Abstract
Background:
In patients with femoral intertrochanteric fractures treated by cephalomedullary (CM) nailing, abduction force reportedly decreased by 25–30% during the postoperative follow-up period. The purpose of the current study is to evaluate the cross-sectional area (CSA) and adipose tissue ratio (ATR) of the gluteus medius muscle on the postoperative computed tomography (CT) view, expecting this graphic study will support clinical results.
Materials and methods:
A total of 27 patients with femoral intertrochanteric fractures treated by CM femoral nail implants completed the study. The mean age at osteosynthesis was 83 years (range: 72–94 years). The mean postoperative follow-up period was 23 months. The three CT axial slice views were defined as slices A, B, and C corresponding to proximal, midway, and distal part of gluteus medius, respectively. The CSA and ATR were assessed bilaterally.
Results:
The mean and standard deviation of CSA values (mm2) between the nonoperated/ operated side were as follows: slice A: 2225.8 ± 621.2/1984.5 ± 425.8; slice B: 2145.1 ± 538.3/1854.9 ± 383.9; and slice C: 1711.0 ± 459.0/1434.5 ± 396.9 (p < 0.01 in slices A, B, and C). The mean and standard deviation of ATR values (%) from the nonoperative/ operative side were as follows: slice A: 2.8 ± 1.7/5.2 ± 3.5; slice B: 2.7 ± 1.9/4.6 ± 3.2; and slice C: 3.6 ± 3.0/4.8 ± 3.2 (p < 0.01 in slices A and B and p < 0.05 in slice C).
Conclusion:
Our image findings documented that gluteus medius is significantly changed in CSA and ATR. The damage possibly triggers decrease in muscular strength of hip abduction in the postoperative follow-up period. This measurement is objective, and needed no patient’s endurance and cooperation.
Keywords
Introduction
Abduction muscle tension is important in gait and hip biomechanics. The gluteus medius muscle plays a key role in exerting abduction forces across the hip joint and provides stabilization of the pelvis during single leg stance. 1 Insufficient functioning of the gluteus medius muscle is clinically associated with a positive Trendelenburg sign which leads to a loss of pelvic control, hence an impaired ability to walk. 2 In our study of patients with femoral intertrochanteric fractures treated by cephalomedullary (CM) nailing, muscle strength around the hip joint, especially abduction force, decreased by 25–30% during the postoperative follow-up period. 3 However, accurate measurement of maximum isometric muscle strength requires the expertise of well-trained physical therapists as patients are often elderly, are easily fatigued, and may be uncooperative. Therefore, objective data to support our clinical findings are needed, where the patient’s endurance and cooperation will not affect the measurement results.
Automated computed tomography (CT) image analysis of the cross-sectional area (CSA) and adipose tissue ratio (ATR) of skeletal muscles has been used to measure muscle strength and adaptation. 2 This method is not influenced by patient effort or motivation and may be used to visualize pathologic changes on the operative side compared to the nonoperative contralateral side. In order to investigate differences in degree of thoracic kyphosis and paraspinal muscle composition in asymptomatic and symptomatic lumbar discectomy patients, a cross-sectional CT analysis of the quality of trunk muscles was conducted. 4
Muscle density imaging by CT and muscle adiposity imaged by magnetic resonance imaging (MRI) were used to determine function-related risk factors in women with hip fractures. 5 These previous studies clarify the usefulness of automated imaging (CT and MRI) in evaluating muscle pathology. To our knowledge, there are no articles in the literature related to graphic examination of gluteus medius muscle in patients with femoral intertrochanteric fractures treated by CM nailing.
The purpose of the current study is to evaluate the postoperative muscular CSA and adiposity of the gluteus medius muscle after CM nailing in patients with femoral intertrochanteric fractures. We hypothesized that the CSA decreases and adipose tissue proliferates. This image study will support our clinical study showing postoperative decreased strength of the gluteus medius muscle after CM nailing.
Methods
Patients
A total of 116 patients with femoral intertrochanteric fractures treated by CM femoral nail implants (Targon PF nail, B. B. Aesculap, Tuttlingen, Germany) from March 2012 to September 2015 were extracted from our trauma database for CT evaluation. Exclusion criteria included: (1) <70 years old at the time of surgery; (2) <11 months follow-up period; (3) bilaterally operated hip; (4) dementia; (5) nonambulatory; (6) with existing central or spinal nerve disorder, or Parkinson’s disease as screened by the attending physicians; (7) deceased; and (8) any related pathology that may affect hip muscle strength after physical examination and radiographic assessment. After application of the exclusion criteria, 32 patients were enrolled and 27 patients completed the study.
The mean age of the 27 patients at osteosynthesis was 83 years (range: 72–94 years). Eight patients were male (30%) and 19 were female (70%). The mean postoperative follow-up period was 23 months (range: 11–51 months). The mean body weight was 47.8 kg (range: 35–68 kg), and the mean height was 151.1 cm (range: 129.5–172.0 cm). Twelve patients were operated on the right side and 15 on the left.
CM nailing was performed conventionally, and all patients received the same type of short nail implant (Targon PF nail, B. B. Aesculap) containing two screws into the femoral head. During surgery, a 16.5-mm diameter hole over the tip of greater trochanter was created in the anteroposterior view and between the anterior and posterior thirds of the greater trochanter at the lateral projection. However, this maneuver may cause possible damage to the gluteus medius muscle.
The average period of hospitalization was 2–3 months, during which the postoperative rehabilitation program was initiated, which consisted of a 40-min per day session of muscular coordination and strength training, including early weight bearing assisted by physical therapists. In addition, patients were encouraged to walk in the hospital corridors with the aid of nurses, once their physical condition is stable. Upon discharge, ambulatory patients are recommended to attend rehabilitation activity at the outpatient clinic.
CT measurements
All patients were positioned in the supine in a Phillips MX 8000 (Phillips Medical Systems, Cleveland, Ohio, USA) spiral CT-scanner with the pelvis in the neutral position. The lower limbs were placed in internal rotation so that the patella is situated in the frontal plane. Routine scout scan of the anteroposterior and lateral pelvis was taken to localize hip position. Axial images of the CT (140 kVp, Auto mA, Noise Index 13) were obtained from each patient from the area of the iliac crest to below the lesser trochanter. Slices were 10.0 mm thick at 10.0 mm intervals. The scanned images were Digital Imaging and Communications in Medicine (DICOM) formatted (512 × 512 pixels) using Advantage Work station (version 4.6; GE Health care Japan, Tokyo, Japan) and modified for quantification of CSA of muscle, bone, and fat to the nearest 0.01 cm2. A written informed consent was obtained from each patient.
Prior to CSA measurement of CT scan, examiners routinely adjusted the slice plane of the axial and coronal view with fine control of the cutting angle. Much attention was given to obtain three slices that confirms parallelism with the connecting bilateral symmetrical anatomic landmarks. The three CT slice views evaluated were defined as: (1) slice A: view connecting the bilateral anterior superior iliac spines; (2) slice C: line connecting the superior edges of both acetabulum; and (3) slice B: view midway of slices A and C (Figure 1).

Anatomical locations of the three slices on a representative skeletal 3D CT scan image. Slice A: line connecting the anterior superior iliac spines bilaterally representing the proximal area of the gluteus medius muscle. Slice C: line connecting both sides of the superior edge of the acetabulum proximal to muscular insertion. Slice B: line midway of slices A and C. CT: computed tomography.
The CSA and ATR of the gluteus medius muscle were assessed in three different slices in the axial view. Radiographic technicians were instructed to adjust pelvic rotation to allow for equal views of the bilateral obturator foramens. For added accuracy, to determine the horizontal axis of the pelvis, a reference line between both the superior edges of iliac crest was used as an anatomical landmark to allow viewing of the distal end of pelvic bone or ischial tuberosity in the same CT slice.
CSA for the gluteus medius muscle was measured bilaterally three times by three independent investigators (YT, MF, IS). All tracings of muscle border were cleared for each examiner. Mean of three measurements for each slice for each patient was calculated, and values between the operated and nonoperated side were compared statistically.
The CSA of the gluteus medius muscle was automatically calculated based on the circumferential outline after manual tracing (Figure 2). Neighboring slices were used as references to increase accuracy of tracing the muscle boundaries. Adipose tissue in and around muscle bundles within each muscle group was distinguished by a bimodal image histogram resulting from the distribution of CT numbers in adipose tissue and muscle. 6 The ATR of the gluteus medius muscle was represented as the ratio of the area of intramuscular adipose tissue (−100 to −50 in Hounsfield Units, HU) against the CSA (0–100 HU) 7,8 (Figure 3).
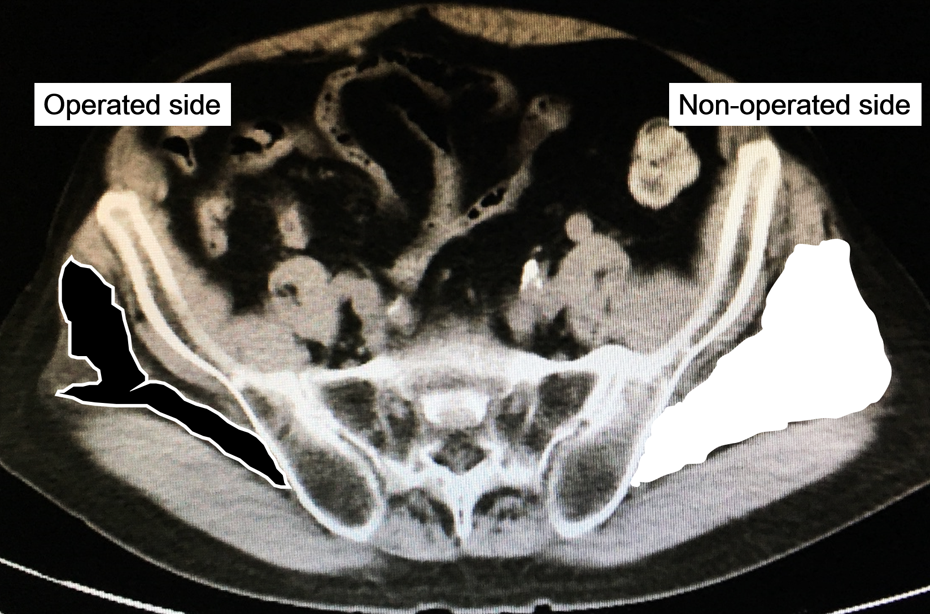
The CSA of the gluteus medius muscle is painted in black or white. The CSA in the operated side (black) is lesser than in the non-operated contralateral side (white). CSA: cross-sectional area.

Adipose tissue proliferation (white arrows) in the operated side contrasted to the nonoperated contralateral side.
Statistical analyses
All data were analyzed using the software program Statistical Package for the Social Sciences (SPSS, version 14.0, Tokyo, Japan). Statistical significance was set at p < 0.05. CSA and ATR values obtained from the gluteus medius muscle were compared with the Wilcoxon Signed Rank test to determine significant difference between the operated and nonoperated side.
Results
The mean and standard deviation of CSA values (mm2) from the nonoperated/operated side were as follows: slice A: 2225.8 ± 621.2/1984.5 ± 425.8; slice B: 2145.1 ± 538.3/1854.9 ± 383.9; and slice C: 1711.0 ± 459.0/1434.5 ± 396.9 (Table 1). CSA values measured from all slices were significantly lesser in the operative side compared to the nonoperative side (p < 0.01 in slices A, B, and C).
Values of CSA of the gluteus medius muscle at slices A to C, in the operated side and nonoperated contralateral side.a

CSA: cross-sectional area.
aThe values are given as the mean and the standard deviation. Twenty-seven CSAs of gluteus medius were analyzed. A significant difference in all slices between the nonoperated side and the operated side.
b p < 0.01.
The mean and standard deviation of ATR values (%) from the nonoperative/operative side were as follows: slice A: 2.8 ± 1.7/5.2 ± 3.5; slice B: 2.7 ± 1.9/4.6 ± 3.2; and slice C: 3.6 ± 3.0/4.8 ± 3.2 (Table 2). ATR in the CSA of the gluteus medius muscle in all slices was significantly higher in the operated side compared to the nonoperated side (p < 0.01 in slices A and B, and p < 0.05 in slice C).
Distribution of ATR in the gluteus medius muscle at slices A to C, in the operated side and nonoperated contralateral side.a

ATR: adipose tissue ratio; CSA: cross-sectional area.
aThe values are given as the mean and the standard deviation. Twenty-seven CSAs of gluteus medius were analyzed. A significant difference between the non-operated side and the operated side.
b p < 0.01.
c p < 0.05.
The interobserver repeatability of measurements was acceptable and shown as follows. Interobserver variations in measuring CSA of gluteus medius were assessed using the coefficient of variation (CV%), 0.72 (95% CI: 0.43–1.0, p < 0.01). Interobserver variations in measuring ATR of gluteus medius were calculated using the CV%, 0.68 (95% CI: 0.40–0.96, p < 0.01).
Discussion
Changes to muscles at trunks and legs after surgical procedures have been previously reported. 4,5 We hypothesized that similar changes may occur to the gluteus medius muscle on the operative side of patients with intertrochanteric fractures treated by femoral CM nailing may occur. One of the important findings is pathologic change from the proximal to the distal gluteus medius observed as both a decrease in CSA and a replacement of muscular tissue with adipose tissue. CSA is expected to be directly related to muscle volume and the radiological density of the muscle itself, which may indirectly represent muscle strength. 2 Several articles reported changes in CSA measured by CT scan imaging. Lang et al. 9 stressed that CT values of CSA correlate with measures of muscle strength and lower extremity performance. Other studies were conducted to search for factors related to CSA. 10,11 However, there are a few studies with CT scan imaging that document changes in thigh muscles between the fractured and non-fractured hip. 7 To our knowledge, there have been no reports involving CT scan examination of the gluteus medius muscle in postoperative femoral intertrochanteric fractures.
In the current study, the CSA of the gluteus medius muscle was significantly decreased among the middle and distal part, found as continued oval muscular defect, reflecting direct damage during greater trochanteric reaming. Cause of the defect is supported by the following anatomical research. McConnell et al. 12 documented that 27% of 34 cadaveric specimens had injury to the gluteus medius tendon at the junction of the greater trochanter after 17 mm of reaming, when entry point for gamma nail insertion was at the tip of the greater trochanter. This coincides with the findings reported by Sawauchi et al. 13 where examination of 10 cadaveric specimens showed a 30% damage of the gluteus medius muscle. Many surgeons have frequently noticed hollow reamer filled with certain volume of muscle belly while creating nail hole. On the other hand, Perez et al. 14 reported no damage to the gluteus medius tendon in their cadaveric study. However, the entry point was slightly medial to the tip of the greater trochanter using a 14-mm diameter reamer, which was smaller for standard CM nailing. Consequently, their results may not be applicable to conventional CM nailing procedures.
The proximal gluteus medius muscle showed changes in both CSA and ATR. Since it is away from the direct path of the intramedullary reamer, this pathologic change is indirect. In clinical and animal studies of rotator cuff tears, tendinous tears at the insertion characterized by pathological fatty infiltration, muscle atrophy, and fibrosis in proximal muscular tissues were observed. 15,16 Therefore, these observations show that tendinous damage partially induces fatty degeneration of proximal muscular tissues.
The reliability of CT scan imaging for muscle evaluation has been previously evaluated. In a cadaveric study by Mitsiopoulos et al., 17 scanned images of skeletal muscles were compared with actual anatomic evaluation, and the results strongly support the use of MRI and CT imaging as reference methods for measurement of appendicular skeletal muscle and interstitial and subcutaneous adipose tissues. Goodpaster et al. 11 also confirmed the effectiveness of repeated CT measurements in muscle evaluation and demonstrated a coefficient variability of less than 1% in radiological evaluation. In a report by Rasch et al., 18 the simplicity of circumscribing large muscle bellies and easy identification of the bony landmarks of the pelvis allow for repeated measurements, with minimal errors. In our research, CT scan imaging not MRI was selected, as MRI is not feasible due to the presence of the metallic femoral implant.
We believe it is difficult to prolong postoperative period until achieving the plateau of muscular recovery. Because the duration needed for CSA recovery depends on the cause of muscular damage. In general, muscle atrophy may be triggered by mechanical unloading (e.g. by non-weight bearing), postoperative immobilization (e.g. casting or bed rest), direct injury to the muscle, and may occur as a consequence of normal aging. 19 Several reports anticipated the decrease in CSA will recover less rapidly 18,20 and stressed that a significant increase in muscle volume may occur within 2 years after total hip arthroplasty (THA) and reach an equilibrium. Mikkola et al. 21 reported that knee extension strength and calf muscle CSA were lower on the fractured leg in a cross-sectional study among older adults 3.5-years post-hip fracture. Nevertheless, the difficulty for aged patients to continue long-term follow-up should be taken into consideration. A few years of follow-up seem difficult in some of aged patients. Thus, the minimal follow-up period was set to 1 year in this particular aged group.
There are several limitations to our current study. First, the sample size is relatively small. As this may be attributed to our stringent inclusion criteria, patients met these conditions were limited. Approximately three-fourths of patients were excluded for follow-up due to the inability to walk, deceased state, and worsening of cognitive functioning. However, the follow-up ratio marked nearly 90% among the selected patients. This figure is higher than similar study, 22 as Aspenberg et al. 23 reported a very high dropout rate and a low compliance to visits among aged patients during a 6-month follow-up period. Second, only relatively healthy ambulatory adults were recruited to the study at baseline, as the prime purpose of this study is to exclusively clarify the influence of surgery on the gluteus medius muscle. A large number of disabled patients in our trauma database (increased age and presence of neurovascular disorder) were excluded in the present investigation, as accurate measurement of the gluteus medius muscle could not be obtained. Omission of these patients may limit the power of our study to examine muscular changes among the elderly. A different study design to include these patients and to search for a general course of this operative procedure needs to be done. Third, none of our examiners were blinded to the operated side. This bias is difficult to avoid, as neighboring slices have to be evaluated in necessary cases. Fourth, we could not prove pre-injury CSA is symmetric, although a few articles showed a difference in both lower extremities. 24 The CT scans in this present study included almost equal left and right laterality and revealed approximately similar CSA between operated and nonoperated sides in gluteus maximus and minimus. Fifth, measurement is not under the same time frame. CSA defect revealed lasting damage, as a few patients in our database had similar diminished CSA of the gluteus medius muscle even after a decade of follow-up, though not included in this present study.
In conclusion, our imaging findings documented a possible cause for the decrease in muscular strength of the gluteus medius muscle occurring in the postoperative follow-up period after CM nailing. Damage to the gluteus medius tendon was deemed unavoidable with placement of the CM nail and should also be recognized as a potential cause of postoperative morbidity. Shoulder surgeons 18,25 expanded and supported our findings in that adipose infiltration is a strong negative predictor of muscle recovery. Zielinski et al. 22 predicted that diminished strength may affect older and more disabled patients, causing debilitation due to their decreased ability to compensate. Surgeons should carefully follow-up postoperative functions of patients, as suggested in this current study.
Footnotes
Acknowledgements
We thank Mr. Masahiko Imoto, a radiological technologist at Konan hospital, for his long-term contribution.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
