Abstract
Background:
Percutaneous radiofrequency ablation (RFA) has been shown to be an effective treatment for soft tissue lesions and also benign bone tumors, especially osteoid osteoma. There are limited data regarding this technique in other bone tumors, specifically larger and more aggressive ones.
Purposes:
To describe the use of RFA as a definitive treatment and an alternative to traditional open surgery for the treatment of chondromyxoid fibroma (CMF), a benign but locally aggressive bone tumor.
Case presentation:
An 11.5-year-old girl was diagnosed with a 4-cm lytic bone lesion of the distal fibula. Evaluation, including biopsy, revealed CMF. It was managed by fluoroscopy-guided RFA only. Six-year follow-up demonstrated complete healing without damage to the adjacent distal fibular growth plate.
Discussion and conclusions:
RFA induces local heat in the ablation field and causes tissue necrosis. The depth of heat penetration and the size of heated sphere are accurately controlled by modern types of ablation probes and accurate positioning. The current report demonstrates the ability to use this percutaneous technique for larger and more aggressive bone tumors than has been indicated previously.
Introduction
Chondromyxoid fibroma (CMF) is a benign cartilaginous neoplasm. Its incidence is less than 1% of all tumoral and tumorlike lesions of bone. It’s a benign but locally aggressive tumor, causing pain that would lead to radiographic detection. The typical radiographic presentation of CMF is an eccentric and well-circumscribed lesion, located in the metaphysis of long bones, which often has a sclerotic periphery. The most common locations are the proximal tibia, distal femur, foot, and pelvis. 1
The mainstay of percutaneous radiofrequency ablation (RFA) is induction of focal tissue necrosis by thermal energy deployed by the RFA electrode. Despite its advantages, RFA is not amenable to all bone tumors. Efficient ablation may not be achieved in tumors larger than 1 cm (limited sphere of penetration). Nowadays, RFA, as a single treatment, is the gold standard of care of osteoid osteoma, 2,3 achieving success rates of 73–95%. 4
The use of RFA is sometimes inadequate in tumors located in the vicinity of large vessels for reasons such as uncontrolled heat deployment and heat dissipation, also known as the “sink effect” (blood flow in the large vessels renders RFA-induced temperature drop below the target ablation temperature). 5,6 Nevertheless, RFA continues to gain recognition due to technical improvements such as probe repositioning during the same setting, employment of smaller-sized probe tips, expandable electrodes, and the use of a water-cooled probe tip. 7 –9 RFA allows shorter recovery periods as compared to surgical excisions, especially for lesions located in critical areas such as the physeal plate or articular cartilage. 10 All the above mentioned advantages revived interest in applying this technique in cartilaginous bone tumors as an alternative to surgery. The use of RFA for the treatment of chondroblastoma (a cartilaginous lesion affecting epiphysis or apophysis of long bones of teenagers) spearheaded the application of this technique in other lesions. Additional reports followed, suggesting the efficacy and safety of RFA in the treatment of other tumors such as osteoblastoma, giant-cell tumor, and enchondroma. 11 –15 At present, however, RFA is not routinely pursued for the definitive treatment of benign solid bone tumors greater than 2 cm in their largest diameter.
We hereby present our experience in the use of RFA for CMF of the distal fibula in proximity to the growth plate.
Patient presentation
An 11-year-old girl complained of right ankle pain that appeared 6 months prior to consultation and was related to a fall. Her pain limited strenuous activities such as running. Plain radiograph (Figure 1) and computed tomography (CT) (Figure 2) showed medullary lytic lesion of the distal fibula; bone scan revealed pathologically increased radionuclide uptake in the same location. Magnetic resonance imaging (MRI) demonstrated a gadolinium-enhancing 4 × 2 × 1.8 cm intramedullary lesion, surrounded by reactive periosteal sclerosis. The lesion was adjacent to the physeal growth plate and increased in size during a 3-month follow-up. The patient underwent bone biopsy through a 4-mm skin incision and bone penetration with an 8G Jamshidi-type hollow biopsy needle with CT imaging guidance. Histology diagnosed the lesion as CMF. Since the lesion was in proximity to the growth plate, there was a major concern of harming the fibula growth potential with formal surgical excision. Despite the lesion’s size (>4 cm in length), definitive RFA was offered as an alternative. She underwent RFA under general anesthesia and fluoroscopy guidance. A 20-mm outer diameter electrode with water-cooled tip electrode (Electrode Kit CT–1520, Tyco Valleylab, Boulder, Colorado, USA) was used for ablation. In order to cover the entire lesion area, heating process was performed up to 90ºC for 6 min. This was repeated in two successive locations along the lesion at the same session. The patient was discharged the following day, and pain subsided gradually within 5 days. A week after, she returned to normal daily activities, didn’t report recurrence of pain. Follow-up imaging demonstrated normal remodeling of bone and filling of the lesion. Physeal growth plate remained unharmed and the fibula attained normal length and ankle relations (Figure 3).
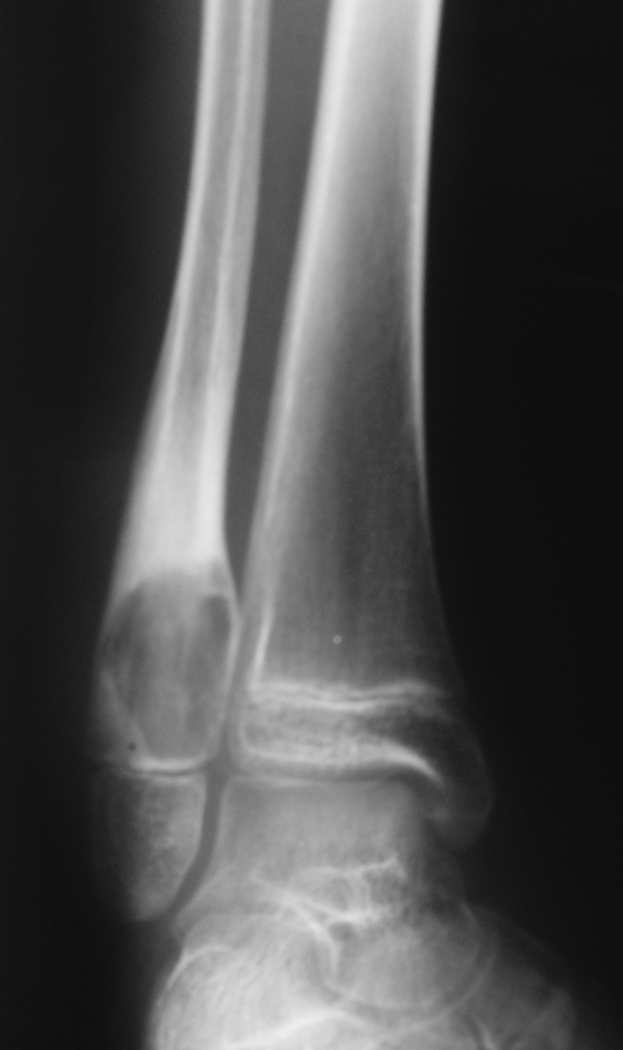
Ankle anteroposterior x-ray at diagnosis showing large lytic lesion of the distal metaphysis of fibula.

Coronal CT of the ankle showing similar findings as in the x-ray. CT: computed tomography.

Follow-up radiographs of the ankle after 1 year (a) and 6 years (b) showing gradual healing of the lesion.
Discussion
We hereby describe a case of large distal fibular CMF. It was successfully managed by RFA as an alternative to surgical tumor resection. There are no previous reports on RFA treatment of CMF.
Treatment options for CMF are surgical curettage (with and without bone grafting or polymethacrylate filling) and also en bloc excision. Curettage with or without bone grafting confers a high recurrence rate of 20% to 25%. 16,17 CMF occurring in the lateral malleolus is rare. Three other such cases have been reported. 18 –20 One underwent curettage, insertion of a wire, and polymethyl methacrylate cementation. The second underwent curettage alone (with local recurrence), and later had en bloc resection of the distal fibula, followed by reconstruction. The third underwent curettage, followed by instillation of phenol, insertion of a wire, and polymethyl methacrylate cementation.
Lately, RFA has been gaining recognition as a valid definitive treatment not only for osteoid osteoma 21 but also for other benign and malignant bone tumors such as chondroblastoma and bone metastases. RFA technique has been further adjusted to accommodate palliation of pain due to metastatic skeletal lesions, usually as a second line alternative, when radiation dose and side effects render re-irradiation not applicable. 22,23 As experience is continuously gained with standard RFA, it was noticed that tissues in the tumor surroundings are unintentionally burned and charred during the procedure due to the high temperature deployed by the probe and limiting RFA depth. 5 As this problem became evident, treatment efficacy and safety were improved by multiple repositions of the probe 24 and using a smaller outer diameter electrode of 10 mm with a manually controlled heating cycle. This allows improved lesion ablation while limiting surrounding tissue damage. This is also a major limitation of standard electrodes; their limited sphere of ablation. Larger lesions (i.e. over 4 cm) would require multiple repositioning with overlapping treatment areas in all planes. This limitation prolongs procedure time and may lead to incomplete tumor ablation. In the current case, we ended up using three electrode repositioning with good result.
Two other strategies were employed to improve controlled ablation of tissue. The first method is the use of water as a cooling source; water circulating through the probe’s tip controls the temperature at and around the tip, thus preventing its coverage by char and improving its efficacy. 25 Given the higher temperatures conducted further away from the electrode tip, a greater diameter of tissue ablation compared to non-cooled tips is achieved. This technique was successfully applied in adult osteoid osteoma ablation 26,27 and may attain larger free margins for treating tumors with high recurrence rate such as CMF. The second option is to reposition the probe in certain sites along a large lesion to ensure complete ablation coverage. To ascertain tumor eradication, the RFA plan took into account a larger ablation diameter and indeed, during the 6 years of follow-up, radiographs continued to show complete healing of the original lytic lesion with restoration of normal bone (Figure 3). Bone grafting is at high risk of infections, recurrence, fractures, pseudoarthrosis, and articular stiffness. Thirty-six percent of the patients who underwent resection and reconstruction of the distal fibula for benign or malignant tumors sustained reduced ankle mobility. 28
In the era of cost-effectiveness, percutaneous RFA has markedly reduced inpatient stay from several days following surgery with bone grafting to only a few hours. RFA also allows immediate weight bearing on the operated extremity. 10 The long visible scar carries complications such as infection and dehiscence and discoloration (RFA scar is similar in size to that of a 4 mm biopsy needle). Open surgery and curettage creates a void that has to be filled with bone graft/substitute, this is not the case with RFA where no filling is required. The reason is that the healing occurs without generating a void. In this case, follow-up by MRI and/or CT (while no bone graft has been used) is easier for interpretation.
In summary, the current report infers that RFA may be safely and successfully applied for larger cartilaginous tumors such as CMF.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
