Abstract
Purpose:
We aimed to evaluate the early clinical and radiological outcomes of arthroscopic one-stage treatment with hyaluronic acid-based cell-free scaffold (HACS) in combination with a concentration of autologous bone marrow aspirate (CBMA) technique compared to nanofracture (NF).
Methods:
A retrospective evaluation was made of all the patients with focal osteochondral lesions of the talus that were treated between January 2012 and January 2015. Ninety-eight patients met the criteria and were enrolled in the study. Forty-six patients (group 1) were treated with arthroscopic one-stage treatment with CBMA in combination with an HACS. Fifty-two patients (group 2) were treated with NF. Patient demographics and cartilage defect characteristics, the AOFAS and VAS scoring systems were compared between groups. In the evaluation of cartilage repair tissue, the magnetic resonance observation of cartilage repair tissue (MOCART) scoring system was used.
Results:
No significant differences were determined between the two groups in terms of age (p = 0.874), body mass index (p = 0.621), defect size (p = 0.485), defect depth (p = 0.674), follow-up time (p = 0.512). A significant clinical difference was determined between the two groups according to the AOFAS and VAS scores (p = 0.028, p = 0.046, respectively). The mean MOCART score of group 1 was significantly higher (p = 0.041).
Conclusions:
Both NF and HACS with CBMA techniques are beneficial in treatment of osteochondral lesions of the talus. Better clinical and radiological results, in addition to higher cartilage quality, could be obtained with HACS with CBMA technique compared to NF.
Introduction
Chondrocytes have a low capacity for regeneration; therefore, the treatment of osteochondral lesions is still challenging in orthopaedic practice. 1 There has been an increasing interest in various treatment modalities, which have been developed for osteochondral lesions in recent years and combinations of these have started to be used. 2 –4
Microfracture provides direct stimulation of mesenchymal stem cells in the subchondral bone and promotes regeneration of cartilage tissue. 5 In order to provide deeper subchondral access with smaller diameters, nanofracture (NF) method has been proposed. By this technique, main mechanical limitations of the microfracture method could be eliminated. 6
Cell-free scaffold methods have become popular with the advantages of low costs and that they allow the possibility of one-stage surgery, providing the maturation of cartilage tissue formation within the scaffold of mesenchymal stem cells, coming from the subchondral tissue. 7,8 In the current treatment of osteochondral lesions of the talus, the use of hyaluronic acid-based cell-free scaffold (HACS) can be used as an alternative method. 9 In addition, to increase the provision of mesenchymal stem cells, autologous bone marrow aspirate (CBMA) has started to be applied in combination with scaffold applications. 6 Although good results have been reported from this combination in knee cartilage defects, 10,11 to the best of our knowledge, there is no comparative study in literature, which has been conducted on talar osteochondral lesions.
In this study, it was aimed to evaluate the early clinical and radiological outcomes of arthroscopic one-stage treatment with HACS in combination with CBMA technique, compared to NF in osteochondral lesions of the talus.
Materials and methods
This was a retrospective study of patients, having focal osteochondral lesions of the talus, treated between January 2012 and January 2015.
Patients were included between the age of 18 and 45 years, surgically treated with an Outerbridge grade 3–4 lesion greater than 1.5 cm2 affecting talar dome, body mass index (BMI) < 30, with a follow-up period of at least 6 months, and who underwent a rehabilitation program. Patients were excluded who were lost in follow-up, if there was femorotibial malalignment (normal range: 0°–10°) 12 or malalignment on the axis of ankle (normal range: 88°–100°), 13 a history of previous ankle surgery, a need for a revision surgery, instability in the ankle, or a kissing lesion.
A total of 98 patients met the criteria and were enrolled in the study. All patients were operated by two individual senior surgeons (A and B).The preferred treatment method for such lesions was HACS + CBMA by surgeon A and NF by surgeon B. Thus, patients were examined in two groups as group 1 of 46 patients, arthroscopic one-stage treatment was applied with HACS (Hyalofast®, Anika Therapeutics Inc., Bedford, Massachusetts, USA), in combination with CBMA (BMAC®, Harvest Terumo, Plymouth, Massachusetts, USA), and group 2 of 52 patients, NF (Nanofx®, Arthrosurface Inc., Franklin, Massachusetts, USA) was applied.
Data of all the patients in both groups were obtained from the records and the age, BMI, defect sizes, and depth values were recorded. The patients were contacted and the AOFAS and VAS (0 = No pain, 10 = Worst pain) scoring systems were applied. For evaluation of the quality of the cartilage tissue, patients were evaluated using 3 Tesla magnetic resonance imaging (MRI) machine (Trio, Siemens, Erlangen, Germany) and a 15-channel transmit-receive birdcage knee coil. All patients were positioned with a fully outstretched leg and the ankle joint in 90° position. Sagittal planes 2D T1-weighted turbo spin-echo, 2D T2-weighted gradient-echo, 3D proton density-weighted, 3D-STIR, 3D-VIBE, 3D-T1 (dGEMRIC), and 3D-T2 mapping images were obtained. Post contrast T1-weighted and dGEMRIC images were obtained after intravenous administration of 20 ml Gd-DTPA (Magnevist, Bayer-Schering, Germany). In the evaluation of cartilage quality, according to the MRI, the magnetic resonance observation of cartilage repair tissue (MOCART) scoring system was used. 14 Patient demographics, cartilage defect characteristics, AOFAS, VAS, and MOCART scoring systems were compared between two groups.
In the standard ankle arthroscopy procedure, 60 ml of bone marrow was withdrawn from the patients of group 1 by aspiration from the posterior iliac crest into tubes and centrifuged at 3200 r/min for 15 min. At the end of the procedure, approximately 6 ml product was obtained at four- to sixfold concentration. The cartilage defects were debrided and the subchondral bone was reached. Lesion size and depth were measured in millimeters with a probe. All patients were undergone microfracture by a 30° awl prior to the application of HACS + CBMA. The scaffold was then prepared by cutting to a shape and size to completely fill the defect. By injecting approximately 3 ml bone marrow concentrate over the scaffold, it was ensured that it had spread all over. Then, the composite was placed on the defect area, arthroscopically.
In the patients in group 2, following debridement of the cartilage defects with the standard ankle arthroscopy procedure, a cannulated pick with a 15° angled tip and a 1-mm thick needle, which is advanced through the pick, was used for NF. Standardized and 9 mm deep subchondral perforations were performed with such system. 6
Approval was given by the institutional review board and informed consent was obtained from each patient.
The data were evaluated by a professional and independent statistician using SPSS 16.0 (SPSS, Inc., Chicago, Illinois, USA). The Kolmogorov–Smirnov test was used for variable groups and Student’s t-test was used for non-variable groups during statistical analysis. A level of p < 0.05 was accepted as statistically significant.
Results
The mean age of the patients was 28.8 ± 6.2 years in group 1 and 30.4 ± 7.6 years in group 2. Mean BMI was 24.2 ± 4.3 in group 1 and 25.3 ± 3.6 in group 2. The mean defect size was 1.9 ± 0.3 cm2 in group 1 and 2.1 ± 0.4 cm2 in group 2. The mean depth of the defect was 0.8 ± 0.2 cm in group 1 and 0.6 ± 0.3 cm in group 2. The mean follow-up period was 42.2 ± 9.2 months in group 1 and 40.1 ± 11.6 months in group 2. No significant differences were determined between the two groups in terms of demographic characteristics and lesion sizes (p = 0.874, p = 0.621, p = 0.885, p = 0.674, p = 0.512, respectively; Table 1).
Comparison of the mean patient demographics and cartilage defect characteristics.

BMI: body mass index.
No major complication was seen in any patient. Superficial infection developed in two patients in group 1 and this was successfully treated with oral antibiotherapy.
A significant clinical difference was determined between the two groups according to the AOFAS score (p = 0.028). The mean AOFAS score was 93.7 ± 11.6 in group 1 and 86.1 ± 7.4 in group 2. The mean VAS score was 1.3 ± 0.6 in group 1 and 0.8 ± 0.5 in group 2 and the difference was significant (p = 0.046; Table 2).
Comparison of the postoperative clinical and radiological scores of the groups.

MOCART: magnetic resonance observation of cartilage repair tissue.
A regenerated tissue with a mean T2 map was considered compatible with hyaline-like cartilage and integration of the regenerated tissue was complete in 82% of the cases in group 1 (38 of 46; Figure 1). Generally, the regenerated tissue was well organized but relatively non-homogeneous and minimally edematous. Regeneration of tissue was determined in all the patients in group 2. The structure of the repair tissue was not homogeneous, and the surface was damaged in 88% (46 of 52) of the patients in group 2 (Figure 2). Integration of the regenerated tissue to the border zone was complete in 66% (34 of 52) of the patients in group 2. The regenerative cartilage tissue was considered as fibrous repair tissue in all the patients in group 2. The mean MOCART score was 82 ± 7.1 in group 1 and 73 ± 5.4 in group 2 and the difference was statistically significant (p = 0.041; Table 2).

Coronal T2 mapping of a patient from group 1, showing regenerative tissue similar to that assumed to be indicative of healthy hyaline cartilage.
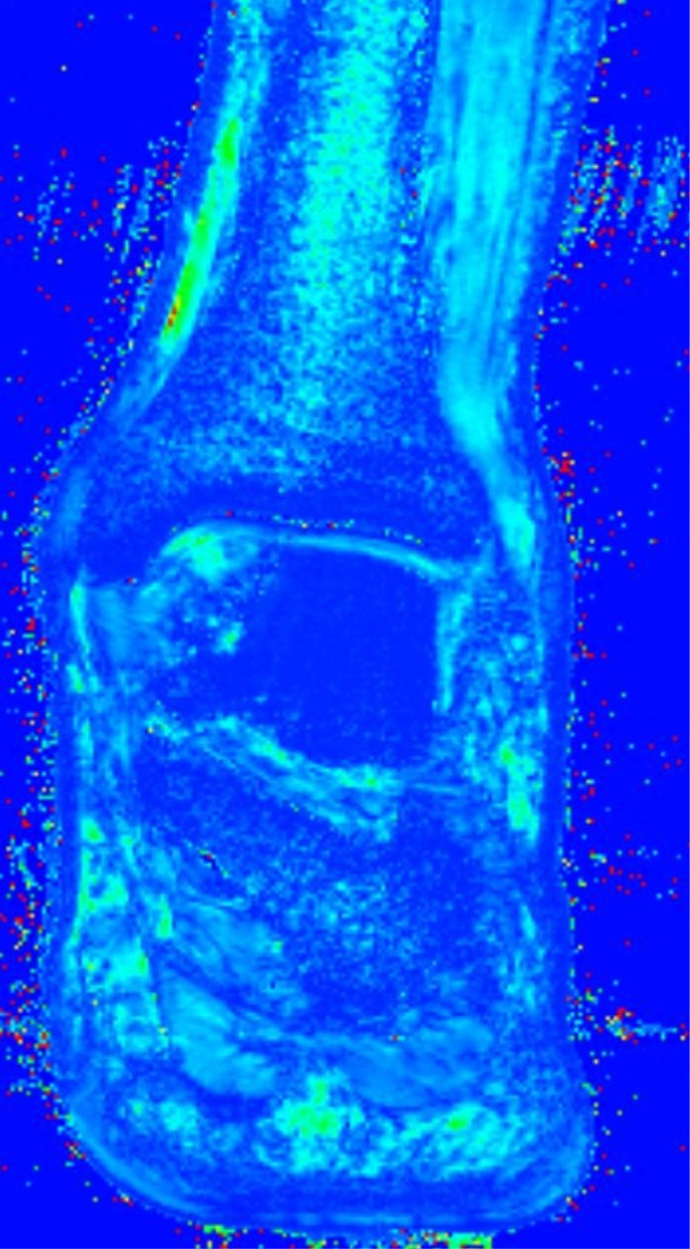
Coronal T2 mapping of a patient from group 2, showing an area of regenerative fibrocartilage tissue.
Discussion
Our results showed that both NF and HACS with CBMA provided effective pain relief and good clinical results in the treatment of talus osteochondral lesions. However, better organized hyaline like chondral tissue and higher MOCART scores could be obtained with HACS + CBMA.
The treatment of cartilage lesions is still controversial. However much autologous chondrocyte implantation reduces pain and provides clinical improvement and hyaline cartilage formation, it still requires a two-stage operation and the costs are high. Therefore, alternative treatments continue to be developed. 1,15 –17 In this context, due to the content of mesenchymal stem cells, the use of a bone marrow concentrate absorbed by the scaffold has been proposed. In a study by Buda et al., a one-stage procedure was applied using CBMA absorbed in a cell-free scaffold together with platelet-rich fibrin and in the 2-year follow-up, it was reported that together with good clinical results, 70% closure of the defects had been achieved. 18 Similarly, Giannini et al showed regenerated cartilage tissue in various degrees of remodeling (Including hyaline cartilage), following use of CBMA. 17 In a study of knee chondral lesions, after microfracture applications, CBMA absorbed in a scaffold was applied and at the end of 1 year, significant clinical recovery and the formation of hyaline-type cartilage were reported. 19 On the other hand, Zedde et al. concluded that NF is an effective and innovative repair technique, allowing deeper perforation into subchondral bone with less trabecular fragmentation and compaction when compared to microfracture. 20 In the current study, we evaluated a very specific group of patients and obtained good clinical results with both techniques, comparable to the literature. Furthermore, the clinical scores were determined to be better in the patients applied with HACS + CBMA.
Although the definitive means of determining cartilage tissue quality is by tissue biopsy, when that cannot be applied, it has been determined that MRI evaluation also gives accurate results. Marlovits et al. concluded that high-resolution MRI and well-defined MRI variables is a reliable, reproducible, and accurate tool for assessing cartilage repair tissue and they stated that they observed a good correlation of some MRI variables to the clinical outcome. 14 In a study of talar chondral lesions in 20 patients, CBMA was applied after microfracture and at the end of a 2-year follow-up period; the patients were evaluated with MRI T2 mapping. It was reported that this method is extremely successful in the determination of tissue quality and it has been reported that it is a simple, accurate, and non-invasive method that could be successfully used after regenerative surgical procedures. 21 In the current study, the regeneration tissue was evaluated with MRI and while hyaline-like cartilage tissue has been obtained in group 1 patients and the appearance in group 2 patients was consistent with a fibrous cartilage structure. Following the application of HACS + CBMA, higher MOCART values were obtained according to the cartilage formed, compared to that formed following the NF method.
The major limitations of this study were the limited number of patients, limited duration of follow-up, and the retrospective design. The outcome scores were only compared at final follow-up and no data on preoperative clinical scores. Further prospective studies are required with larger patient series and control groups with longer follow-up periods to confirm the findings of this study. In addition, if the content and structure of the cartilage tissue were to be determined by biopsy rather than MRI examination, it would add another valuable dimension to the research.
Conclusion
Both NF and HACS with CBMA techniques are beneficial in treatment of osteochondral lesions of the talus, especially considering single stage superiority. However, in comparison with NF technique, better clinical and radiological results can be obtained with HACS + CBMA.
Footnotes
Authors’ note
Study was done in Katip Celebi University, Izmir Ataturk Training and Research Hospital, Department of Orthopaedics and Traumatology and Yildirim Beyazıt University, Faculty of Medicine, Department of Orthopaedics and Traumatology.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
