Abstract
Introduction:
The purpose of this present study is to investigate the efficacy of vancomycin-loaded VK100 silicone cement drug delivery system in the treatment of implant-related methicillin-resistant Staphylococcus aureus (MRSA) osteomyelitis in rats.
Materials and Methods:
Thirty-six adult (18–20 weeks old) female Sprague-Dawley rats were included in the study. All rats underwent experimental osteomyelitis surgery via injecting 100 µL bacterial suspension of MRSA into the medullary canal. After a 2-week duration for the formation of osteomyelitis model, rats were assigned randomly into four groups: control (C), systemic vancomycin (V), local vancomycin-loaded VK100 silicone cement (vVK100), and systemic vancomycin and local vancomycin-loaded VK100 silicone cement (V+vVK100). The following treatment protocols were administered to each group for 4 weeks. For group C, 0.9% saline solution equivalent to the volume of vancomycin dose (approximately 1 ml/kg) was administered intraperitoneally twice daily (12-h intervals). For group V, 15 mg/kg of vancomycin was administered intraperitoneally twice daily (12-h intervals). For group vVK100, vVK100 polymer was included so that the intramedullary canal of the rats are affected. For group V+vVK100, vVK100 polymer was included so that the intramedullary canal of the rats are affected and 15 mg/kg of vancomycin was administered intraperitoneally twice daily (12-h intervals). After 4 weeks of treatment, clinical, radiologic, microbiologic, and histopathologic evaluations were performed for all groups.
Results:
Results of this study revealed that all scores of the evaluation criteria for the treatment groups (groups V, vVK100, and V+vVK100) decreased due to the treatment protocols when compared to group C. These results show the effectiveness of all treatment protocols for the implant-related chronic MRSA osteomyelitis. However, there were no statistical difference between these three protocols.
Conclusions:
vVK100 polymer, as a local antibiotic delivery system, seems to be an effective method for the treatment of implant-related chronic MRSA osteomyelitis.
Introduction
Methicillin-resistant Staphylococcus aureus (MRSA) is the most common bacterial agent isolated from implant-related chronic osteomyelitis cases after orthopedic surgery. Despite the progression made in surgical techniques and proper antibiotic prophylaxis, chronic osteomyelitis is still a challenging problem for orthopedic surgeons. 1 –3
Conventional therapy for implanted-related osteomyelitis includes removal of all implants, drainage of pus, debridement of all nonviable tissues, obliteration of death spaces, reconstruction of bone defects, and proper antibiotic treatment against causative bacterial agent. Beside surgical interventions, antibiotherapy still remains the most important component of the treatment. Intravenous (IV) antibiotics with sufficient bactericidal concentration should reach the infection site to eradicate the infection. However, diminished vascularity at the target site, usually seen in infections, is an important obstacle for this effective action. Furthermore, antibiotics should be continued for at least 4–6 weeks. 1 –3
Antibiotics can be administered by systemic route, direct application to the target site, or in a combined fashion. The use of local antibiotic delivery systems provides higher drug concentrations at the target site, thus eliminating the risks and the side effects associated with long-lasting oral or parenteral therapies such as systemic toxicity. Furthermore, they act as a spacer and fill the dead spaces created by radical debridement of all nonviable tissues. 1 –3
Systemic vancomycin and vancomycin-loaded local antibiotic delivery systems are the most common treatment for implant-related chronic osteomyelitis. Currently antibiotic-loaded polymethyl methacrylate (PMMA) is the commonly used delivery system in orthopedic practice. However, it has some disadvantages. Hardening of PMMA is an exothermic reaction, and during polymerization PMMA can reach high temperature as high as 80°C. This feature of PMMA limits its use with thermolabile antibiotics. Second, the release properties of PMMA have been questioned for its sufficiency in several previous studies. It has been proposed that almost 90–95% of the loaded antibiotic is stuck in the PMMA and cannot be released at the target site. 4 –10 Thus, new drug delivery systems having better release properties and compatibility with a large spectrum of antibiotics, including thermolabile ones, are currently under investigation.
More recently, a silicone cement called VK100 was introduced in the market by Bonwrx® (Arizona, USA). This dual-paste cement contains dimethyl methylvinyl siloxanes (87%), barium sulfate powder (14%), and a platinum catalyst (15 ppm as metal) in the first component, and dimethyl methylvinyl siloxanes (78%), barium sulfate powder (15%), and a methylhydrogen siloxane cross linker (7%) in the second component. This silicone cement has some advantages over classical PMMA cement. After hardening, the structure is not completely rigid and provides a certain amount of compressibility. It is hypothesized that with this feature it could prevent the adjacent segment fractures commonly seen at percutaneous vertebroplasty and kyphoplasty procedures. Unfortunately, preliminary results for bone augmentation applications (elastoplasty) are very poor, with more than 60% leakage into the spinal canal and pulmonary embolism. The second most advantageous feature is that it does not emit heat during hardening process. Thus, it could prevent heat necrosis and possible neural damage. In addition, a large number of antibiotics including the thermolabiles could be used in the antibiotic delivery systems, unlike the classical PMMA. 11,12 In the present literature, no study focuses on VK100 silicone cement as an antibiotic delivery system.
We hypothesized that VK100 silicone cement may be an alternative antibiotic delivery system. Thus, we investigated the efficacy of vancomycin-loaded VK100 drug delivery system in the treatment of implant-related MRSA osteomyelitis in rats.
Materials and methods
Thirty-six adult (18–20 weeks of age) female Sprague-Dawley rats weighing between 250 and 350 g were included in the study. The animals were fed a standard laboratory diet and water, and they were housed separately in the standard cage in an air-conditioned room with 12-h light–dark cycle, where the temperature (20–22°C) and relative humidity (65–70%) were kept constant. Before the study initiation, all experimental protocols were approved by local ethics committee (Institutional Animal Care and Use Committee of Gulhane Military Medical Academy, Acceptance No: 28 June 2013/43). The study was carried out in the “Center for Experimental Animals” at the same institution.
After the formation of osteomyelitis model, rats were randomly assigned into four groups each consisting of nine subjects, respectively, control (C), systemic vancomycin (V), local vancomycin-loaded VK100 silicone cement (vVK100), and systemic vancomycin and local Vancomycin-loaded VK100 silicone cement (V+vVK100) (Table 1).
Study groups.

C: control; V: systemic vancomycin; vVK100: local vancomycin-loaded VK100 silicone cement; V+vVK100: systemic vancomycin and local vancomycin-loaded VK100 silicone cement.
Preparation of the MRSA suspension
MRSA (N315) (NBCI Taxonomy ID: 158879) suspension was prepared to 0.5 McFarland (108 cfu/ml) turbidity level.
Experimental osteomyelitis model
Tibial osteomyelitis model was created using the similar techniques used by Kalteis et al., 13 Monzon et al., 14 and Oguz et al.. 15 All rats underwent previously described techniques and procedures as follows.
Rats were anesthetized with intraperitoneal injection of 10 mg/kg xylazine and 50 mg/kg ketamine hydrochloride mixture and placed at supine position. After sterilization and draping of the left limb, 1 cm incision was made over the proximal tibia just distal to the patellar tendon insertion. A 2-mm Kirschner wire was used to create a cortical window, then the intramedullary canal of the tibia was reamed with 1.6 mm K wire as far to the distal metaphysis. MRSA suspension of 100 µl was injected directly into the intamedullary canal to create bacterial contamination. A 5-mm 26G needle was inserted into the medullary cavity and the cortical window was closed with bone wax. Wound was closed with nonabsorbable sutures (Figure 1).

(a) Skin incision, (b) cortical window, (c) injection of 100 µl MRSA suspension, (d) insertion of the 5-mm needle into the medullary cavity, (e) closure of cortical window with bone wax. MRSA: methicillin-resistant Staphylococcus aureus.
For 3 days postoperatively, oral ibuprofen (15 mg/kg) was administered for pain control. The infection was allowed to progress for 2 weeks. At the end of 2 weeks, with clinical and radiologic findings, osteomyelitis was confirmed in all rats.
Preparation of the vVK100 silicone cement
For each rat, before the implant removal surgeries, 30 mg/kg of vancomycin was weighed at precision scales and kept at sterile flacons. Vancomycin 30 mg/kg and VK100 polymer 200 mg were mixed homogenously under sterile conditions just before the implant removal surgeries, to maintain the liquidity of the cement for injection.
Implant removal and vVK100 silicone cement application
After giving 2 weeks’ duration for the formation of osteomyelitis model, all groups underwent implant removal surgeries.
After implant removal, premixed vVK100 silicone cement was injected into the left medullary canal of the rats of vVK100 and V+vVK100 groups. The cement was prepared just before injection to maintain the liquidity. We used 2 cc sterile injector with a 16G plastic IV cannula at every single rat and tried to fill the destructed anterior tibial cortex and the whole medullary canal with the polymer to the far metaphyseal region.
Treatment protocols
After the implant removal and vVK100 application surgeries, we administered the following treatment protocols to each group for 4 weeks: For group C, 0.9% saline solution equivalent to the volume of vancomycin dose (approximately 1 ml/kg) was administered intraperitoneally twice daily (12 h intervals). For group V, 15 mg/kg of vancomycin was diluted with 1 ml/kg of 0.9% saline solution and administered intraperitoneally twice daily (12 h intervals). For group vVK100, the vVK100 polymer was inserted to the affected intramedullary canal of the rats. For group V+vVK100, the vVK100 polymer was inserted to the affected intramedullary canal of the rats and 15 mg/kg of vancomycin was diluted with 1 ml/kg of 0.9% saline solution and administered intraperitoneally twice daily (12 h intervals).
Specimen harvesting and killing
At the end of the 4-week treatment protocols, all rats underwent surgery for specimen harvesting. They were anesthetized with intraperitoneal injection of 10 mg/kg xylazine and 50 mg/kg ketamine hydrochloride mixture and placed in supine position. After sterilization and draping of the left limb, left tibia was meticulously dissected and stripped of the soft tissues. Bone tissue specimens were obtained from the anterior cortex of the tibia with mini rongeur and kept under sterile conditions for later microbiologic evaluations. The remaining whole tibia was kept in 10% buffered formalin solution for later histopathologic evaluations.
After harvesting procedure, all rats were killed by overdose (100 mg/kg) of intraperitoneal ketamine hydrochloride injection.
Evaluations
Clinical evaluation
During harvesting procedure, bone and soft tissue findings were noted according to our criteria (Table 2). Total clinical scores were calculated for each rat. Statistical comparisons were made according to the total scores and each separate criteria scores.
Clinical evaluation criteria.

Radiologic evaluation
Radiologic evaluations were performed three times: at preoperative first day (Day 0 – preoperative) of experimental osteomyelitis model, before implant removal surgeries (Day 14 – pretreatment) and at the end of treatment protocol (Day 42 – posttreatment), before the specimen harvesting and killing.
Radiographs were taken under intraperitoneal ketamine anesthesia using a digital mammography. All radiographs were taken from a film-to-focus distance of 30 cm and X-ray beam was centered to the proximal third of the tibia with a standard dose (30 Kv/160 mps). We preferred digital mammography over digital X-ray machines for the high-resolution and more detailed digital images.
Radiographs were evaluated by a single investigator who was blinded to the assignment of study groups according to the modified radiologic chronic osteomyelitis criteria described by An and Friedman (Table 3; Figure 2). 16 –19
An and Friedman’s modified radiologic chronic osteomyelitis criteria.


(a) Radiologic appearance of the left tibia of a group vVK100 rat at day 0; (b) radiologic appearance of the left tibia of the same rat at day 14, note the implant and the osteolysis due to osteomyelitis; (c) radiologic appearance of the left tibia of the same rat at day 42, note the vVK100 polymer and sclerosing osteolytic regions due to treatment. vVK100: local vancomycin-loaded VK100 silicone cement.
Total pretreatment (Day 14) and posttreatment (Day 42) radiologic scores were calculated for each rat. Statistical comparisons were made according to the total scores and for each criterion score separately.
Microbiologic evaluation
All microbiologic samples were weighed at precision scales and recorded. Samples were homogenized with 1 ml tryptic soy broth at a brass mortar with Muller In Class II biosecure cabins.
Quantitative cultivation was made by homogenized samples for enumeration of colonies. Samples taken using 1 μl calibrated loop and dispersed as a pine tree pattern on 5% sheep blood agar (SBA) and eosin methylene blue (EMB) agar. Also samples taken using 10-μl pipette and dispersed as cage pattern on 5% SBA and EMB. From the homogenized samples, 10 μl was taken and diluted in 1 ml tryptic soy broth. The same procedure was performed with the diluted samples, respectively (1 and 10 μl cultivated). Inoculated plates were incubated at 35°C for 24–48 h in an atmosphere of 5% carbon dioxide. At the end, the colonies were counted quantitatively. Gram staining, catalase test, tube coagulase test, and Chapman agar cultivation were made for identification. Kirby-Bauer disk diffusion susceptibility test was used for antimicrobial susceptibility testing according to the Clinical and Laboratory Standards Institute (CLSI, Pennsylvania, USA) 2013 guidelines. Samples were prepared to 0.5 McFarland (108 cfu/ml) turbidity level and were streaked onto entire surface of Mueller-Hinton agar (MHA) via swabs homogenously. Samples were incubated at 35°C for 24 h and assessed for CLSI 2013 criteria. For each sample, grown colonies were quantitatively counted and weighed and colony-forming unit per gram (cfu/g) was calculated. For each rat, statistical comparisons were made according to the obtained numbers of colony-forming unit per gram.
Histopathologic evaluation
Tissue specimens were evaluated macroscopically and sampled properly for microscopic assessment. Samples were fixed in 10% buffered formalin solution for at least 72 h and decalcified in acid solution (10% CH2O2 + 8% HCl + 82% tap water). Afterward the samples were embedded in paraffin blocks using tissue embedding system, and 4 µm sections were obtained using microtome. Proper sections were taken on the slides and stained with hematoxylin–eosin at the Autostainer device. Slides were evaluated with Standard Light Microscope according to microscopic histological assessment parameters and scoring system criteria of Smeltzer et al. (Table 4). 20 –22 Total histopathologic scores were calculated for each rat. Statistical comparisons were made according to the total scores and each criterion score separately.
Histological assessment parameters and scoring system according to Smeltzer et al. 20

Statistical analysis
Shapiro-Wilk test was used to evaluate the normality of the data. For descriptive statistics, mean ± standard deviation (SD) and median (minimum-maximum) were calculated for continuous variables and the percentage for discontinuous variables. Comparison of variables between groups was made using one-way analysis of variance, Kruskal–Wallis and chi-square tests. When statistical difference was detected between the groups, Mann–Whitney U test with Bonferroni correction and Tukey’s honest significant difference tests were used to detect the two different groups. The Wilcoxon signed-rank test was used when comparing pre- and posttreatment variables of related groups. Significance level was <0.05.
Results
There were no complications during the surgeries and treatment period. Osteomyelitis was induced in all rats. All 36 rats survived the experimental period and included for analyses.
Clinical scores
Statistically significant difference was found between the four study groups when compared according to the total clinical scores (p < 0.001).
Between group C and group V (p < 0.001), group C and group vVK100 (p = 0.002), and group C and group V+vVK100 (p < 0.001) statistically significant difference was found (After Bonferroni correction, p < 0.008 is determined statistically significant.). There was no statistically significant difference between treatment groups (groups V, vVK100 and V+vVK100).
Median values of the total clinical scores for control group and treatment groups were 2 and 0, respectively. Total clinical scores of treatment groups decreased due to treatment protocols compared to the control group. These results show the effectiveness of treatment protocols according to clinical evaluations.
There was no statistically significant difference between treatment groups, and these results revealed no significant efficiency difference between treatment protocols according to clinical evaluations (Table 5).
Comparison of total clinical scores between groups.

C: control; V: systemic vancomycin; vVK100: local vancomycin-loaded VK100 silicone cement; V+vVK100: systemic vancomycin and local vancomycin-loaded VK100 silicone cement.
aKruskal–Wallis test.
Radiologic scores
Statistically significant difference was found between the four study groups when compared according to the posttreatment (Day 42) total radiologic scores (p = 0.001).
Between group C and group V (p = 0.012), group C and group vVK100 (p = 0.006), and group C and group V+vVK100 (p = 0.001) statistically significant difference was found (p < 0.05 is determined statistically significant.). There was no statistically significant difference between treatment groups.
Mean values of posttreatment total radiologic scores for groups C, V, vVK100, and V+vVK100 were 5.7 ± 1.8, 3.0 ± 2.0, 2.7 ± 1.5, and 2.3 ± 1.5 (mean ± SD), respectively. Posttreatment total radiologic scores of treatment groups decreased due to treatment protocols compared to the control group. These results show the effectiveness of treatment protocols according to radiologic evaluations.
There was no statistically significant difference between treatment groups, and these results revealed no significant efficiency difference between treatment protocols according to radiologic evaluations (Table 6).
Comparison of posttreatment total radiologic scores between groups.
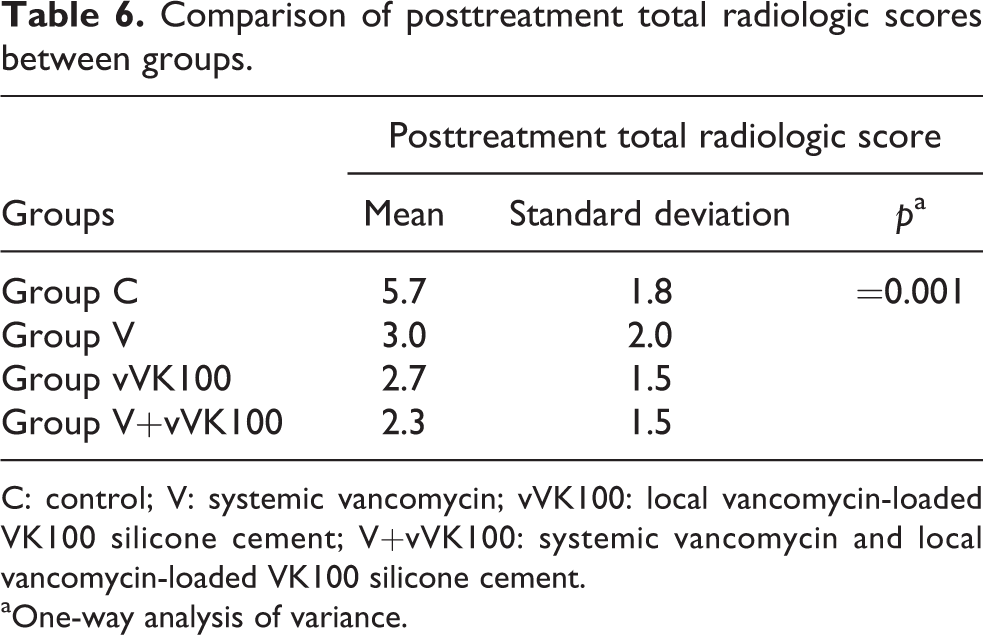
C: control; V: systemic vancomycin; vVK100: local vancomycin-loaded VK100 silicone cement; V+vVK100: systemic vancomycin and local vancomycin-loaded VK100 silicone cement.
aOne-way analysis of variance.
In addition, four study groups were compared separately according to pre- and posttreatment total radiologic scores.
For group C, the mean value for pre- and posttreatment total radiologic scores was 2.8 ± 0.6 and 5.7 ± 1.8 (mean ± SD), respectively. This increase explains the radiologic findings of untreated progressive osteomyelitis and the difference was statistically significant (p = 0.007).
For group V, the mean value for pre- and posttreatment total radiologic scores was 5.3 ± 1.4 and 3.0 ± 2.0 (mean ± SD), respectively. This decrease explains the regression of radiologic scores due to the effective systemic vancomycin treatment. The difference was statistically significant (p = 0.007).
For group vVK100, the mean value for pre- and posttreatment total radiologic scores was 4.4 ± 1.5 and 2.7 ± 1.5 (mean ± SD), respectively. This decrease explains the regression of radiologic scores due to the effective application of local vVK100. The difference was statistically significant (p = 0.035).
For group V+vVK100, the mean value for pre- and posttreatment total radiologic scores was 4.6 ± 1.5 and 2.3 ± 1.5 (mean ± SD), respectively. This decrease explains the regression of radiologic scores due to the effective systemic vancomycin treatment and local application of vVK100. The difference was statistically significant (p = 0.011) (Table 7).
Comparison of pre- and posttreatment total radiologic scores of separate groups.
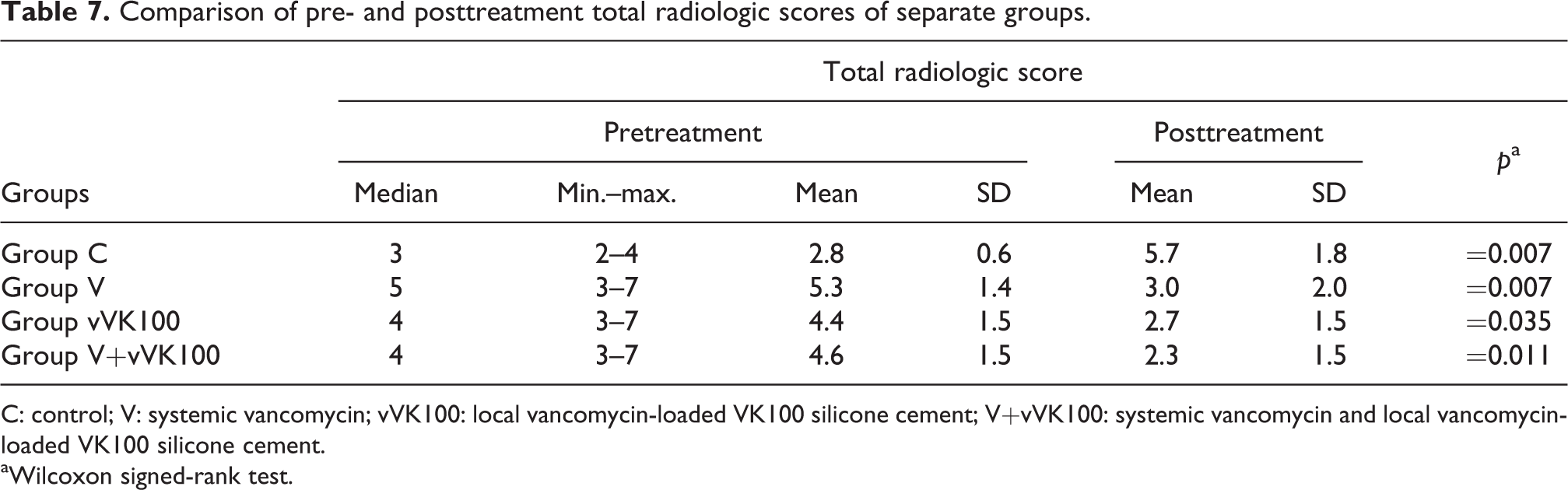
C: control; V: systemic vancomycin; vVK100: local vancomycin-loaded VK100 silicone cement; V+vVK100: systemic vancomycin and local vancomycin-loaded VK100 silicone cement.
aWilcoxon signed-rank test.
Microbiologic scores
All samples revealed gram-positive cocci in clusters and catalase, and tube coagulase and Chapman tests were positive.
All samples were confirmed as vancomycin sensitive MRSA (oxacycline and cefoxitin resistant) with Kirby-Bauer disk diffusion susceptibility test, according to the CLSI 2013 guidelines.
Statistically significant difference was found between four study groups when compared based on the colony-forming units per gram (cfu/g) (p < 0.001). Between group C and group V (p = 0.001), group C and group vVK100 (p = 0.002), and group C and group V+vVK100 (p < 0.001) statistically significant difference was found (After Bonferroni correction, p < 0.008 is determined statistically significant.). There was no statistically significant difference between treatment groups.
Most evident colony formation was observed in group C (5 × 106 cfu/g). Median values for Groups C, V, vVK100 and V+vVK100 were 2 × 106, 5 × 104, 9 × 104, and 2 × 104 cfu/g, respectively.
Compared to the control group, the quantity of the colony-forming units per gram of treatment groups decreased due to the treatment protocols. These results show the effectiveness of treatment protocols according to microbiologic evaluations. There was no statistically significant difference between the treatment groups, and these results reveal there is no significant efficiency difference between treatment protocols according to microbiologic evaluations (Table 8).
Comparison of cfu/g quantity between groups.

C: control; V: systemic vancomycin; vVK100: local vancomycin-loaded VK100 silicone cement; V+vVK100: systemic vancomycin and local vancomycin-loaded VK100 silicone cement; cfu/g: colony-forming units per gram.
aKruskal–Wallis test.
Histopathologic scores
No statistically significant difference was found between the four study groups when compared based on the total pathologic scores (p = 0.092).
Median values of total pathologic scores for the control and treatment groups were 4 and 2, respectively. Compared to the control group, the total pathologic scores of treatment groups decreased due to the treatment protocols. These results show the effectiveness of treatment protocols according to histopathologic evaluations. There was no statistically significant difference between treatment groups, and these results revealed no significant efficiency difference between treatment protocols according to histopathologic evaluations (Table 9).
Comparison of total pathologic scores between groups.

C: control; V: systemic vancomycin; vVK100: local vancomycin-loaded VK100 silicone cement; V+vVK100: systemic vancomycin and local vancomycin-loaded VK100 silicone cement.
aKruskal–Wallis test.
Discussion
The purpose of this present study is to investigate the efficacy of vancomycin-loaded VK100 silicone cement drug delivery system in the treatment of implant-related MRSA osteomyelitis in rats.
Clinical, radiologic, microbiologic and histopathologic evaluations revealed that vVK100 as a local antibiotic delivery system seems to be an effective method in the treatment of implant-related chronic MRSA osteomyelitis.
Studies about local antibiotic release systems in literature contain different kinds of scaffolds and antibiotics. At their in vivo study, Liu et al. used poly (
In our study clinical, radiologic, microbiologic, and histopathologic evaluations revealed that all scores at each particular evaluation criterion decreased for the treatment groups (groups V, vVK100, and V+vVK100) due to the treatment protocols, compared to control group. These results correlate with the current literature.
There are numerous studies comparing local antibiotic release systems and systemic antibiotic treatment. Cevher et al. compared vancomycin-loaded biosoluble chitin microbeads and intramuscular vancomycin treatment of rat osteomyelitis model and reported that microbeads were more efficient. 4 Lucke et al. compared local and systemic gentamycin treatment at an implant-related rat osteomyelitis model. For local application, they loaded gentamycin to PLGA and coated the titanium implant with it. They report that local treatment was more successful. 27 Kundu et al. performed in vitro and in vivo studies using ceftriaxone-sulbactam-loaded bioactive glass and reported that it provided higher minimum inhibition concentration values and contributed new bone formation compared to IV antibiotherapy. 28
On the contrary of the literature mentioned above, our study revealed that there is no statistically significant difference between our local, systemic, or combined fashion treatment protocols of implant-related chronic rat osteomyelitis model.
Based on our study results, we believe that both in vivo and in vitro release kinetics of vVK100 polymer should be thoroughly researched.
At the present time, PMMA is the only polymer that has clinical usage, but it has many disadvantages. Polymerization of PMMA emits high degrees of heat and affects the surrounding soft tissues. In addition, it could decrease the efficacy and its period of thermolabile antibiotics. Due to this, a limited number of antibiotics could are to be used with it. Gentamycin, tobramycin, vancomycin, and teikoplanin are the most commonly used ones. In addition, other amino glycosides, penicillins and cephalosporins can be used with PMMA. Fluoroquinolones, tetracyclines and polymyxin B are thermolabile and cannot be used with PMMA. In addition, uncontrolled micro fractures may occur at the release surface of PMMA. In this case, 90–95% of the antibiotic remains in the inner depths of PMMA, and the amount of the released dose are uncontrolled. 4 –10
In their study, Radin et al. compared titanium alloy coated with vancomycin-loaded calcium phosphate and vancomycin-loaded PMMA beads. They reported that calcium phosphate ceramic releases much more antibiotics compared to PMMA. 9 Laurencin et al. compared gentamycin-loaded biosoluble polyanhydride and gentamycin-loaded PMMA in an experimental rat osteomyelitis model. They observed that gentamycin-loaded polyanhydride treated osteomyelitis and offered bone debridement before the usage of gentamycin-loaded PMMA. 29 Saito et al. applied vancomycin-loaded PMMA beads to five patients for the treatment of chronic osteomyelitis. They did not observe any evident healing but demonstrated bactericidal effect when vancomycin-loaded HA was applied. 24 El-Ghannam et al. loaded self-soluble bioactive silica–calcium phosphate nanocomposite with vancomycin and compared it with vancomycin-loaded PMMA. They reported it contained higher rates of antibiotics and released therapeutic doses for 35 days more, compared to vancomycin-loaded PMMA. 30
To our knowledge, there is no study concerning the utilization of VK 100 silicone cement for antibiotic delivery system. In addition, there is no in vivo study. We have used VK100 polymer for local antibiotic release system, and based on our results we suggest VK100 polymer to PMMA as an alternative antibiotic release system.
Efficacy comparison of VK100 polymer and other local antibiotic release systems like PMMA was not within the scope of our study. Studies directed at this scope should be considered, with more subjects and groups.
In addition, studies utilizing thermolabile antibiotics with VK100 polymer should be planned. Hence, like PMMA, the polymer has no exothermic side effects. We believe with this feature VK100 polymer can demonstrate the clinical success of PMMA.
Studies comparing the local antibiotic release system with VK100 polymer and systemic antibiotic treatments should also focus on the correlation between blood levels and side effects of the related antibiotics. In addition, local usage of various antibiotics with numerous systemic side effects should be tested with various doses.
Its applicability with thermolabile antibiotics and injectable application is among its advantages.
The most frustrating disadvantage of the polymer is the handling difficulty. Its sticky and viscous structure complicates the mixing process with antibiotics and applications around soft tissues. To overcome the mixing difficulties, premixed antibiotic containing VK100 polymer syringes could be designed and manufactured. In addition, nozzles with various calibers could be designed for different site applications.
In conclusion, according to our data vVK100 polymer, as a local antibiotic delivery system, is an effective method in the treatment of implant-related chronic MRSA osteomyelitis. We suggest that new studies and improvements considering the handling difficulties should be conducted, and vVK100 polymer could be an alternative to PMMA in the present orthopedic practice.
Footnotes
Acknowledgments
The authors are grateful to Dr Inanc Guvenc, MD, for assistance with radiologic imaging and coordination with radiology department; Dr Abdullah Kilic, MD, and A. Korhan Sig, MD, for assistance with the preparation of the MRSA suspension, microbiologic evaluations, and coordination with microbiology laboratories; Dr Cansel Kose Ozkan, PhD, for assistance with preparation of vancomycin flacons and coordination with pharmacology department; Dr Yildirim Karslioglu, MD, for assistance with histopathologic evaluations and coordination with pathology laboratories; and Dr Cengizhan Acikel, MD, and Dr Guven Oysul, MD, for assistance with statistical analysis. Authors especially thank to Rose Carmichael, president of Bonwrx® Inc., for providing VK100 polymers.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All experimental protocols were approved by local ethics committee (Institutional Animal Care and Use Committee of Gulhane Military Medical Academy, Acceptance No: 28 June 2013/43).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Gulhane Military Medical Academy Scientific Research Board financially supported this work (Project No: AR-2013/35).
