. It occurs almost in 10% of all fractures over the age of 65. 1,2 The traumatic injuries in this age can result in fracture comminution on single or both tuberosity due to decreased bone quality. Three-part or four-part complex proximal humeral fractures in the elderly are often not amenable to open reduction and internal fixation because of the bone quality, potential loss of fixation, risk of nonunion, and risk of avascular necrosis. 1,3 Another option is hemiarthroplasty (HA). HA in the setting of a comminuted proximal humeral fracture in the elderly is a technically demanding procedure because functional outcomes are unreliable in correlation between anatomical tuberosity union and other degenerative rotator cuff pathologies. Tuberosity union will be influenced by several factors in elderly patients such as the decreased bone quality and limited bone healing potential. Several literatures stated that HA in elderly proximal humeral fractures showed inconsistent results in functional outcome, power, and range of motions. 4,5
Reverse Total Shoulder Arthroplasty (rTSA) has developed as an alternative option for the treatment of acute traumatic proximal humeral fracture in the elderly patient population, 6 since the functional outcome of rTSA in this circumstance is less dependent on tuberosity healing and rotator cuff integrity compared to HA. rTSA relies on the deltoid function to restore the shoulder function. Gallinet et al. compared the range of motions and functional outcomes between rTSA and HA. 6 They concluded that rotation is better in the HA group than in the rTSA group. 7 They attempted to repair the tuberosity and to prepare the tuberosity to heal appropriately. The comparative study between union tuberosity group and unrepaired/nonunion tuberosity group showed that the union tuberosity group had significantly better rotational range. 7
Based on this evidence, several studies tried to propose techniques to promote tuberosity union on the rTSA in the setting of acute traumatic proximal humeral fracture. 8 The lower nonunion rate was not only caused by biomechanical factors but also by biological factors. The limited potential for tuberosity healing became a factor that needs to be emphasized. The randomized clinical trial study, evaluating the role of teriparatide in fracture healing on the osteoporotic patients, showed that this bone anabolic agent can be considered as a supplement option to promote fracture healing in this condition. 9
The purpose of this study was to evaluate the benefit of teriparatide application to promote tuberosity union in rTSA on four-part proximal humeral fractures to achieve better functional outcome.
Materials and methods
A retrospective study of a prospectively collected shoulder arthroplasty medical record was performed. All surgeries were performed by a single shoulder surgeon (B.C.) from January 2014 to December 2016. Indications for rTSA as a treatment for the proximal humeral fracture are a physiological age of 70 years or older, the four-part fracture, the three-part fracture with tuberosity comminution, and articular head-split fracture. There are contraindications to rTSA in the elderly population such as dementia patients, nonfunctional deltoid muscle, and preexisting axillary nerve injury. The medical records identified 15 consecutive patients who were treated for complex proximal humerus fractures with a shoulder replacement. Three patients were excluded due to neglected nonunion and malunion complex proximal humeral fractures. Patients were included if they had a minimum of 6 months of radiographic follow-up. The selection of a minimum of 6-month radiographic follow-up was necessary to observe tuberosity healing. One patient was excluded because of insufficient follow-up, leaving 11 patients included in this analysis. Among the evaluated patients, five patients were administered teriparatide as a bone-forming agent. Teriparatide administration to the patients started on the first postoperative day. The doses were 20 μg/day and injected subcutaneously. The duration of teriparatide application was 6 weeks for each patient. The contraindications for teriparatide application were also evaluated on all the patients such as serum calcium above reference levels, liver enzymes more than double the upper reference level, joint disease or any disease affecting bone metabolism, addiction to drugs or alcohol, medication with bisphosphonates before the fracture, mental dysfunction, inability to understand the language, or any other factors that would make informed consent impossible. Informed consent regarding publication was obtained from all individual participants included in the study.
Preoperatively, the author evaluated plain radiographs and three-dimensional computed tomography scans to assess the fracture pattern. Four-part fractures, in accordance with Neer’s description, were greater tuberosity, lesser tuberosity, humeral head, and humeral shaft separated. Additional pathologic change, such as dislocation and glenoid fracture, was reported. Postoperative radiographs and clinical outcomes were reviewed from the most recent follow-up visit. Postoperative radiographs (anteroposterior, Grashey, axillary, and scapular Y) were evaluated for radiographic evidence of tuberosity healing by comparing initial postoperative radiographs with the most recent radiographs. Greater tuberosity healing was considered complete if there was no evidence of displacement of the tuberosity, and trabeculation was noted between the humeral shaft and the greater tuberosity.
At the most recent follow-up, patients completed American Shoulder Elbow Surgeon (ASES) Shoulder Score as patient outcome survey. Measurements of postoperative active range of motion were performed with a manual goniometry for forward elevation, abduction, and external rotation, whereas internal rotation was measured by the reach test, identifying the highest midline lumbar or thoracic level obtained.
Operative techniques
All surgeries were performed using a standard rTSA prosthesis. The proximal humerus was routinely exposed through a delto-pectoral approach, and the tuberosities were identified and mobilized. The humeral head was then removed and taken to the back table, where morselized cancellous bone graft was harvested with a rongeur. The glenoid was then exposed, and the baseplate and glenosphere were implanted.
The intramedullary canal was then gently prepared with intramedullary reamers to define the diameter of the humeral canal. The final reamer diameter defined the size of the cement restrictor that was eventually used during cementing. Broaches were then placed such that the medial aspect of the broach lined up with the intact portion of the calcar (or estimated position in cases of calcar bone loss) and the humeral socket.
Next, three 2.5-mm drill holes were created approximately 1.5 cm distal to the fracture line. These drill holes were placed at the bicipital groove, and lateral–medial of the bicipital groove served as anchor points for tuberosity to shaft vertical suture repair. The shaft was prepared for cement by placing a cement plug then irrigating and drying the canal. The stem was then brought to the operative field. With use of a cement gun, cement was pressurized into the canal from distal to proximal. The stem was then impacted into the canal in 0°–20° of retroversion using a stem inserter, referred to the axis of the forearm, and held in position until the cement had hardened. The metaphyseal flare of the mono-block implant rested on the intact portion of the calcar to establish appropriate height of the implant. The cement, which typically extrudes from the vent holes, was removed. The appropriate polyethylene humeral insert was implanted based on motion and stability.
Then, we performed preparation of the tuberosities for suture fixation. Two types of suture alignments were used to balance suture fixation: vertical (greater tuberosity to humeral shaft) and cerclage (loop around the stem that connects both tuberosities). All sutures were passed at this time through the bone–tendon junction of the greater and lesser tuberosity. Balance stabilization was attempted during this procedure using six-configuration for loop-around suture and figure of eight for vertical suture (Figure 1). Additional bone graft was packed around the implant and proximal humerus when the greater tuberosity was either thin or severely comminuted in an effort to restore the appropriate offset of the greater tuberosity to prevent overreduction and to promote biological healing while creating osteoinductive and osteoconductive environment.
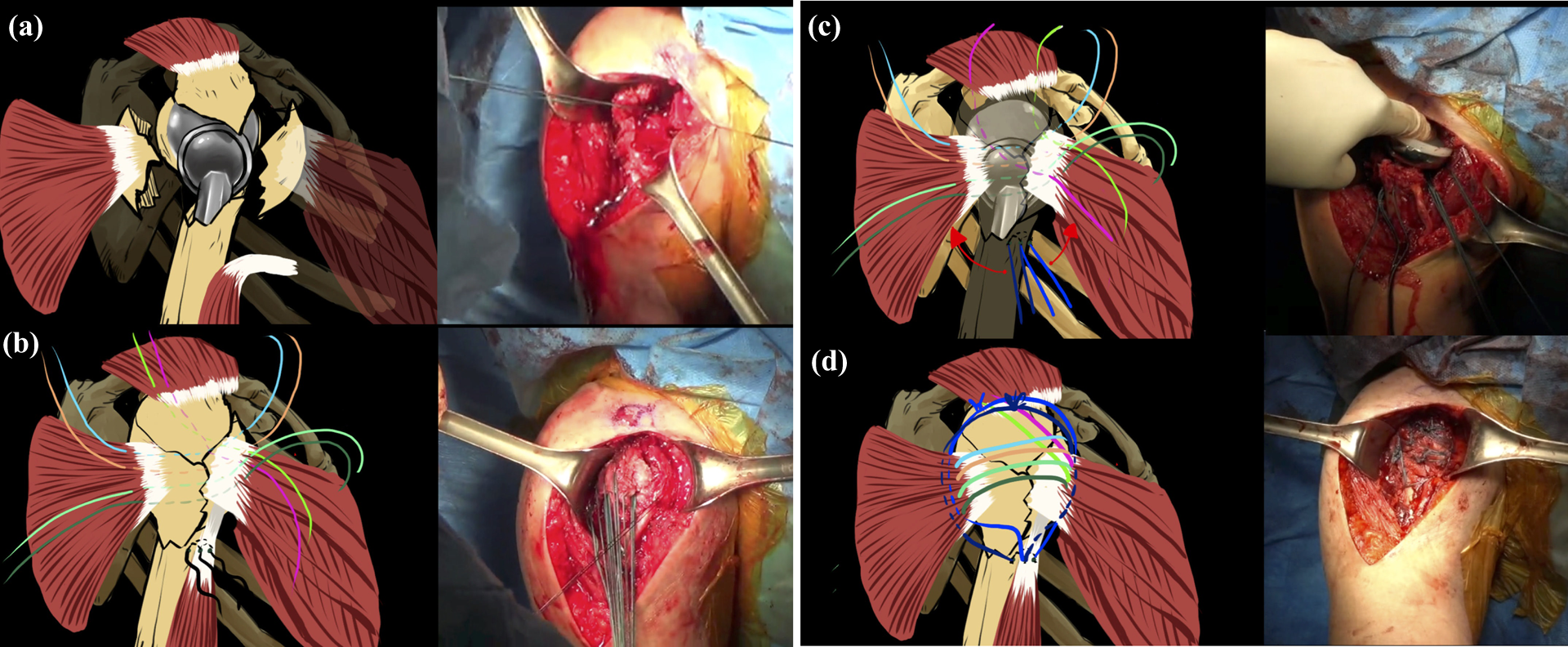
Steps six balance configuration tuberosity fixation.
Postoperative management
The patient began the supervised exercise for elbow, wrist, and hand active range of motion exercise on postoperative day 1. At this time, we immobilized the affected shoulder joint with abduction brace. After 4 weeks, the patient’s immobilizer was removed, and they began outpatient physical therapy working in passive range of motion and used their arms for the light activities of daily living while maintaining 5-pound lifting restriction. After 6 weeks, the patient progressed to active assisted range of motion exercises. Twelve weeks postoperatively, they advanced to deltoid and shoulder strengthening exercises.
Statistical analysis
Results were expressed as mean ± standard deviation or frequency (percentage) as appropriate. For continuous data, according to the results of Shapiro–Wilk test for normal distribution, group differences were analyzed by t-test or Mann–Whitney U test. For the categorical data, the Fisher Exact test was used to compare the variables between the study groups. Statistical analysis was performed with SPSS version 20.0 for Windows (SPSS Inc., Chicago, Illinois, USA) and Prism (ver. 7.0, Graph Pad Software, California, USA). P values <0.05 were considered statistically significant.
Results
Eleven patients were included in this study. They were consisted of two groups, five patients in the group administered teriparatide (Figure 2) and six patients in the group not administered teriparatide (Figure 3). The average age of the patients was 74.5 years (range: 60–82 years). All patients were injured by falls on the shoulder, and they underwent surgical treatment within 14 days of their respective injuries. All patients had four-part displaced proximal humeral fracture (AO classification 11-C2). The mean follow-up period was 19 months (range: 12–26 months). Furthermore, follow-up plain radiographs were checked every month until tuberosity union was achieved. The obtained range of motion that was compared statistically was followed up when tuberosity union was achieved.

Case in teriparatide group that achieved tuberosity union after rTSA. (a) CT-scan of the right shoulder showed proximal humeral fracture; (b) X-ray right shoulder anteroposterior; (c) immediate right shoulder X-ray post-rTSA; (d) right shoulder X-ray showed tuberosity union; (e) forward flexion Range of Motion evaluation; and (f) external rotation limitation in abduction 90° evaluation. rTSA: reverse shoulder arthroplasty.

Case in no-teriparatide group that did not achieved tuberosity union after rTSA. (a) CT-scan of the right shoulder showed proximal humeral fracture; (b) X-ray right shoulder anteroposterior; (c) immediate right shoulder X-ray post-rTSA; (d) right shoulder X-ray showed tuberosity union; (e) forward flexion Range of Motion evaluation; and (f) external rotation limitation in adduction evaluation. rTSA: reverse shoulder arthroplasty.
From X-ray evaluation, we found the superior tuberosity migration in three (50%) patients, the tuberosity resorption in one (17%) patient, and tuberosity union in two (34%) patients in the no-teriparatide group, whereas tuberosity union occurred in all (100%) patients in the teriparatide group. We found the clinical difference between teriparatide and no-teriparatide groups in time to achieve the initial callus (33 ± 18.3 days vs. 150 ± 42.4 days) and the tuberosity consolidation (165.8 ± 70.3 days vs. 315 ± 106.1 days; Figure 4).

Comparison of time to achieve initial callus and tuberosity union between teriparatide and no-teriparatide groups (in days).
From the statistical analysis, we found no significant difference in mean age of both the groups. The median of external rotation range of motion in the teriparatide group was significantly higher than in the no-teriparatide group (29.5° vs. 7.5°; p = 0.004). The median of external rotation range of motion in 90° abduction in the teriparatide group was significantly higher than in the no-teriparatide group (55° vs. 12.5°; p < 0.020). The mean forward flexion range of motion in the teriparatide group was significantly higher than in the no-teriparatide group (120° vs. 105°; p = 0.002; Figures 3 and 4). The mean ASES score in the teriparatide group was significantly higher than in the no-teriparatide group (84.5 vs. 74.6; p = 0.019; Table 1).
Comparison between teriparatide and no-teriparatide groups.

n: number of patients with available data; SD: standard deviation; Q1: lower quartile; Q3: upper quartile.
aUnpaired t-test.
bFisher-Exact test.
cMann–Whitney U test.
d p < 0.05.
Discussion
This study showed that four (67%) of six patients in group of rTSA without teriparatide administration experienced superior tuberosity migration and then tuberosity resorption. From this circumstance, we found that the capacity of biological healing for tuberosity itself was inconsistent in elderly patients, although we had already put the autograft bone grafted between the repaired tuberosity. Several studies focused only on the structural construct integrity to repair the greater and lesser tuberosity with a common goal of secure tuberosity fixation such as multiple sutures together with updated implant designs, developed in attempt to increase the healing rates of tuberosity. 8 –12 Findings from our clinical experience showed that several factors influenced the healing of tuberosity in acute proximal humeral fractures while performing rTSA. We noticed that not only biomechanical factors but also biological problems influenced the healing of tuberosity.
Due to this consideration, we administered teriparatide, the anabolic bone-forming agent, as a supplement option to obtain and accelerate tuberosities union in four-part acute proximal humeral fractures that performed rTSA. The mechanism of action of teriparatide can increase the osteoblast number, inhibit osteoblast apoptosis, and reactivate lining cells to resume their matrix-synthesizing function. Besides, animal experiments showed a dramatic improvement in skeletal repair by teriparatide. Postmenopausal women with a distal radial fracture in need of closed reduction, but not surgery, were assigned to 8 weeks of teriparatide; the median time to complete cortical bridging was analyzed, and the time to heal was shorter than placebo. 13,14
We administered 20 μg teriparatide, injected subcutaneously during 6 weeks after performing rTSA in acute proximal humeral fractures. All patients in this group obtained tuberosity union. The time to achieve initial callus formation and tuberosity union was also decreasing clinically. A previous study showed that teriparatide, administered to the osteoporotic distal femur fracture, could accelerate initial callus formation, bridge callus formation, and consolidation. 13 –16
From these cases, we evaluated the patient’s range of motion and functional outcome. We found that there were significant differences between teriparatide and no-teriparatide groups in external rotation (29.5° vs. 7.5°; p = 0.004), external rotation in 90° abduction (55° vs. 12.5°; p < 0.020), forward flexion (120° vs. 105°; p = 0.002) range of motions, and ASES (84.5 vs. 74.6; p = 0.019). We noticed that there was limited range of motion in external rotation (aside the body and abduction 90°) in patients in who tuberosity union was not achieved. This result was similar to a previous study which showed rTSA has an inferior result in rotation compared to shoulder HA, if the tuberosity is nonunion or is not being repaired. Additionally, the remaining patients revealed that tuberosity union was achieved, and improvements in rotatory function and strength of the shoulder were obtained. Another previous study also stated that there were improvements in active forward elevation, external rotation, and external rotation strength, which limited the chances of early prosthetic dislocation in complex shoulder fractures treated by rTSA if tuberosities were repaired and achieved union. 6,7 On the other hand, there was benefit to obtain tuberosity union as rapid as possible because the rehabilitation program will progress to active range of motion exercise (forward flexion, abduction, and external–internal rotation) after the tuberosity achieved union stage. The rapid progression of the patient to perform the rehabilitation to gain abduction and forward flexion active range of motion influenced the patient’s functional outcome.
Limitations of this study were related to the small population of patients and the size of comparison group that might have limited the statistical power. However, the total subjects of this study had fulfilled the minimal power sample, and this study examined a series of patients treated by one surgeon. Data were reviewed retrospectively. All patients included in this study were seen at a tertiary hospital, which may not be a representation of the general population. In the X-ray evaluation, we could not compare the group statistically because the patient’s number of tuberosity union in no-teriparatide group was too small to be compared with the teriparatide group. The range of motion between the two groups was obtained in 6 months without considering whether the tuberosity at that time was consolidated or not.
Conclusions
Teriparatide administration supports tuberosity repair results with a high tuberosity healing rate. Achieving tuberosity union as rapid as possible will optimize functions of the remaining rotator cuff tendon and promote early rehabilitation to restore the rotatory function after rTSA in complex proximal humeral fractures.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
