Abstract
Purpose:
To clarify the relationship between anterior cruciate ligament (ACL) remnant tissue at the time of surgery and preoperative evaluation of ACL-injured knee.
Methods:
A total of 123 patients were enrolled and classified into four groups based on the classification of ACL remnant tissue. To quantify the signal–noise quotient (SNQ), five regions of interest (ROIs), such as tibial, mid, femoral ACL remnant, tendon of quadriceps femoris to normalize the signal intensity of ACL remnant tissue, and background measurements, were measured. Preoperative side-to-side difference (SSD) and SNQ were analyzed.
Results:
Significant differences were observed in preoperative SSD of the four groups (p = 0.021), and a post hoc analysis revealed that SSD in Crain type 2 was significantly smaller than that in Crain type 4 (p = 0.014). SSD in Crain type 3 was also significantly smaller than that in Crain type 4 (p = 0.0030). There were significant differences in SNQ at the tibial, mid, and femoral portion in Crain types 2 and 3 (p < 0.001); SNQ at the tibial portion in Crain type 2 was significantly lower than that at the mid portion (p = 0.024); and SNQ at the tibial portion in Crain type 3 was significantly lower than that at the mid portion
Conclusion:
The femoral attachment of the ACL remnant tissue may influence the SNQ of the tibial remnant tissue and the stability of the ACL-injured knee.
Introduction
Injury of the anterior cruciate ligament (ACL) is very common. 1 The short-term results after ACL reconstruction are favorable. 2 However, not all patients are satisfied with their knee stability after surgery. 3 One of the ideal goals of ACL reconstruction is to restore normal knee stability and function. To achieve this, anatomical ACL reconstruction, such as double-bundle ACL reconstruction, 4 was developed, and good clinical results have been reported. 5 However, issues such as persistent graft necrosis, 6 insufficient recovery of joint position sense, 7 and risk of reinjury of the graft 8,9 remain after ACL reconstruction. Therefore, new therapeutic approaches to ACL reconstruction are needed to solve these problems. Recently, ACL remnant tissue has attracted attention due to the possibility of accelerating the graft remodeling phase. 10,11 In a rat model, type III collagen, α-smooth muscle actin, and neurofilament antigen were significantly more highly expressed in the remnant augmented group. 12 In a rabbit model, remnant tissue preservation significantly increased expression of messenger ribonucleic acid of the vascular endothelial growth factor receptor (VEGF), which enhanced regeneration of blood vessels. 13 Additionally, ACL remnant tissue is thought to be a possible source of reinnervation because there are proprioceptive mechanoreceptors. 14 In a sheep ACL reconstruction model, preservation of the ACL remnant tissue enhanced cell proliferation, revascularization, and regeneration of the proprioceptive organs. 15 Furthermore, a nearly native tibial enthesis structure can be preserved in the preservation of ACL remnant tissue that contributes a higher initial stiffness in the remnant tissue-grafted tendon complex, 16 thus reducing anterior laxity.
Consequently, several investigators have developed ACL reconstruction with remnant tissue preservation and reported better clinical outcomes than that of conventional ACL reconstruction. 17 –20 However, controversy remains on whether remnant tissue can accelerate the remodeling phase of the graft in all generations. For example, there is a report that the ACL remnant in adolescent patients had a significantly higher potential for proliferation and multilineage differentiation. 21 In a rat ACL reconstruction model, ACL remnant tissue from younger rats enhanced early bone–tendon healing compared to older rats. 22 Therefore, surgeons should consider patient’s age when planning ACL reconstruction with remnant tissue preservation, as the procedure is more complex than a conventional one. Magnetic resonance imaging (MRI) is thought to be useful to morphologically examine the remodeling of the graft. 23 In a previous study, MRI indicated that preserved remnant bundles showed progressive remodeling in the ACL graft. 24 However, there are few reports on MRI evaluations of ACL remnant tissue.
The purpose of this study was to clarify the relationship between Crain classification of ACL remnant tissue at the time of surgery and preoperative knee stability and MRI of ACL-injured knee. We hypothesized that there were significant differences concerning the preoperative knee stability and MRI of ACL-injured knee between the groups classified according to Crain classification.
Methods
The institutional review board of the ethics committee of our institution approved this retrospective comparative study (approval number: 160614).
Patient selection
Patients with unilateral ACL injury who met the criteria for arthroscopic reconstruction and underwent ACL reconstruction at a single institution from May 2014 to March 2015 were included in this study. Patients with previous injury or surgery in the opposite knee were excluded to avoid bias in assessing patient-reported outcomes.
All patients were diagnosed as having ACL injury based on clinical findings, such as positive Lachman test and MRI. Physical examination and interpretation of radiological evaluations were performed by orthopedic specialist, authorized by the Japanese Orthopaedic Association, and well-experienced knee surgeons. None of them is a coauthor of the present study. The exclusion criteria were patients with multiligamentous injury, such as posterior cruciate ligament (PCL), posterolateral and posteromedial knee complex injuries, and medial and lateral collateral ligament injuries who simultaneously underwent another ligament reconstruction; patients with associated fractures of the femur or tibia; patients with severe chondral lesion that needed an additional procedure at the time of ACL reconstruction, obvious varus or valgus knee osteoarthritis; and patients with past history of knee surgery. Initially, 180 patients were selected from the medical records, but 40 patients were excluded as they did not meet the inclusion criteria. Additionally, 17 patients were excluded because they had not undergone initial MRI. The remaining 123 patients were enrolled in this study.
There were 62 male and 61 female patients, with 49 right and 74 left ACL injuries. The average age of the patients was 27.9 ± 13.5 years (12–70 years old). The average time from injury to MRI testing was 7.99 ± 11.3 weeks (0–52 weeks), and the average time from injury MRI testing to surgery was 5.88 ± 6.13 weeks (1–36 weeks). The patient characteristics are shown in Table 1.
Patient characteristics.a

MRI: magnetic resonance imaging.
aData are expressed as mean ± standard deviation.
We retrospectively investigated the relationship between ACL remnant tissue, classified according to four types that Crain et al. reported 25 at the time of surgery, and the preoperative condition of the knee joint as determined by MRI evaluations of the ACL remnant tissue.
Radiological evaluation
The MRI examinations were performed on a 1.5-T instrument (Echelon RX, Hitachi, Japan). The knee was placed in neutral position in an extremity coil. The sagittal images were acquired using fast spin-echo proton density-weighted imaging (PDWI). The repetition time range and echo time values were 2000 ms and 10 ms, respectively, for PDWI. The signal–noise quotient (SNQ) was calculated using the region of interest (ROI) technique. The intra-articular signal of the ACL remnant tissue was divided into three zones to analyze the signal intensity. The tibial, mid, and femoral thirds were defined as three zones. The signal from the quadriceps femoris tendon was measured after placing the ROI in the patellar upper limit level to normalize the signal intensity of the ACL remnant tissue. For background measurements, the ROI was placed approximately 2 cm anterior to the patellar tendon. Diameter of the ROI circle was set at 3.3 mm (Figure 1). All five ROI measurements were performed using a Picture Archiving and Communication System (PACS) monitor (GE Healthcare, Barrington, Illinois, USA) and a mouse cursor. To quantify SNQ from the ROI measurements, the following equation
24
reported by Ahn et al. was used.


Sagittal MRI of the knee shows the positions of the five regions, which included the tibial portion, mid portion, femoral portion, QT, and background. QT: quadriceps tendon; MRI: magnetic resonance imaging.
SNQ calculations from five ROI measurements were performed using an Excel spreadsheet (Microsoft, Redmond, Washington, USA). Patients were classified into four groups according to the classification of ACL remnant tissue established by Crain et al. Classification of ACL remnant tissue was performed at the time of initial arthroscopic observation just before ACL reconstruction surgery. 26 The ACL remnant tissue was considered to be Crain type 1 in cases where the injured ACL wrapped around the PCL and the normal ACL attachment on the femur was empty, Crain type 2 in cases where we observed ACL healing to the roof of the notch and the lateral wall was empty, Crain type 3 in cases where the attenuated ACL remnant tissue healed to the lateral wall more anterior and distal than its anatomical origin, and Crain type 4 in cases where the ACL remnant tissue was torn and resorption of the femoral end was observed (Figure 2). Age, time from injury to undergoing MRI (weeks), International Knee Documentation Committee (IKDC) Subjective Knee Form, Tegner Activity Scale, and preoperative side-to-side difference (SSD) using TelosTM (Telos GmbH, Laubscher, Holstein, Switzerland), 27 and the SNQ calculated using ROI at the tibial, mid, and femoral portions of the injured ACL was compared among the four groups. Patients were given the IKDC Subjective Knee Form and Tegner Activity Scale self-reported questionnaires and asked to answer on an outpatient basis by nurses who were not involved in this study. Preoperative SSD was evaluated using TelosTM. Patients were lying on the bed with their knee flexed at 20°, and 15 kg (147 N) anterior force was applied to the tibia relative to the femur. Two lines were defined. First, the tibial line was defined as tangent to the posterior edge of the medial tibial plateau and perpendicular to the line tangent to the medial tibial plateau. Second, the femoral line was defined as tangent to the posterior edge of the medial femoral condyle and parallel to the tibial line (Figure 3). The distance between these two lines was measured by a single observer who is blinded to the arthroscopic findings of ACL remnant tissue using PACS with 0.01-mm precision.

The illustration of how SSD is measured using Telos. Patients were lying on the bed with their knee flexed at 20°, and 15 kg (147 N) anterior force was applied to the tibia relative to the femur. Two lines were defined. First, the tibial line was defined as tangent to the posterior edge of the medial tibial plateau and perpendicular to the line tangent to the medial tibial plateau. Second, the femoral line was defined as tangent to the posterior edge of the medial femoral condyle and parallel to the tibial line. SSD: side-to-side difference.

There was a significant difference in preoperative SSDs among the four groups. Post hoc analysis revealed that the SSD was significantly smaller in Crain type 2 than in Crain type 4. SSD was also significantly smaller in Crain type 3 than in Crain type 4. SSD: side-to-side difference.
Statistical analysis
All data were presented as the mean and standard deviations. One-way analysis of variance with post hoc analysis was used to evaluate the differences or correlations between the four groups. The χ 2 test was used to evaluate the differences in the incidence of concomitant medial or lateral meniscus injuries. All statistical analyses were performed using EZR, 28 and the significance level was set at p < 0.05. The adequate sample size with an α error of 0.05, a β error of 0.80, and an effect size of 0.25 was calculated to be 180 using G* Power 3.1 (Franz Paul, Kiel, Germany). Power analysis revealed that the β error was 0.381 and the power was calculated to be 0.619.
Results
There were no significant differences in age, time from injury to undergoing MRI, preoperative IKDC subjective scale, and Tegner activity scale among the four groups (Table 2). There were no significant differences in the incidences of accompanying lateral meniscus lesion and chondral lesion among the four groups (p = 0.124 and 0.428, respectively). On the other hand, there were significant differences in the incidences of accompanying medial meniscus lesions among the four groups (p < 0.001), and post hoc analysis revealed that the incidence of medial meniscus lesion was significantly larger in Crain type 4 than in Crain type 2 (p < 0.001). The incidence of medial meniscus lesion was also significantly larger in Crain type 4 than in Crain type 3 (p < 0.001). However, there was no significant difference concerning the incidence of medial meniscus lesion between Crain types 2 and 3 (p = 0.816).
Results between the four groups according to Crain classification of ACL remnant tissue.a,b

ANOVA: analysis of variance; SNQ: signal/noise quotient; ACL: anterior cruciate ligament; MRI: magnetic resonance imaging; IKDC: International Knee Documentation Committee.
aData are expressed as mean (standard deviation).
bComparison between groups using one-way ANOVA.
There were significant differences in the preoperative SSDs among the four groups (p = 0.021), and post hoc analysis revealed that the SSD was significantly smaller in Crain type 2 than in Crain type 4 (p = 0.014). The SSD was also significantly smaller in Crain type 3 than in Crain type 4 (p = 0.0030). However, there was no significant difference in the SSD between Crain types 2 and 3 (p = 0.79; Figure 4).

There were significant differences in SNQs among the tibial, mid, and femoral portions in Crain type 2. Post hoc analysis revealed that the SNQ in Crain type 2 was significantly lower at the tibial portion than at the mid portion. SNQ: signal–noise quotient.
There were no significant differences in the SNQs calculated using the ROI at the tibial, mid, and femoral portions between Crain types 1 and 4. On the other hand, there were significant differences in the SNQ calculated using ROI at the tibial, mid, and femoral portions between Crain types 2 and 3 (p < 0.001; Table 3). Post hoc analysis revealed that SNQ in Crain types 2 and 3 were significantly lower at the tibial portion than at the mid portion (p = <0.05; Figures 4 and 5). Furthermore, there were significant differences in SNQs using ROI at the tibia among the four groups (p = 0.034), and post hoc analysis revealed that SNQ at the tibial portion was significantly lower in Crain types 2 and 3 than in Crain type 1 (p < 0.05; Figure 6). On the other hand, there were no significant differences in the SNQs at the femoral and mid portions among the four groups (Table 4).
Results of SNQ at three points of ACL remnant tissue.a,b

ANOVA: analysis of variance; SNQ: signal/noise quotient; ACL: anterior cruciate ligament.
aData are expressed as mean (standard deviation).
bComparison between groups using one-way ANOVA.
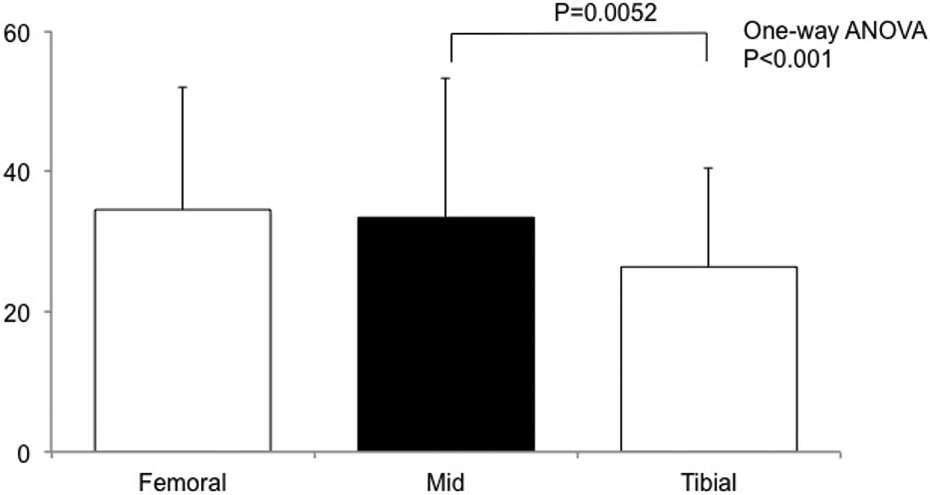
There were significant differences in the SNQs among the tibial, mid, and femoral portions in Crain type 3. Post hoc analysis revealed that the SNQ in Crain type 3 was significantly lower at the tibial portion than at the mid portion. SNQ: signal–noise quotient.

There were significant differences in the SNQs using ROIs at the tibia among the four groups, and post hoc analysis revealed that the SNQ at the tibial portion was significantly lower in Crain type 2 than in Crain type 1. Additionally, the SNQ at the tibial portion was significantly lower in Crain type 3 than in Crain type 1. SNQ: signal–noise quotient; ROI: region of interest.
Results of the SNQ at each of the three portions.a,b

ANOVA: analysis of variance; SNQ: signal/noise quotient.
aData are expressed as mean (standard deviation).
bComparison between groups using one-way ANOVA.
Discussion
In this study, SSD in Crain types 2 and 3 was significantly smaller than that in Crain type 4. Also, there were significant differences in SNQ at the tibial, mid, and femoral portion in Crain types 2 and 3. These results were in accordance with our hypothesis.
Preoperative sagittal MRI was used to assess the integrity of the ACL remnant tissue in patients with a clinical diagnosis of ACL injury and compared the preoperative MRI findings with the arthroscopic findings obtained at the time of surgery. MRI has high sensitivity and specificity in diagnosing ACL injuries. 29,30 However, despite recent interest in ACL reconstruction with remnant tissue preservation, 18,20 there is limited information about the diagnostic values of preoperative MRI for the remnant tissue. 31 Normal ACL appears as a band comprising low signal lines, 32 while injured ACL gives a high signal in the MRI, which indicates fluid and hemorrhage confined to the intact synovial sheath. 33,34 Additionally, there is a report that MRI is also considered to be an important tool for predicting the pattern of ACL remnant tissue or bundle injury pattern with satisfactory precision. Therefore, the MRI signal intensity of ACL remnant tissue is supposed to reflect its integrity. There is a report that the ACL remnant tissue of Crain types 1 or 2 contributed to anteroposterior knee stability evaluated at 30° of knee flexion for up to 1 year after injury using a KT-2000 arthrometer 35 and MRI. This study revealed that Crain types 2 and 3 ACL remnant tissue retained relatively better anterior stability than that of Crain types 1 and 4. There is a difference in testing apparatus, and it may affect the difference between that study and the present study. In addition, the SNQs at the tibial portions were significantly lower than those at the mid portions in Crain types 2 and 3. These results implied that the tibial portions of the ACL remnant tissue retained better integrity in Crain types 2 and 3 than in Crain types 1 and 4.
Tibial enthesis is of importance for the gradual increase in anterior drawer force applied to the ACL. 16 In a clinical study on ACL reconstruction with a tibial remnant tissue-preserving technique, a significant difference was detected in functional outcome and proprioception in patients with >20% tibial remnant tissue preserved. 17 Hence, there is a possibility that the preservation of a tissue with better tibial integrity in MRI is the key to better clinical outcome in ACL reconstruction with remnant tissue preservation, especially in cases of better knee stability and higher stiffness against anteroposterior drawing force.
From the results of our study and past reports, we suppose that both femoral attachment and lower SNQ at the tibia may contribute better knee stability. However, which of these contributes greater is uncertain. Therefore, this remains a point of interest in future studies.
Limitations
This study has several limitations. First, no radiologist performed interpretations of the MRI and all the calculations and measurements. But interpretations of the MRI were performed by an orthopedic specialist, authorized by the Japanese Orthopaedic Association, and well-experienced knee surgeons. Second, ROI was evaluated only from sagittal MRI and not from oblique coronal or oblique sagittal MRI in which accuracy to distinguish the anteromedial and posterolateral bundle is said to be higher. 36 It is difficult to diagnose a proximally torn ACL with a relatively little disruption that adheres to the PCL or maintains a gross alignment on the sagittal sequence. In these cases, more accurate diagnosis may be made in the axial or coronal planes. 37 Third, we did not include patients with findings of rotatory stability obtained on an outpatient basis because we could not evaluate quantitatively, 38 and the results of these procedures depend on the examiner’s experience. 39 However, the Pivot shift test with a three-dimensional electromagnetic measurement system under general anesthesia revealed that there was no significant difference between the Crain classification groups. 40 Fourth, ROI and SNQ were evaluated by a single examiner, but the examiner evaluated the MRI several times before becoming aware of the arthroscopic findings. Fifth, the time interval between injury and MRI may have been too large (8 ± 11 weeks; 0–52 weeks), and we did not investigate the chronic cases in which patients underwent MRI >1 year after injury because the biomechanical contribution of the ACL remnant tissue is supposed to be lost 1 year after injury. 26 Sixth, the examiners for imaging and arthroscopy were not blinded to the results. Seventh, this study was an underpowered study (0.619), so more data should be collected to achieve sufficient power.
Beyond these limitations, however, we believe that the results of this study may be helpful for surgeons who plan to perform ACL reconstruction with remnant tissue preservation when they use sagittal images of preoperative MRI using ROI technique to determine if the tibial ACL remnant tissue has good integrity and would be of value if preserved. Further study is required to clarify how these findings influence the postoperative stability after ACL reconstruction with remnant tissue preservation.
Conclusions
The femoral attachment of ACL remnant tissue may influence the SNQ of tibial remnant tissue and stability of the ACL-injured knee.
Footnotes
Authors’ contributions
TT had made the study design, carried out acquisition of the data, made the analysis, and interpreted the data. MK and KT had participated in conceiving the study design and revising the data and manuscript. All authors had read and approved the final manuscript.
Acknowledgment
We thank Keiichi Hagiwara, MD, PhD, Shinya Yanagisawa, MD, PhD, Atsuko Ogoshi, MD, Tomoyuki Nakagawa, MD, and Kazuhiro Katayama, MD for the initial examination and performing surgery.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
